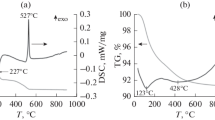
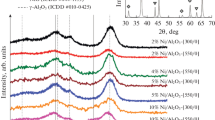
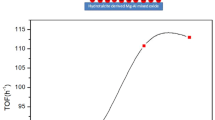
Abstract
Catalytic systems 2 wt % Pd/Al2O3 were prepared using noncalcined boehmite (NC) and two types of alumina support: one was prepared by the calcination of boehmite at 600°C (C) and the other produced by Engelhard (E). To prepare 2 wt % Pd/HPC–Al2O3 samples, these supports were modified by impregnation by a heteropoly compound (HPC) (20 wt % Н8[Si(W2O7)6] ⋅ 6Н2О). The effect of the Al2O3 structure and its modification by the heteropoly compound on the physicochemical properties, activity, selectivity and stability of catalysts in the reaction of multiphase hydrodechlorination of 1,3,5-trichlorobenzene (TCB) was studied. All catalysts showed activity in the considered reaction with the predominant formation of benzene but were deactivated in the reaction medium. Modification by the heteropoly compound resulted in increased stability and was especially effective for catalyst supported on Al2O3(E). The method of scanning electron microscopy (SEM) was used to determine the morphological differences of supports. According to the data of transmission electron microscopy, all catalysts contained palladium in the form of particles less than 20 nm in size. The particle size and width of the size distribution increases in the series Pd/Al2O3(NC) < Pd/Al2O3(C) < Pd/Al2O3(E). Modification by the heteropoly compound was favorable for the decrease in the size of palladium particles. The method of temperature-programmed reduction with hydrogen (TPR-H2) showed that all catalysts included in their composition palladium hydride along with more strongly surface-bound metal forms that are reduced at elevated temperatures, and their content decreases after modification by the heteropoly compound and increases after catalytic tests. According to diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS), the deposition of a heteropoly compound leads to a change in the type of Lewis acid sites on the alumina surface and in the electronic state of palladium. According to the results of infrared spectroscopic studies of adsorbed CO, the relatively large particles of Pd0 are the main form on the surface of nonmodified catalysts. The catalysts modified by the heteropoly compound contain single Pd+ and Pd2+ cations, and the fraction of Pd0 is substantially smaller. The specific features of the Lewis acidity of the catalyst surface determine the possibility of 1,3,5-trichlorobenzene adsorption and activation on the support and the spillover of hydrogen from Pd0. An increase in the catalyst stability as a result of support modification by the heteropoly compound can be explained by the appearance of new active sites in the interaction of palladium with the heteropoly compound or its thermal decomposition products.








Similar content being viewed by others
REFERENCES
Perosa, A., Selva, M., and Maschmeyer, T., Chemosphere, 2017, vol. 173, p. 535.
Dai, C., Zhou, Y., Peng, H., Huang, S., Qin, P., Zhang, J., Yang, Y., Luo, L., and Zhang, X., J. Ind. Eng. Chem., 2018, vol. 62, p. 106.
Zinovyev, S.S., Shinkova, N.A., Perosa, A., and Tundo, P., Appl. Catal., B, 2005, vol. 55, p. 39.
Tundo, P., Perosa, A., Selva, M., and Zinovyev, S.S., Appl. Catal., B, 2001, vol. 32, p. L1.
Cobo, M., Quintero, A., and Correa, C.M.D., Catal. Today, 2008, vols. 133–135, p. 509.
Wu, Y., Gan, L., Zhang, S., Song, H., Lu, C., Li, W., Wang, Z., Jiang, B., and Li, A., J. Hazard. Mater., 2018, vol. 356, p. 17.
Cheng, L., Jin, Z., and Wang, X., Catal. Commun., 2013, vol. 41, p. 60.
Han, B., Liu, W., Li, J., Wang, J., Zhao, D., Xu, R., and Lin, Z., Water Res., 2017, vol. 120, p. 199.
Nieto-Sandoval, J., Munoz, M., de Pedro, Z.M., and Casas, J.A., Chemosphere, 2018, vol. 213, p. 141.
De Corte, S., Sabbe, T., Hennebel, T., Vanhaecke, L., De Gusseme, B., Verstraete, W., and Boon, N., Water Res., 2012, vol. 46, p. 2718.
Wu, K., Qian, X., Chen, L., Xu, Z., Zheng, S., and Zhu, D., RSC Adv., 2015, vol. 5, p. 18 702.
El-Sharnouby, O., Boparai, H. K., Herrera, J., and O’Carroll, D.M., Chem. Eng. J., 2018, vol. 342, p. 281.
Mori, T., Kubo, J., and Morikawa, Y., Appl. Catal., A, 2004, vol. 271, p. 69.
Śrębowata, A., Juszczyk, W., Kaszkur, Z., and Karpiński, Z., Catal. Today, 2007, vol. 124, p. 28.
Diaz, E., Mohedano, A.F., Casas, J.A., Shalaby, C., Eser, S., and Rodriguez, J.J., Appl. Catal., B, 2016, vol. 186, p. 151.
Hashimoto, Y., Uemichi, Y., and Ayame, A., Appl. Catal., A, 2005, vol. 287, p. 89.
Ordóñez, S., Sastre, H., and Díez, F.V., React. Kinet. Catal. Lett., 2000, vol. 70, p. 61.
Lokteva, E.S., Erokhin, A.V., Kachevsky, S.A., Yermakov, A.Y., Uimin, M.A., Mysik, A.A., Golubina, E.V., Zanaveskin, K.L., Turakulova, A.O., and Lunin, V.V., Stud. Surf. Sci. Catal., 2010, vol. 175, p. 289.
Klokov, S.V., Lokteva, E.S., Golubina, E.V., Chernavskii, P.A., Maslakov, K.I., Egorova, T.B., Chernyak, S.A., Minin, A.S., and Konev, A.S., Appl. Surf. Sci., 2019, vol. 463, p. 395.
Babu, N.S., Lingaiah, N., Kumar, J.V., and Prasad, P.S.S., Appl. Catal., A, 2009, vol. 367, p. 70.
Golubina, E.V., Lokteva, E.S., Lunin, V.V., Telegina, N.S., Stakheev, A.Y., and Tundo, P., Appl. Catal., A, 2006, vol. 302, p. 32.
Srikanth, C.S., Kumar, V.P., Viswanadham, B., and Chary, K.V.R., Catal. Commun., 2011, vol. 13, p. 69.
Navalikhina, M.D., Kavalerskaya, N.E., Lokteva, E.S., Peristyi, A.A., Golubina, E.V., and Lunin, V.V., Russ. J. Phys. Chem. A, 2012, vol. 86, p. 1792.
Tundo, P., Perosa, A., and Zinovyev, S., J. Mol. Catal. A: Chem., 2003, vols. 204–205, p. 747.
Mrabet, D., Vu, M.-H., Kaliaguine, S., and Do, T.-O., J. Colloid Interface Sci., 2017, vol. 485, p. 144.
Sepúlveda, J.H. and Fígoli, N.S., Appl. Surf. Sci., 1993, vol. 68, p. 257.
Boudart, M. and Hwang, H.S., J. Catal., 1975, vol. 39, p. 44.
Zheng, Q., Farrauto, R., and Deeba, M., Catalysts, 2015, vol. 5, p. 1797.
Bhogeswararao, S. and Srinivas, D., J. Catal., 2015, vol. 327, p. 65.
Hurst, N.W., Gentry, S.J., Jones, A., and McNicol, B.D., Catal. Rev., 1982, vol. 24, p. 233.
Jin, H., Yi, X., Sun, X., Qiu, B., Fang, W., Weng, W., and Wan, H., Fuel, 2010, vol. 89, p. 1953.
Morterra, C. and Magnacca, G., Catal. Today, 1996, vol. 27, p. 497.
Morterra, C., Bolis, V., and Magnacca, G., Langmuir, 1994, vol. 10, p. 1812.
Busca, G., Finocchio, E., and Escribano, V.S., Appl. Catal., B, 2012, vols. 113–114, p. 172.
Barrera, A., Montoya, J.A., del Angel, P., Navarrete, J., Cano, M.E., Tzompantzi, F., and López-Gaona, A., J. Phys. Chem. Solids, 2012, vol. 73, p. 1017.
Liu, C., Zhang, C., Sun, S., Liu, K., Hao, S., Xu, J., Zhu, Y., and Li, Y., ACS Catal., 2015, vol. 5, p. 4612.
Wu, X., Zhang, L., Weng, D., Liu, S., Si, Z., and Fan, J., J. Hazard. Mater., 2012, vols. 225–226, p. 146.
Binet, C., Jadi, A., and Lavalley, J.-C., J. Chim. Phys., 1989, vol. 86, p. 451.
Bourguignon, B., Carrez, S., Dragnea, B., and Dubost, H., Surface Sci., 1998, vol. 418, p. 171.
Bertarione, S., Scarano, D., Zecchina, A., Johánek,V., Hoffmann, J., Schauermann, S., Frank, M.M., Libuda, J., Rupprechter, G., and Freund, H.-J., J. Phys. Chem. B, 2004, vol. 108, p. 3603.
Lear, T., Marshall, R., Antonio Lopez-Sanchez, J., Jackson, S.D., Klapötke, T.M., Bäumer, M., Rupprechter, G., Freund, H.-J., and Lennon, D., J. Chem. Phys., 2005, vol. 123, p. 174 706.
Clarke, J.K.A., Farren, G.M., and Rubalcava, H.E., J. Phys. Chem., 1967, vol. 71, p. 2376.
van Hardeveld, R. and Hartog, F., Adv. Catal., 1972, vol. 22, p. 75.
Hadjiivanov, K.I. and Vayssilov, G.N., Adv. Catal., 2002, vol. 47, p. 307.
Föttinger, K., Emhofer, W., Lennon, D., and Rupprechter, G., Top. Catal., 2017, vol. 60, p. 1722.
Föttinger, K., Schlögl, R., and Rupprechter, G., Chem. Commun., 2008, p. 32.
Davydov, A.A., IK-spektroskopiya v khimii poverkhnosti okislov (IR Spectroscopy in the Chemistry of Oxide Surfaces), Novosibirsk: Nauka, 1984.
Coq, B., Ferrat, G., and Figueras, F., J. Catal., 1986, vol. 101, p. 434.
Panias, D., Asimidis, P., and Paspaliaris, I., Hydrometall., 2001, vol. 59, p. 15.
Yuan, G. and Keane, M.A., J. Catal., 2004, vol. 225, p. 510.
de Pedro, Z.M., Diaz, E., Mohedano, A.F., Casas, J.A., and Rodriguez, J.J., Appl. Catal., B, 2011, vol. 103, p. 128.
Gómez-Sainero, L.M., Seoane, X.L., Fierro, J.L.G., and Arcoya, A., J. Catal., 2002, vol. 209, p. 279.
Tarlani, A., Abedini, M., Khabaz, M., and Amini, M.M., J. Colloid Interface Sci., 2005, vol. 292, p. 486
Pizzio, L.R., Cáceres, C.V., and Blanco, M.N., Appl. Catal., A, 1998, vol. 167, p. 283.
Liu, L., Wang, B., Du, Y., Zhong, Z., and Borgna, A., Appl. Catal., B, 2015, vols. 174–175, p. 1.
Hoang-Van, C. and Zegaoui, O., Appl. Catal., A, 1997, vol. 164, p. 91.
Srinivas, S.T., Jhansi Lakshmi, L., Lingaiah, N., Sai Prasad, P.S., and Kanta Rao, P., Appl. Catal., A, 1996, vol. 135, p. 201.
FUNDING
This work was performed on the state assignment “Catalysis and Physical Chemistry of the Surface” AAAA-A16-116092810057-8. The authors acknowledge support from Lomonosov Moscow State University Program of Development.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by Andrey Zeigarnik
Rights and permissions
About this article
Cite this article
Golubina, E.V., Lokteva, E.S., Gurbanova, U.D. et al. Multiphase Hydrodechlorination of 1,3,5-Trichlorobenzene on Palladium Catalysts Supported on Alumina: Effect of the Support Properties and Modification by Heteropoly Acid Based on Silicon and Tungsten. Kinet Catal 60, 297–314 (2019). https://doi.org/10.1134/S0023158419030066
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158419030066