Abstract
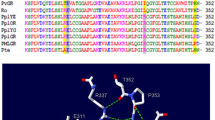
Firefly luciferases display a typical change in bioluminescence color to red at acidic pH, high temperatures and in the presence of heavy metals. Recently, the proton and metal sensing site responsible for the pH-sensitivity of firefly luciferases, which involves the salt bridges between E311-R337 and H310-E354, was identified. However, it is unclear what other residues contribute to the distinct degrees of pH-sensitivity observed in other firefly luciferases. A multialignment of primary structures of a large set of pH-sensitive and pH-insensitive beetle luciferases showed that the conserved E270 among adult firefly luciferases is substituted by Gly (railroad worms)/Gln (click-beetles) in pH-insensitive ones. Site-directed mutagenesis studies using Macrolampis sp2 and Amydetes vivianii firefly luciferases indeed showed that E270 is important for the pH-dependent activity and spectral profiles: the substitution E270A/G drastically decreases the spectral pH-sensitivity, and extends the activity profile above pH 9.0. These mutations also decrease the sensitivity to metals such as zinc, mercury and cadmium. Modelling studies showed that the residue E270 is located in a three-glutamate motif (269EEE271) at the N-terminal of α-helix-10. The results suggest that at acidic pH, the protonation of E270 carboxylate may extend a turn of the helix at the N-terminal, misaligning the pH-sensor and luciferin phenolate binding site residues: S286, I288 and E311. In contrast, the substitution of E270A/G may unwind a turn of the α-helix-10, indirectly increasing the interaction of the pH-sensor and other residues at the bottom of the luciferin binding site, stabilizing the green light emitting conformation.
Similar content being viewed by others
References
K. V. Wood, The chemical mechanism and evolutionary development of beetle bioluminescence, Photochem. Photobiol. Sci. 1995 62 662
V. R. Viviani, The origin, diversity and structure function relationships of insect luciferases, Cell. Mol. Life Sci. 2002 59 1833
V. R. Viviani and E. J. H. Bechara, Bioluminescence of Brazilian fireflies (Coleoptera: Lampyridae): spectral distribution and pH effect on luciferase-elicited colors. Comparison with elaterid and phengodid luciferases, Photochem. Photobiol. Sci. 1995 62 490
A. Roda, P. Pasini, M. Mirasole, E. Michelini and M. Guardigli, Biotechnological application of bioluminescence and chemiluminescence, Trends Biotechnol. 2004 22 295
V. R. Viviani and Y. Ohmiya, Beetle luciferases: Colorful lights on biological processes and diseases of referencing Photoproteins in Bioanalysis, ed. S. Daunert and S. K. Deo, Wiley, New York, 2006, vol. 3, pp. 49–60
G. V. M. Gabriel and V. R. Viviani, Novel application of pH-sensitive firefly luciferases as dual reporter genes for simultaneous ratiometric analysis of intracellular pH and gene expression/location, Photochem. Photobiol. Sci. 2014 14 1661–1670
G. V. M. Gabriel, R. Yasuno, Y. Ohmiya and V. Viviani, Novel application of Macrolampis sp2 firefly luciferase for intracellular pH-biosensing in mammalian cells, Photochem. Photobiol. Sci. 2019 18 1212–1217
J. R. de Wet, K. V. Wood, D. R. Helinsky and M. DeLuca, Cloning of firefly luciferase cDNA and expression of active luciferase in Escherichia coli, Proc. Natl. Acad. Sci. U. S. A. 1985 82 7870
H. Tatsumi, T. Masuda, N. Kajiyama and E. Nakano, Luciferase cDNA from Japanese firefly Luciola cruciata: cloning, structure and expression in E. coli, J. Biolumin. Chemilumin. 1989 3(2) 75–78
H. Tatsumi, N. Kajiyama and E. Nakano, Molecular cloning and expression in Escherichia coli of a cDNA clone encoding luciferase of a firefly, Luciola lateralis, Biochim. Biophys. Acta 1992 1131 161
J. H. Devine, G. D. Kutuzova, V. A. Green, N. N. Ugarova and T. O. Baldwin, Luciferase f9om the East European firefly Luciola mingrelica: cloning and nucleotide sequence of cDNA, overexpression in E. coli and purification of the enzyme, Biochim. Biophys. Acta 1993 1173 121
Y. Ohmiya, N. Ohba, H. Toh and F. I. Tsuji, Cloning, expression and sequence analysis o cDNA for the Japanese fireflies, Pyrocoelia miyako and Hotaria parvula, Photochem. Photobiol. Sci. 1995 62 309
G. B. Sala-Newby, C. M. Thomson and A. K. Campbell, Sequence and biochemical smilarities between the luciferases of the glow-worm Lampyris noctiluca and the firefly, Photinus pyralis, Biochem. J. 1996 313 761
Y. Li, L. M. Buck, H. J. Scaeffer and F. R. Leach, Cloning and sequencing of a cDNA for the firefly luciferase from Photuris pennsilvanica, Biochim. Biophys. Acta 1997 1339 39
V. R. Viviani, A. C. R. Silva, G. L. O. Perez, R. V. Santelli, E. F. H. Bechara and F. C. Reinach, Cloning and molecular characterization of the cDNA for the Brazilian larval Click-beetle Pyrearinus termitilluminans luciferase, Photochem. Photobiol. Sci. 1999 70 254
V. R. Viviani, E. J. H. Bechara and Y. Ohmyia, Cloning, sequence analysis and expression of active Phrixothrix railroad-worms luciferases: relationship between bioluminescence spectra and primary structures, Biochem 1999 38 8271
V. R. Viviani, F. G. Arnoldi, M. Brochetto-Braga and Y. Ohmiya, Cloning and characterization of the cDNA for the Brazilian Cratomorphus distinctus larval firefly luciferase: similarities with the European Lampyris noctiluca and Asiatic Pyrocoelia luciferases, Comp. Biochem. Physiol., Part B: Biochem. Mol. Biol. 2004 139 151
D. T. Amaral, R. A. Prado and V. R. Viviani, Luciferase from Fulgeochlizus bruchi (Coleoptera:Elateridae), a Brazilian click-beetle with a single abdominal lantern: molecular evolution, biological function and comparison with other click-beetle luciferases, Photochem. Photobiol. Sci. 2012 11 1259
V. R. Viviani, T. L. Oehlmeyer, F. G. C. Arnoldi, M. R. Brochetto-Braga, A new firefly luciferase with bimodal spectrum: identification of structural determinants of spectral sensitivity in firefly luciferases, Photochem. Photobiol. Sci. 2005 81 843
B. S. Alipour, S. Hosseinkhani, M. Nikkhah, H. Naderi-Manesh, M. J. Chaichi and S. K. Osaloo, Molecular cloning, sequence analysis, and expression of a cDNA encoding the luciferase from the glow-worm, Lampyris turkestanicus, Biochem. Biophys. Res. Commun. 2004 325 215
V. R. Viviani, D. T. Amaral, R. A. Prado and F. G. C. Arnoldi, A new blueshifted luciferase from the Brazilian Amydetes fanestratus (Coleoptera: Lampyridae) firefly: molecular evolution and structural/functional properties, Photochem. Photobiol. Sci. 2011 10 1879
E. Conti, N. P. Franks and P. Brick, Crystal structure of firefly luciferase throws light on a superfamily of adenylate-forming enzymes, Structure 1996 4 287
T. Nakatsu, S. Ichiyama, J. Hiratake, A. Saldanha, N. Kobayashi, K. Sakata and H. Kato, Structural basis for the spectral difference in luciferase bioluminescence, Nature 2006 13 372
M. Kheirabadi, Z. Sharafian, H. Naderi-Manesh, U. Heineman, U. Gohlke and S. Hosseinkhani, Crystal structure of native and a mutant of Lampyris turkestanicus luciferase implicate in bioluminescence color shift, Biochim. Biophys. Acta, Proteins Proteomics 2013 1834 2729–2735
C. Carrasco-López, J. C. Ferreira, N. M. Lui, S. Schramm, R. Berraud-Pache, I. Navizet, S. Panjikar, P. Naumov and W. M. Rabeh, Beetle luciferases with naturally red- and blue-shifted emission, Life Sci. Alliance 2018 1 4
B. R. Branchini, R. A. Magyar, M. H. Murtiashaw, S. M. Anderson and M. Zimmer, Strucutral basis for the inhibition of firefly luciferase by a general anesthetic, Biochem 1998 37 15311
T. P. Sandalova and N. N. Ugarova, Model of the active site of firefly luciferase, Biochem 1999 64 962
B. R. Branchini, R. A. Magyar, M. H. Murtiashaw and N. C. Portier, The Role of Active Site Residue Arginine 218 in Firefly Luciferase Bioluminescence, Biochem 2001 40 2410
B. R. Branchini, T. L. Southworth, M. H. Murtiashaw, H. Boije and S. E. Fleet, A mutagenesis study of the luciferin binding site residues of firefly luciferase, Biochem 2003 42 10429
N. N. Ugarova and L. Y. Brovko, Protein structure and bioluminescent spectra for firefly bioluminescence, Luminescence 2002 17 321
M. DeLuca, Hydrophobic nature of the active site of firefly luciferase, Biochem 1969 8 160
E. H. White, E. Rapaport, T. A. Hopkins and H. H. Seliger, Chemi- and bioluminescence of firefly luciferin, J. Am. Chem. Soc. 1968 91 2178
F. McCapra, D. J. Gilfoyle, D. W. Young, N. J. Church and P. Spencer, The chemical origin of color differences in beetle luminescence, Bioluminescence and Chemiluminescence: Fundamental and applied aspects, ed. A. K. Campbell, L. J. Kricka and P. E. Stanley, 1994, p. 387
G. Orlova, J. D. Goddard and L. Y. Brovko, Theoretical study of the amazing firefly bioluminescence: the formation and structures of the light emitters, J. Am. Chem. Soc. 2003 125 6962
T. Hirano, Y. Hasumi, K. Ohtsuka, S. Maki, H. Niwa, M. Yamaji and D. Hashizume, Spectroscopic studies of the light-color modulation mechanism of firefly (beetle) bioluminescence, J. Am. Chem. Soc. 2009 131 2385
V. R. Viviani, D. R. Neves, D. T. Amaral, R. A. Prado, T. Matsuhashi and T. Hirano, Bioluminescence of beetle luciferases with 6′-Amino-D-luciferin analogues reveals excited keto-oxyluciferin as the emitter and phenolate/luciferin binding site interactions modulate bioluminescence colors, Biochem 2014 53(32) 5208–5220
V. R. Viviani, A. F. Simões, V. R. Bevilaqua, G. V. M. Gabriel, F. G. C. Arnoldi and T. Hirano, Glu311 and Arg337 Stabilize a Closed Active-site Conformation and Provide a Critical Catalytic Base and Countercation for Green Bioluminescence in Beetle Luciferases, Photochem. Photobiol. Sci. 2016 55 4764
V. R. Viviani, G. V. M. Gabriel and V. R. Bevilaqua, et al. The proton and metal binding sites responsible for the pH-dependent green-red bioluminescence color tuning in firefly luciferases, Sci Rep. 2018 8 17594
R. Shakeri, S. Hosseikhani and S. K. Ardestani, Role of 270 and 271 residues on function of Lampyris turkestanicus luciferase, Luminescence 2014 29 91
Z. Sohbani-Damavandifar, S. Hosseinkhani and R. H. Sajedi, Proposed ionic bond between Arg300 and Glu270 and Glu271 are not involved in inactivation of a mutant firefly luciferase (LRR), Enzyme Microb. Technol. 2016 86 17
P. J. White, D. J. Squirrell, P. Arnaud, C. R. Lowe and J. A. H. Murray, Improved thermostability of the North American firefly luciferase: saturation mutagenesis at position 354, Biochem. J. 1996 319 343–350
A. Roy, A. Kucukural and Y. Zhang, I-TASSER: a unified platform for automated protein structure and function prediction, Nat. Protoc. 2010 5 725–738
C. H. Rodrigues, D. E. Pires and D. B. Ascher, DynaMut: predicting the impact of mutations on protein conformation, flexibility and stability, Nucleic Acids Res. 2018 46 W1 W350–W355
I. Sánchez-Linares, H. Pérez-Sánchez, J. M. Cecilia and J. M. García, High-throughput parallel blind virtual screening using BINDSURF, BMC Bioinf. 2012 13 S13
A. C. Wallace, R. A. Laskowski and J. M. Thornton, LIGPLOT: a program to generate schematic diagrams of protein-ligand interactions, Protein Eng., Des. Sel. 1995 8(2) 127–134
W. L. DeLano, The Pymol Molecular Graphics System, Schrodinger Inc., 2002
A. M. Marcelino and L. M. Gierasch, Roles of beta-turn in protein folding: from peptide models to protein engineering, Biopolymers 2008 89(5) 380–391
Y. Oba, N. Mori, M. Yoshida and S. Inouye, Identification and Characterization of a Luciferase Isotype in the Japanese Firefly, Luciola cruciata, Involving in the Dim Glow of Firefly Eggs, Biochem 2010 49 107885
V. R. Viviani, F. G. C. Arnoldi, A. J. S. Neto, T. L. Oehlmeyer, E. J. H. Bechara and Y. Ohmiya, The structural origin and biological functions of pH-sensitivity in beetle luciferases, Photochem. Photobiol. Sci. 2008 7 159
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Viviani, V.R., Pelentir, G.F., Oliveira, G. et al. Role of E270 in pH- and metal-sensitivities of firefly luciferases. Photochem Photobiol Sci 19, 1548–1558 (2020). https://doi.org/10.1039/d0pp00190b
Published:
Issue Date:
DOI: https://doi.org/10.1039/d0pp00190b