Abstract
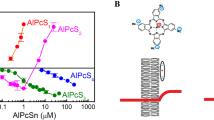
The impact of double bonds in fatty acyl tails of unsaturated lipids on the photodynamic inactivation of ion channels formed by the pentadecapeptide gramicidin A in a planar bilayer lipid membrane was studied. The presence of unsaturated acyl tails protected gramicidin A against photodynamic inactivation, with efficacy depending on the depth of a photosensitizer in the membrane. The protective effect of double bonds was maximal with membrane-embedded chlorin e6-monoethylenediamine monoamide dimethyl ester, and minimal — in the case of water-soluble tri-sulfonated aluminum phthalocyanine (AlPcS3) known to reside at the membrane surface. By contrast, the protective effect of the hydrophilic singlet oxygen scavenger ascorbate was maximal for AlPcS3 and minimal for amide of chlorin e6 dimethyl ester. The depth of photosensitizer position in the lipid bilayer was estimated from the quenching of photosensitizer fluorescence by iodide. Thus, the protective effect of a singlet oxygen scavenger against photodynamic inactivation of the membrane-inserted peptide is enhanced upon location of the photosensitizer and scavenger molecules in close vicinity to each other.
Similar content being viewed by others
Abbreviations
- AlPcS3 :
-
tri-sulfonated aluminum phthalocyanine
- BLM:
-
bilayer lipid membrane
- DOPC:
-
dioleoylphosphatidylcholine
- DPhPC:
-
diphytanoylphosphatidylcholine
- PDA:
-
photodynamic action
References
Krasnovsky, A. A., Jr. (1998) Singlet molecular oxygen in photobiochemical systems: IR phosphorescence studies, Membr. Cell Biol., 12, 665–690.
Ehrenberg, B., Anderson, J. L., and Foote, C. S. (1998) Kinetics and yield of singlet oxygen photosensitized by hypericin in organic and biological media, Photochem. Photobiol., 68, 135–140.
Kearns, D. R. (1971) Physical and chemical properties of singlet molecular oxygen, Chem. Rev., 71, 395–427.
Lavi, A., Weitman, H., Holmes, R. T., Smith, K. M., and Ehrenberg, B. (2002) The depth of porphyrin in a membrane and the membrane’s physical properties affect the photosensitizing efficiency, Biophys. J., 82, 2101–2110.
Bronshtein, I., Afri, M., Weitman, H., Frimer, A. A., Smith, K. M., and Ehrenberg, B. (2004) Porphyrin depth in lipid bilayers as determined by iodide and parallax fluorescence quenching methods and its effect on photosensitizing efficiency, Biophys. J., 87, 1155–1164.
Dror, S. B., Bronshtein, I., Garini, Y., O’Neal, W. G., Jacobi, P. A., and Ehrenberg, B. (2009) The localization and photosensitization of modified chlorin photosensitizers in artificial membranes, Photochem. Photobiol. Sci., 8, 354–361.
Lissi, E. A., Encinas, M. V., Lemp, E., and Rubio, M. A. (1993) Singlet oxygen O2(1-Delta-G) bimolecular processes — solvent and compartmentalization effects, Chem. Rev., 93, 699–723.
Wilkinson, F., Helman, W. P., and Ross, A. B. (1995) Rate constants for the decay and reactions of the lowest electronically excited singlet-state of molecular-oxygen in solution — an expanded and revised compilation, J. Phys. Chem. Ref. Data, 24, 663–1021.
Davies, M. J. (2005) The oxidative environment and protein damage, Biochim. Biophys. Acta, 1703, 93–109.
Giulivi, C., Sarcansky, M., Rosenfeld, E., and Boveris, A. (1990) The photodynamic effect of rose bengal on proteins of the mitochondrial inner membrane, Photochem. Photobiol., 52, 745–751.
Moreno, G., Poussin, K., Ricchelli, F., and Salet, C. (2001) The effects of singlet oxygen produced by photodynamic action on the mitochondrial permeability transition differ in accordance with the localization of the sensitizer, Arch. Biochem. Biophys., 386, 243–250.
Rodrigues, T., De Franca, L. P., Kawai, C., De Faria, P.A., Mugnol, K. C., Braga, F. M., Tersariol, I. L., Smaili, S. S., and Nantes, I. L. (2007) Protective role of mitochondrial unsaturated lipids on the preservation of the apoptotic ability of cytochrome c exposed to singlet oxygen, J. Biol. Chem., 282, 25577–25587.
Strassle, M., and Stark, G. (1992) Photodynamic inactivation of an ion channel: gramicidin A, Photochem. Photobiol., 55, 461–463.
Rokitskaya, T. I., Antonenko, Y. N., and Kotova, E. A. (1993) The interaction of phthalocyanine with planar lipid bilayers - photodynamic inactivation of gramicidin channels, FEBS Lett., 329, 332–335.
Kunz, L., Zeidler, U., Haegele, K., Przybylski, M., and Stark, G. (1995) Photodynamic and radiolytic inactivation of ion channels formed by gramicidin A: oxidation and fragmentation, Biochemistry, 34, 11895–11903.
Rokitskaya, T. I., Antonenko, Y. N., and Kotova, E. A. (1997) Effect of the dipole potential of a bilayer lipid membrane on gramicidin channel dissociation kinetics, Biophys. J., 73, 850–854.
Rokitskaya, T. I., Block, M., Antonenko, Y. N., Kotova, E. A., and Pohl, P. (2000) Photosensitizer binding to lipid bilayers as a precondition for the photoinactivation of membrane channels, Biophys. J., 78, 2572–2580.
Shapovalov, V. L., Rokitskaya, T. I., Kotova, E. A., Krokhin, O. V., and Antonenko, Y. N. (2001) Effect of fluoride anions on gramicidin photoinactivation sensitized by sulfonated aluminum phthalocyanines, Photochem. Photobiol., 74, 1–7.
Antonenko, Yu. N., Kotova, E. A., and Rokitskaya, T. I. (2005) Photodynamic action as a basis for relaxation method of the study of gramicidin channels, Biol. Membr. (Moscow), 22, 275–289.
Pashkovskaya, A. A., Sokolenko, E. A., Sokolov, V. S., Kotova, E. A., and Antonenko, Y. N. (2007) Photodynamic activity and binding of sulfonated metallophthalocyanines to phospholipid membranes: contribution of metal-phosphate coordination, Biochim. Biophys. Acta, 1768, 2459–2465.
Pashkovskaya, A. A., Maizlish, V. E., Shaposhnikov, G. P., Kotova, E. A., and Antonenko, Y. N. (2008) Role of electrostatics in the binding of charged metallophthalocyanines to neutral and charged phospholipid membranes, Biochim. Biophys. Acta, 1778, 541–548.
Antonenko, Y. N., Kotova, E. A., Omarova, E. O., Rokitskaya, T. I., Ol’shevskaya, V. A., Kalinin, V. N., Nikitina, R. G., Osipchuk, J. S., Kaplan, M. A., Ramonova, A. A., Moisenovich, M. M., Agapov, I. I., and Kirpichnikov, M. P. (2014) Photodynamic activity of the boronated chlorin e6 amide in artificial and cellular membranes, Biochim. Biophys. Acta, 1838, 793–801.
Rokitskaya, T. I., Kotova, E. A., Agapov, I. I., Moisenovich, M. M., and Antonenko, Y. N. (2014) Unsaturated lipids protect the integral membrane peptide gramicidin A from singlet oxygen, FEBS Lett., 588, 1590–1595.
Moisenovich, M. M., Ol’shevskaya, V. A., Rokitskaya, T. I., Ramonova, A. A., Nikitina, R. G., Savchenko, A. N., Tatarskiy, V. V., Jr., Kaplan, M. A., Kalinin, V. N., Kotova, E. A., Uvarov, O. V., Agapov, I. I., Antonenko, Y. N., and Shtil, A. A. (2010) Novel photosensitizers trigger rapid death of malignant human cells and rodent tumor transplants via lipid photodamage and membrane permeabilization, PLoS One, 5, E127–7.
Mueller, P., Rudin, D. O., Tien, H. T., and Wescott, W. C. (1963) Methods for the formation of single bimolecular lipid membranes in aqueous solution, J. Phys. Chem., 67, 534–535.
Rokitskaya, T. I., Antonenko, Y. N., and Kotova, E. A. (1996) Photodynamic inactivation of gramicidin channels: a flash-photolysis study, Biochim. Biophys. Acta, 1275, 221–226.
Pashkovskaya, A., Kotova, E., Zorlu, Y., Dumoulin, F., Ahsen, V., Agapov, I., and Antonenko, Y. (2010) Light-triggered liposomal release: membrane permeabilization by photodynamic action, Langmuir, 26, 5726–5733.
Kotova, E. A., Kuzevanov, A. V., Pashkovskaya, A. A., and Antonenko, Y. N. (2011) Selective permeabilization of lipid membranes by photodynamic action via formation of hydrophobic defects or pre-pores, Biochim. Biophys. Acta, 1808, 2252–2257.
Ytzhak, S., and Ehrenberg, B. (2014) The effect of photodynamic action on leakage of ions through liposomal membranes that contain oxidatively modified lipids, Photochem. Photobiol., 90, 796–800.
Kuznetsova, N. A., Gretsova, N. S., Derkacheva, V. M., Kaliya, O. L., and Lukyanets, E. A. (2003) Sulfonated phthalocyanines: aggregation and singlet oxygen quantum yield in aqueous solutions, J. Porphyr. Phthalocyanines, 7, 147–154.
Zorin, V., Michalovsky, I., Zorina, T., and Khludeyev, I. (1996) The distribution of chlorin e6 derivatives in biological systems — investigation of pH-effect, Proc. SPIE, 2625, 146–155.
Vilensky, A., and Feitelson, J. (1999) Reactivity of singlet oxygen with tryptophan residues and with melittin in liposome systems, Photochem. Photobiol., 70, 841–846.
Sokolov, V. S., Block, M., Stozhkova, I. N., and Pohl, P. (2000) Membrane photopotential generation by interfacial differences in the turnover of a photodynamic reaction, Biophys. J., 79, 2121–2131.
Sokolov, V. S., and Pohl, P. (2009) Membrane transport of singlet oxygen monitored by dipole potential measurements, Biophys. J., 96, 77–85.
Krasnovsky, A. A., Kagan, V. E., and Minin, A. A. (1983) Quenching of singlet oxygen luminescence by fatty acids and lipids - contribution of physical and chemical mechanisms, FEBS Lett., 155, 233–236.
Bamberg, E., and Lauger, P. (1973) Channel formation kinetics of gramicidin A in lipid bilayer membranes, J. Membr. Biol., 11, 177–194.
Nagle, J. F., and Tristram-Nagle, S. (2000) Structure of lipid bilayers, Biochim. Biophys. Acta, 1469, 159–195.
Rose, R. C. (1987) Solubility properties of reduced and oxidized ascorbate as determinants of membrane permeation, Biochim. Biophys. Acta, 924, 254–256.
Moan, J., and Berg, K. (1991) The photodegradation of porphyrins in cells can be used to estimate the lifetime of singlet oxygen, Photochem. Photobiol., 53, 549–553.
Krasnovsky, A. A., Jr. (2006) Photodynamic regulation of biological processes: primary mechanisms, in Problems of Regulation in Biological Systems (Rubin, A. B., ed.) Moscow-Izhevsk.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T. I. Rokitskaya, A. M. Firsov, E. A. Kotova, Y. N. Antonenko, 2015, published in Biokhimiya, 2015, Vol. 80, No. 6, pp. 882–890.
Rights and permissions
About this article
Cite this article
Rokitskaya, T.I., Firsov, A.M., Kotova, E.A. et al. Photodynamic inactivation of gramicidin channels in bilayer lipid membranes: Protective efficacy of singlet oxygen quenchers depends on photosensitizer location. Biochemistry Moscow 80, 745–751 (2015). https://doi.org/10.1134/S0006297915060097
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297915060097