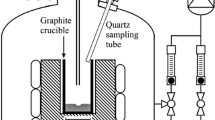
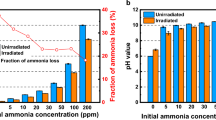
Abstract
A mathematical model has been developed for the radiolysis of an aqueous solution containing the VVER primary coolant components ammonia and boric acid. The test results show that the average deviation of the model calculation results from experimental data does not exceed 35% when processing about 250 experimental data points. This model correctly predicts the temporal evolution of the VVER coolant quality indicators (concentrations of H2, O2, and NH3) and can be used to analyze their behavior at various parameters of the VVER core (temperature, power, boron content, etc.).






Similar content being viewed by others
REFERENCES
Aliev, T.N., Evdokimov, I.A., Likhanskii, V.V., and Makhin, V.M., Vopr. At. Nauki Tekh., 2009, no. 25, p. 96.
Pikaev, A.K., Kabakchi, S.A., and Makarov, I.E., Vysokotemperaturnyi radioliz vody i vodnykh rastvorov (High-Temperature Radiolysis of Water and Aqueous Solutions), Moscow: Energoatomizdat, 1988.
Pikaev, A.K., Usp. Khim., 1960, vol. 29, no. 4, p. 508.
Bulanov, A.V., Kolesov, B.I., Lukashenko, M.L., et al., At. Energ., 2000, vol. 88, no. 5, p. 353.
Dmitriev, M.T., Zh. Prikl. Khim., 1963, vol. 36, p. 1123.
Etoh, Y., Karasawa, H., Ibe, E., et al., J. Nucl. Sci. Technol., 1987, vol. 24, no. 8, p. 672.
Dwibedy, P., Kishore, K., Dey, G.R., and Moorthy, P.N., Radiat. Phys. Chem., 1996, vol. 48, no. 6, p. 743.
Buxton, G.V. and Steuart, C.R., J. Chem. Soc., Faraday Trans., 1996, vol. 92, no. 9, p. 1519.
Buxton, G.V. and Steuart, C.R., J. Chem. Soc., Faraday Trans., 1997, vol. 93, no. 8, p. 1535.
Buxton, G.V. and Lynch, D.A., Phys. Chem. Chem. Phys., 1999, vol. 1, p. 3293.
Buxton, G.V. and Sims, H.E., Phys. Chem. Chem. Phys., 2000, vol. 2, p. 4941.
Dey, G.R., Radiat. Phys. Chem., 2011, vol. 80, no. 3, p. 394.
www.researchgate.net/publication/332875887. Accessed August 31, 2020.
Kabakchi, S.A., Matematicheskoe modelirovanie radiatsionnogo vozdeistviya na vodnye teplonositeli yadernykh energeticheskikh ustanovok. Uchebnoe posobie (Mathematical Modeling of Radiation Effect on Water-Based Coolants of Nuclear Power Plants: A Teaching Guide), Moscow: NITs Kurchatovskii Institut, 2018.
Buxton, G.V. and Sellers, R.M., Radiat. Phys. Chem., 1987, vol. 29, no. 2, p. 137.
Habersbergerova, A. and Bartonicek, B., Nukleonika, 1981, vol. 26, nos. 7–8, p. 783.
Galanin, M.P. and Konev, S.A., On one of the numerical methods for solving ordinary differential equations, Preprint of Keldysh Inst. for Applied Mathematics, Moscow, 2017, no. 18. https://doi.org/10.20948/prepr-2017-18
Chenier, M.P. and Ouellette, D.C., J. Chem. Soc., Faraday Trans., 1993, vol. 89, no. 8, p. 1193.
Sunaryo, G.R., Katsumura, Y., Hiroishi, D., and Ishigure, K., Radiat. Phys. Chem., 1995, no. 1, p. 131.
Byakov, V.M. and Nichiporov, F.G., Radioliz vody v yadernykh reaktorakh (Radiolysis of Water in Nuclear Reactors), Moscow: Energoatomizdat, 1990.
http://kinetics.nist.gov/solution. Accessed August 31, 2020.
Romanovskii, B.V., Osnovy khimicheskoi kinetiki (Fundamentals of Chemical Kinetics), Moscow: Ekzamen, 2006.
Hickel, B. and Sehested, K., Radiat. Phys. Chem., 1992, vol. 39, no. 4, p. 355.
Buxton, G.V. and Lynch, D.A., J. Chem. Soc., Faraday Trans., 1998, vol. 94, p. 3271.
Kabakchi, S.A. and Bulgakova, G.P., Radiatsionnaya khimiya v yadernom toplivnom tsikle (Radiation Chemistry in Nuclear Fuel Cycle), Moscow: RKhTU im. D.I. Mendeleeva, 1996.
Pagsberg, P.B., in Aspects of Research at Risø: A Collection of Papers Dedicated to Professor T. Bjerge on His Seventieth Birthday, No. 256 of Series Risø-R Reports, Roskilde: Forskningscenter Risø, 1972, p. 209.
Strehlow, H. and Wagner, I., Z. Phys. Chem., 1982, vol. 132, no. 2, p. 151.
Simic, M. and Hayon, E., J. Am. Chem. Soc., 1971, vol. 93, no. 23, p. 5982.
Halpern, J. and Rabani, J., J. Am. Chem. Soc., 1966, vol. 88, no. 4, p. 699.
Czapski, G. and Peled, E., Isr. J. Chem., 1968, vol. 6, no. 4, p. 421.
Elliot, A.J., Radiat. Phys. Chem., 1989, vol. 34, no. 5, p. 753.
Schwarz, H.A., J. Phys. Chem., 1991, vol. 95, no. 17, p. 6697.
Chen, R., Avotinsh, Y., and Freeman, G.R., Can. J. Chem., 1994, vol. 72, no. 4, p. 1083.
Gordeev, A.V., Extended Abstract of Cand. Sci. (Chem.) Dissertation, Moscow: Inst. of Physical Chemistry, 1994.
Men’kin, V.B., Makarov, I.E., and Pikaev, A.K., Khim. Vys. Energ., 1991, vol. 25, no. 1, p. 60.
Stief, L.J. and Payne, W.A., J. Chem. Phys., 1976, vol. 64, no. 12, p. 4892.
Wellman, C.R., Ward, J.R., and Kuhn, L.P., J. Am. Chem. Soc., 1976, vol. 98, no. 7, p. 1683.
Ishida, K., Wada, Y., Tachibana, M., et al., J. Nucl. Sci. Technol., 2006, vol. 43, no. 1, p. 65.
Macdonald, R.G. and Miller, O.A., Radiat. Phys. Chem., 1985, vol. 26, no. 1, p. 63.
Gurvich, L.G., Karachevtsev, G.V., Kondrat’ev, V.N., et al., Energii razryva khimicheskikh svyazei, potentsialy ionizatsii i srodstvo k elektronu (Chemical Bond Energies, Ionization Potentials and Electron Affinity), Moscow: Nauka, 1974.
Egorov, Yu.A., Osnovy radiatsionnoi bezopasnosti atomnykh elektrostantsii (Basics of Radiation Safety of Nuclear Power Plants), Moscow: Energoizdat, 1982.
Hochanadel, C.J., J. Phys. Chem., 1952, vol. 56, no. 5, p. 587.
Schwarz, H.A., J. Phys. Chem., 1962, vol. 66, no. 2, p. 255.
Hayon, E., Trans. Faraday Soc., 1967, vol. 60, p. 1059.
Hart, E.J., McDonell, W.R., and Gordon, S., Proceedings of International Conference on the Peaceful Uses of Atomic Energy, Geneva: United Nations, 1955, no. 7, p. 597.
Belloni, J. and Haissinsky, M., Int. J. Radat. Phys. Chem., 1959, vol. 1, p. 519.
STO (Industry Standard) 1.1.1.03.004.0980-2014: Water Chemistry of the Primary Circuit at the Unit Commissioning Stage of Project AES-2006 Nuclear Power Plant: Coolant Quality Standards and Means of Ensuring Them, Moscow: Kontsern Rosenergoatom, 2014.
Christensen, H., Molander, A., Lassing, A., et al., Water Chemistry of Nuclear Reactor Systems (Proc. 7th Int. Conf. Bournemouth, 1996), London: BNES, 1996, vol. 1, p. 138.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by S. Zatonsky
Rights and permissions
About this article
Cite this article
Grachev, V.A., Sazonov, A.B. Radiolysis of Aqueous Ammonia Solutions: Mathematical Modeling. High Energy Chem 55, 472–481 (2021). https://doi.org/10.1134/S0018143921060072
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0018143921060072