Abstract
Collinear facilitation is an enhancement in the visibility of a target by laterally placed iso-oriented flankers in a collinear (COL) configuration. Iso-oriented flankers placed in a non-collinear configuration (side-by-side, SBS) produce less facilitation. Surprisingly, presentation of both configurations simultaneously (ISO-CROSS) abolishes the facilitation rather than increases it - a phenomenon that can’t be fully explained by the spatial properties of the target and flankers. Based on our preliminary data and recent studies, we hypothesized that there might be a novel explanation based on the temporal properties of the excitation and inhibition, resulting in asynchrony between the lateral inputs received from COL and SBS, leading to cancelation of the facilitatory component in ISO-CROSS. We explored this effect using a detection task in humans. The results replicated the previous results showing that the preferred facilitation for COL and SBS was abolished for the ISO-CROSS configuration. However, presenting the SBS flankers, but not the COL flankers 20 msec before ISO-CROSS restored the facilitatory effect. We propose a novel explanation that the perceptual advantage of collinear facilitation may be cancelled by the delayed input from the sides; thus, the final perception is determined by the overall spatial-temporal integration of the lateral interactions.
Similar content being viewed by others
Introduction
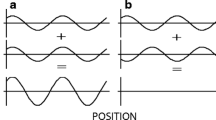
Collinear flankers (COL) enhance the visibility of a local target (Gabor patch) that is placed between them1. Such detection facilitation is found when the low-contrast target is presented simultaneously with or after high-contrast collinear flankers (see the example in each figure)1,2,3,4,5,6,7,8,9,10,11,12,13. Lateral interactions are both excitatory and inhibitory1,8,13,14,15,16,17,18,19,20,21,22,23,24,25, suggesting that they are mediated by the long-range horizontal connections formed by pyramidal neurons within layers 2–317,18,19,22,26,27,28. However, later results suggest that the long-range layer 4 circuitry plays a different functional role than that of iso-orientation biased layers 2–329. It was also found that feedback from higher cortical areas may also play a role30.
Facilitative and suppressive center-surround interactions may be organized differently to subserve different functional roles. Facilitative interactions may be organized mainly along the neuron’s optimal orientation, forming a collinear integration field (mirroring the psychophysical “association field” for collinear contour perception)1,6,7,22,31,32,33,34. Suppression is a more general phenomenon that is found for many center-surround combinations of spatial configurations, orientations and spatial separations. It was suggested that suppression may have different functions, depending on the contextual parameters35.
Studies using single-unit14,16,17,21,31,33,36, intracellular22,37,38,39 and imaging techniques40,41,42,43,44 showed that neuronal response is modulated by signals from outside the RF. Since lateral interactions are both excitatory (E) and inhibitory (I)1,8,13,14,15,16,17,18,19,20,21,22, the final outcome, suppression, or facilitation depends on the E/I balance. Although usually it was found that E/I were well balanced45, the balance depends on complex spatio-temporal parameters. In the spatial domain, the spatial configuration of the target and surround while in the collinear configuration results mainly in facilitation, whereas non-collinear configurations result in suppression22,34. The strength of the effect depends on the distance between the target and the surround22,34 and the contrast of the target and surround; however, low target contrast (mainly in the collinear configuration) reveals facilitation, whereas high contrast reveals suppression32,33,35,46,47. In the temporal domain, it was found that the propagation of the lateral input is slow8,13,22,34,38,48,49, the delay increases with increasing separation between the target and the surround13,22,38,49 and that it depends on the global arrangement of the center and surround, being faster for the collinear configuration22,34,50,51,52. Moreover, the E/I balance may be modified by training47,53,54.
Thus, the emerging results regarding the spatio-temporal properties of excitation and inhibition suggest that the inhibition results from both local3,22,33,35,47,55 and from lateral interactions22,55, whereas the lateral inhibition is mainly from iso-oriented receptive fields, having strong, transient and fast time constants, probably via shunting inhibition22,55. The excitation also derives from lateral interactions predominantly from iso-oriented receptive fields3,22,33,35,47,55,56; however, the excitation is sustained and has a slower time constant8,13,22,33,47. Thus, the E/I level is determined by the spatio-temporal parameters in the network; hence, the level is not influenced by whether the output is suppressive or facilitative3,22,33,46,47.
An alternative view of the facilitatory effect suggests that the high-contrast flankers directly stimulate neurons that are involved in detecting the target (due to overlapping receptive fields) and thus act as a low-contrast pedestal57.
Several studies have demonstrated that non-collinear iso-oriented flankers positioned at the side (side-by-side, SBS) can also facilitate detection3,7,13,58, but mostly less than COL. Surprisingly, presenting COL and SBS configurations simultaneously, producing a cross configuration (ISO-CROSS) abolishes the facilitation rather than increases it35. Another study found that the arrangement of Gabor elements surrounding the target cancels the facilitatory effect10. Taken together, the cancelation effect of collinear facilitation was rather surprising and is difficult to explain by the lateral excitation or large receptive field underlying the facilitatory effect10,35.
Another possible explanation, still in the spatial domain, is based on the results of recent Yes/No experiments exploring the collinear facilitation. When observers were asked to report the presence (yes) or absence (no) of a near threshold target (a Gabor patch), both their hit rate (reporting “yes” on the target present trials) and their false alarm rate (reporting “yes” on the target absent trials) increase in the presence of nearby collinear flankers9,47,59,60,61. It was suggested that the hit rate reflects the collinear facilitation and that the false-alarms mimic the “filling-in effect”47,59,60. One interpretation for the filling-in effect is that collinear flankers produce neuronal activity, via lateral interactions, at locations corresponding to the target even if it is not directly activated by feed forward input9,40,47. A recent study62 uses an equivalent noise approach to explore the relationships between noise and false alarms. The results are consistent with the notion that nearby collinear flankers add both signal and noise to the target location. The increased signal results in higher hit rates; the increased noise results in higher false alarm rates (the filling-in effect). Thus, in considering the ISO-CROSS configuration, one would expect to find an increased filling-in effect (a false alarm rate), thus increasing the “noise”; hence, it may cancel the facilitation. In this study we explored possible explanations for the cancelation effect using a Yes/No paradigm.
Crowding, the inability to recognize objects in a clutter, sets a fundamental limit on conscious visual perception and object recognition63,64. Several studies65,66 used the ISO-CROSS configuration to explore the crowding effect and the relationships between masking and crowding, demonstrating reduced target identification, thus suggesting that masking and crowding are not related. However, our recent spatiotemporal model47, which is based on the properties of lateral interactions, posits that masking and crowding are related in the spatial and temporal domains. The results suggest that under certain conditions, crowding and masking share common neural mechanisms that underlie the spatiotemporal properties of excitation and inhibition. Thus, transforming the facilitation in COL to “no-effect” in ISO-CROSS might be due to a shift in the neuronal output responding to the different spatial combinations that lead to different spatiotemporal outputs of excitation and inhibition.
Temporal information can lead to segregation of objects from their background with a time difference within 5 msec67,68,69,70,71,72. It was suggested that binding visual features into a coherent percept consists of synchronizing the activity of their neural representations. The results indicate that a small temporal asynchrony, below the visual integration timescale, can have a direct effect on grouping73. The results indicate that visual grouping is indeed facilitated when elements of one percept are presented at the same time as others and are temporally different from elements of another percept or from background elements. The authors concluded that the results indicate that binding is due to a global mechanism of grouping caused by synchronous neural activation73. It was largely assumed that collinear interactions serve as mechanisms involved in grouping contour elements7,33,35,74,75,76,77,78. Although spatial similarity is a fundamental rule in grouping, Polat35 suggested that temporal similarity is an additional important rule for grouping. This suggestion was backed by data showing that under collinear facilitation the neural response variance decreases79, which consequently increases the temporal correlation between the contour elements80. Recent electrophysiological studies22,49 support the notion of the importance of temporal matching between the feedforward and lateral signals, suggesting that temporal matching is critical for reaching final behavioral relevance such as grouping.
Indeed, there are some indications that the “ISO-CROSS phenomenon” might also be influenced by temporal factors. Several studies have shown that temporal processing of COL and SBS are different22,34,50,51,52,72. It was shown that the direction of motion produces bias in the neural response and in human perception. Using intra-cellular recordings from receptive fields in V1 of cats, it was found that there is a shorter delay when the lateral activation was in the collinear direction than in the orthogonal direction22,34,50. An optical imaging study in V1 of monkeys found that the cortical activity, as measured by the onset synchronization, elicited by collinear flankers, preceded that elicited by orthogonal flankers40. MEG recordings in humans showed that the neural latency associated with apparent motion in the direction of co-aligned Gabor elements (mimicking a COL configuration) was faster than the latency of motion in the orthogonal direction (mimicking a SBS configuration)52. Similarly, human perceived motion of Gabor elements in the collinear direction was found to move faster than in the orthogonal direction50,52. Thus, the results from the above studies may suggest the existence of a temporal component that influences the perception of the COL vs ISO-CROSS configurations.
Therefore, in this study we wanted to determine whether the ISO-CROSS phenomenon can be explained by spatial and temporal properties. We hypothesized that there are both spatial and temporal components causing the abolished facilitation in the ISO-CROSS configuration. The spatial component may be caused by 1) an interaction between the SBS and the COL flankers. 2) Since the facilitation is also shown to be associated with a “filling-in” effect,9,29,30,31,32, it is possible that the noise increases in ISO-CROSS, leading to a decreasing signal-to-noise ratio. 3) We also hypothesized that there is a temporal component, i.e. facilitation revealed by ISO-CROSS may occur due to the propagation time that matches the target’s response, thus resulting in facilitation8,47, whereas that of the SBS flankers is not matched; thus, it might cause a temporal asynchrony that affects the collinear facilitation.
Results
We used the Yes/No paradigm9,47,59,60 to measure the hit and false alarm rates for the COL, SBS and ISO-CROSS configurations intermixed by trials. The results are presented in Fig. 1 (N = 14). Figure 1a shows the probability of the hit and false alarm rates, showing that the hit rates for the COL and SBS configurations were significantly higher than for the ISO-CROSS configuration (blue, green, red-filled bars for COL, SBS and ISO-CROSS, respectively) (COL: p < 0.0000, SBS; p = 0.0002, paired t-test). The false alarm rate (dashed bars) was also significantly higher for the COL than for the ISO-CROSS configuration (p = 0.00004, paired t-test) but was not significantly different between SBS and ISO-CROSS (p = 0.25, paired t-test). When comparing the COL and the SBS configurations, the hit and false alarm rates were significantly higher for COL (hit rate: p = 0.027, false alarm rate; p = 0.041, paired t-test). The decision criteria (Criterion (Cr), Fig. 1b) for the ISO-CROSS configuration is significantly higher than the COL and SBS configuration (COL; p < 0.0000; SBS; p = 0.014, paired t-test) indicating that the subject’s reports are more “target no-present” for the ISO-CROSS. The Cr for COL is significantly lower than for SBS (p = 0.014; paired t-test) indicating that the subject’s reports are more “target present” for the COL. Since Cr was the lowest for COL and Cr is correlated with the effect of collinear facilitation9, the results of the Cr and the hit rate provide support for previous studies showing that facilitation is strongest for COL. Importantly, the fact that the false alarm rate decreased, whereas Cr increased in ISO-CROSS does not support the suggestion that strong excitation from COL and SBS contributes simultaneous signals to the perceptive field10,81,82,83 that processes ISO-CROSS; thus, it may be responsible for the cancelation effect of facilitation in ISO-CROSS. In addition, the suggestion of increased noise (filling-in) by ISO-CROSS is not supported, since the false alarm rate is reduced in ISO-CROSS. Figure 1c shows that the sensitivity (d’) is not significantly different among the three spatial configurations (COL vs SBS, p = 0.86; COL vs ISO-CROSS, p = 0.1; SBS vs ISO-CROSS, p = 0.18). This result is consistent with previous studies showing that d’ is not an optimal measure of collinear facilitation when using the Yes/No paradigm9,47,59,60. The reasoning for these results was described and discussed previously9,47.
Example of the stimuli and results.
Gabor target and flankers positioned at different spatial configurations. (a) Collinear (COL); (b) side-by-side (SBS); (c) COL + SBS producing a cross configuration (ISO-CROSS). The colors of the frame in each stimulus match the color of the relevant bar in figures d–f. (d) Bar charts for the probability of hit (solid bars) and false alarm rates; (e) decision criterion and (f) sensitivity. Error bars denote the mean ± standard error (N = 14).
Specificity of spatial configuration
We investigated whether the configuration effect of the collinear facilitation observed in Polat and Sagi’s study9 and the cancelation effect in ISO-CROSS are due to the positioning of any flankers at the side location or are due to a specific iso-oriented configuration. It is known that lateral facilitation diminishes as the orientation differences between the target and flankers is increased1. We performed another experiment by changing the configuration of the SBS flankers from iso-oriented to be orthogonal to the target (ORTO), resulting in control for the side-by-side configuration (ORTO-SBS). Thus, the control experiment mixed the COL, ORTO and ORTO-CROSS conditions.
The results are presented in Fig. 2. The pattern of the results for ORTO-CROSS is different from that of ISO-CROSS. Unlike the results presented in Fig. 1d, the results (Fig. 2d) for the hit and false alarm rates of COL and ORTO-CROSS are not significantly different (COL vs ORTO-CROSS, hit rate; p = 0.096, false alarm rate; p = 0.42). The hit and false alarm rates for COL and ORTO-CROSS were significantly higher than for the ORTO configuration (hit rate: p = 0.0003, p = 0.0004; false alarm rate: p = 0.01, p = 0.007; paired t-test, respectively). The Cr (Fig. 2e) for COL is slightly more negative compared with the COL in Fig. 1e, but this difference is not significant. In contrast, the Cr for ORTO (Fig. 2e) is significantly higher (more “no” answers) than the Cr for the SBS (Fig 1b) (p = 0.017; unequal sample t-test), suggesting that the orthogonal flanker induces a suppressive effect32,33,60. The Cr for COL and for ORTO-CROSS are significantly lower than the Cr for ORTO (p < 0000, p < 0.0000, paired t-test, respectively). There is no significant difference between the Cr of COL and that of ORTO-CROSS (p = 0.13 paired t-test). The results of d’ are not significantly different among the spatial configurations (Fig. 2f). Thus, the results of this control experiment indicate that the cancellation effect that is described in Fig. 1 is configuration dependent, occurring only for iso-orientations, as expected from the architecture of the collinear facilitation1,7.
Example of the stimuli and control for the spatial configurations.
Gabor target and flankers positioned at different spatial configurations. (a) Collinear (COL); (b) side-by-side but for an orthogonal configuration of the flankers (ORTO); (c) COL + ORTO producing the arto-cross configuration (ORTO-CROSS). The colors of the frame in each stimulus match the color of the relevant bar in figures d–f. (d) Probability of the hit rate (filled bars) and false alarms (dashed bars) for the collinear configuration (COL), ORTO) and combined, COL and SBS producing a ORTO-CROSS configuration. (e) Decision criteria (criterion). (f) Sensitivity (d’). Error bars denote the mean ± standard error (N = 7).
Temporal properties – forward presentation of flankers
We hypothesize that the propagation time of the SBS flankers is slower than that of the COL flankers. Our previous pilot study, using measurements of event-related potential in humans, showed that presenting SBS flankers 20 msec before ISO-CROSS is optimal for restoring the collinear facilitation84. Thus, in the next experiment we presented the COL (COL-F-ISO-CROSS) and the SBS flankers (SBS-F-ISO-CROSS) 20 msec before the appearance of the ISO-CROSS condition (forward masking) and they remained presented with ISO-CROSS for another 60 msec. The regular COL, SBS and ISO-CROSS conditions were also tested (a presentation time of 60 msec); thus, we tested five conditions mixed-by-trial. We also measured the reaction time in this experiment. The results are presented in Fig. 3. The hit rate for the COL configuration is significantly higher than for the SBS (p = 0.018) and ISO-CROSS (p = 0.018); however, the results for false alarm rate are not significantly different (p = 0.13, p = 0.16, respectively). In addition, there is no significant difference between SBS and ISO-CROSS for hit and false alarm rate (p = 0.58, p = 0.97, respectively). Taken together, these results are similar to those presented in Fig. 2d. Likewise, the Cr (Fig. 3b) is lower for SBS and ISO-CROSS (p = 0.031 and p = 0.02, respectively, paired t-test). The Cr is not significantly different between SBS and ISO-CROSS (p = 0.3).
Example of the stimuli and results of the control experiment for the temporal control Gabor target and flankers positioned at different spatial configurations.
Collinear (COL); side-by-side (SBS) COL + SBS resulting in an iso-cross configuration (ISO-CROSS). Collinear flankers (COL) presented 20 msec before the ISO-CROSS; SBS flankers presented 20 msec before the ISO-CROSS. The colors of the frame in each stimulus match the color of the relevant bar in the following figures. Probability of the hit rate (filled bars) and the false alarms (dashed bars), Decision criteria (criterion), Reaction time, Sensitivity, Error bars denote the mean ± standard error (N = 7).
However, a remarkable change occurs when the flankers are presented before ISO-CROSS. Forward presentation of COL flankers 20 msec before ISO-CROSS (COL-F-ISO-CROSS) only slightly increased the hit rate but it did not change it significantly (hit rate: p = 0.17; false alarm rate p = 0.55). In contrast, presenting the SBS flankers (SBS-F-ISO-CROSS) 20 msec before CROSS dramatically and significantly changed the results for the CROSS; the hit rate increased significantly from 0.68 to 0.86 (p = 0.0002, paired t-test); the Cr also decreased significantly from 0.83 to 0.24 (p = 0.011, paired t-test). As a result, the effect of cancelation disappear; the hit rate of the ISO-CROSS configuration, when the SBS presented before, was not significantly different from the COL configuration (p = 0.16). As in the previous experiments, the results for d’ were not significantly different among the configurations. Thus, the results of this control experiment, showing that placing SBS flankers before ISO-CROSS (SBS-F-ISO-CROSS), revealed the expected collinear facilitation. This result supports our hypothesis that a temporal component is involved in the processing of ISO-CROSS. Recent studies measuring intracellular recordings provide support for these results22,23.
In this experiment we also measured the reaction time to estimate the processing speed of each configuration (Fig. 3). The reaction time is significantly faster (40 msec) for the COL configuration than for the SBS configuration (p = 0.019) and 34 msec faster than the ISO-CROSS configuration (p = 0.001, paired t-test, respectively). There is no significant difference between SBS and ISO-CROSS (p = 68). The reaction time is also faster by 40 msec for the COL-F-ISO-CROS and SBS-F-ISO-CROSS than for ISO-CROSS (p = 0.035, p = 0.01, respectively). Thus, these results further support our hypothesis that COL is processed faster than SBS, consistent with previous data showing that the perception of apparent motion in the collinear direction is perceived as faster than SBS52.
Discussion
We found that the hit rates of the collinear configuration are higher than those of the side-by-side configuration. This result is consistent with the configuration preference of the facilitation to collinear configuration showing lower thresholds7, improved threshold summation85, synaptic facilitation22 and higher brain signals74. Here we also confirmed previous results that adding additional flankers to the sides (SBS) of the collinear configuration abolished the superiority of the collinear effect in the ISO-CROSS configuration.
Spatio-temporal model based on the properties of excitation and inhibition
As mentioned in the Introduction, lateral interactions are both excitatory (E) and inhibitory (I)1,8,13,14,15,16,17,18,19,20,21,22. Results suggest that the contextual effects are mediated by the long-range horizontal connections formed by pyramidal neurons within V117,18,19,22,26,27,28,49. The emerging results from these studies indicate that excitation is more selective and is received between non-overlapping neurons connected by long-range connections and have similar optimal orientation selectivity that tends to make preferred connections along the collinear configurations. The inhibitory effect resulted either from the lateral interactions or from local interactions thus is being less selective.
In the temporal domain, it was shown that the time constant of the inhibition is rapid and transient5,8,14,22. In contrast, the time constant of the excitation is relatively delayed and sustained8,13,14,20,86,87 and may abrogate the inhibition with increasing presentation times8,47. Recent research using intracellular recordings22 show that subthreshold responses to oriented stimuli flashed outside the receptive field exhibited a geometrical organization around the preferred orientation axis, mirroring the psychophysical association field for collinear contour perception; however, non-collinear direction may produce fast shunting inhibition. Thus, the final outcome of the E/I depends on the spatio-temporal parameters determined by the spatial configuration of the target and flankers, the spacing between them, the contrast of both the target and flankers and the temporal presentation of the target and flankers (see Fig. 4 for an illustration of the model). In this regard, studies from Fregnac and colleagues22,23 are highly relevant, showing a temporal advantage of about 20 msec of collinear configuration over the parallel configuration.
Model illustration of the spatial temporal properties of the excitation and inhibition underscore the lateral interactions.
Gabor target and flankers positioned at different spatial configurations producing collinear (COL, top), side-by-side (SBS, middle) and COL + SBS, producing iso-cross configuration (ISO-CROSS, bottom). Each row depicts different time intervals of the lateral propagation, immediate (T1), intermediate (T2) and after the excitation arrives to the target location (T3). The color code indicates that a lateral inhibition that is fast produces suppression (red), whereas a lateral excitation that arrives later produces facilitation (green). Intermediate colors (orange and yellow indicate the balancing effect of the inhibition and excitation, revealing no effect).
Previous studies that explored the effect of spatial configurations on the collinear facilitation found that adding additional flankers to the sides of the collinear configuration cancelled the facilitatory effect10,35. This effect was surprising and challenged previous models of spatial vision. There were a few accounts that attempted to explain this effect. One was that the SBS flankers may increase the excitation35, which then shifts the target’s activity from near threshold to a no facilitation zone10,15,33,57,81,82,83. If so, one would expect to find an increase in the hit and false alarm rates in ISO-CROSS. An increase in the false alarm may contribute to higher noise62; thus, it may cancel the facilitatory effect. Here we found an opposite effect, a decrease in the hit and false alarm rates in ISO-CROSS compared with COL. We also found that Cr is increased in ISO-CROSS, suggesting that the level of neural interaction is shifted to a lower facilitation level9. Therefore, an explanation based on the spatial arrangement of SBS contributing to higher excitation is less likely. Another possibility is that the signal from the SBS flankers contribute shunting inhibition that cancel the COL excitation22.
An alternative possibility is based on the temporal properties of excitation and inhibition that were described above (see also Fig. 4). If the effect is due to temporal differences between COL and SBS and the propagation of SBS is slower than that of COL, then when they are combined in the ISO-CROSS configuration, temporal asynchrony similar to backward masking should occur88,89. Backward masking cancels the collinear facilitation, leading to a reduction of the target’s visibility88,89 and reduced hit and false alarm rates that should decrease Cr (less target present reports)88. It is possible that the early activation of side-band inhibition (advanced by 20 msec in SBS-F-CROSS) has an earlier suppressive (shunting) effect that decays when the delayed excitation provided by the collinear flankers (COL) arrives. Thus, in ISO-CROSS, slower propagation of the SBS signal can contribute to shunting inhibition22 that cancels the effect of collinear facilitation. We noted that one of the leading models for backward masking is fast inhibition (similar to shunting inhibition) produced by the mask on the target8,89,90. Indeed, here we found a behavioral effect similar to backward masking, in ISO-CROSS, showing decreased hit and false alarm rates and increased Cr compared with COL and SBS configurations, consistent with what is expected from backward masking89. Since the control experiment showed an opposite effect for the control CROSS (ORTO-CROSS), this result supports the idea that the effect is not solely due to the spatial presentation of flankers on the sides of the collinear configuration—it is also due to the temporal property that they may produce fast (shunting) inhibition.
Moreover, we tested the prediction of a temporal asynchrony. When we presented the SBS flankers 20 msec before ISO-CROSS, the effect of cancelation disappeared. This is in agreement with the idea that the propagation time of the COL signals is faster than that of the SBS flankers50,52. Thus, SBS signals, arriving at a slight delay, produce a temporal asynchrony. Support for our prediction of a temporal advantage of COL is found in the reaction time data showing that the reaction time is faster for COL than for SBS and ISO-CROSS (see Fig. 3c). Additional support is found in our pilot event related potential (ERP) study84, showing that the latency of SBS is larger than that of COL and that presenting the SBS 20 msec before ISO-CROSS recovers the facilitation in ISO-CROSS. These results are consistent with previous results showing an advantage of the collinear response over the non-collinear configuration7,31,35,50,52,74,85; this is supported by recent studies by Fregnac and colleagues22,23.
It was suggested that temporal information can lead to segregation of objects from their background with a time difference within 5 msec67,68,69,70,71. Results indicate that a small temporal asynchrony, below the visual integration timescale, can have a direct effect on the grouping73. The results also provide psychophysical and computational support, suggesting that the visual system implements a mechanism that synchronizes the response onsets to object parts and attenuates or cancels their latency differences72. It was largely assumed that collinear interactions serve as mechanisms involved in grouping contour elements7,33,35,74,75,76,77,78. This suggestion was backed by data showing that under collinear facilitation the neural response variance decreased79, which improved the temporal correlation between the contour elements80. However, our results show that adding the SBS flankers to COL abolished the collinear facilitation, suggesting that the temporal delay of SBS is larger than the time delay that the visual system can attenuate72. Thus, our data suggest that a temporal asynchrony larger than a few msec cannot be attenuated to support grouping.
It is largely assumed that masking and crowding are different63,64,91. Several studies, using a set of Gabor patches, showed that global configuration affects crowding66,92,93,94. The general view from these studies is that crowding is more pronounced when the effect of grouping increases. Related to our study is the use of the ISO-CROSS configuration66 as a stimulus for exploring crowding, which showed that ISO-CROSS impairs orientation discrimination. Our recent results47 suggest that under certain spatiotemporal conditions, visual crowding and masking share common neural mechanisms of excitation and inhibition. Here we kept the spatial separation constant (3λ), at a range that is known to reveal facilitation and we showed that changing the global configuration abolished the perception of collinear facilitation. Therefore, the effect of ISO-CROSS can be seen as crowding, despite that the target is at the threshold and the task is detection. In other words, since the E/I level for facilitation depends on the stimulus parameters and on the network properties, the delayed signal from SBS may contribute to inhibition by shifting the network to a level where there is no facilitation.
It was suggested that collinear facilitation provides an advantage of functional significance7,9,22,31,33,35,74,85,95,96 that may contribute to the assigning of an image’s contours, providing a substrate for further cognitive processing. This experimental result is also supported by the statistics of natural images97. Thus, one would expect that the faster response (of COL) should prevail in the ISO-CROSS configuration. However, surprisingly, we saw that a slower response of SBS modifies the perception of collinear facilitation that is widely believed to play a functional role in contour integration. The change in this perception may therefore suggest another perceptual significance of ISO-CROSS (and crowding) in the processing of surfaces and textures.
Methods
Subjects
Twenty-one subjects with normal or corrected-to-normal visual acuity participated in the experiments. The procedures were approved by the ethics committee of the Sheba Medical Center and all participants gave informed written consent to participate in the study. All experimental protocols were performed in accordance with the guidelines provided by the committee approving the experiments.
Apparatus
Stimuli were displayed as gray-level modulation on a Philips 107P color monitor. The experiments were controlled by a Dell PC. Screen resolution was 1024 × 768 pixels occupying a 9.20 × 12.20 of visual degrees. The refresh rate was 100 Hz. The mean display luminance was 40 cd/m2 in an otherwise dark environment. Gamma correction was applied. The stimuli were viewed from a distance of 150 cm.
Visual stimuli
The stimuli were presented as gray-level images (Gabor patches) with a spatial frequency of 6 cycles per degree (cpd) modulated from a background luminance of 40 cd/m2, with a 60 msec duration. The spread of the Gaussian envelope (б) was equated with the wavelength (λ, 0.166°) of the carrier1. The target’s contrast was adjusted to the participant’s threshold (4–7%) and the contrast of the flankers was always 60%. The target-flanker separation was always 3λ for all spatial configurations: collinear (COL), side-by-side (SBS) and a combination of COL and SBS producing ISO-CROSS (Fig. 1). The orientations of the target and flankers were vertical.
We performed two control experiments: a spatial control experiment in which the side-by-side flankers were orthogonal to the target (ORTO), which resulted in control for ISO-CROSS (ORTO-CROSS). We also performed temporal control experiment in which the COL or the SBS flanker was presented 20 msec before ISO-CROSS (COL-F-ISO-CROSS, SBS-F-ISO-CROSS) and remained presented as part of ISO-CROSS for the remaining 60 msec. Thus, in this experiment five conditions were mixed-by-trial.
Experimental procedures
A Yes/No paradigm was used9,47,59,60,61. Subjects were asked to detect a target that may appear or not (Yes/No) between the flankers in all different configurations (COL, SBS, ISO-CROSS, ORTO and ORTO-CROSS). Target and non-target trials appeared randomly. Participants reported whether the target was present (Yes) or absent (No) by pressing the left and right mouse keys, respectively. They were informed of a wrong answer by auditory feedback after each presentation throughout the experiment. A visible fixation circle appeared in the center before each trial and disappeared when the trial started. The order of the configurations was randomized between trials (the “Mix” procedure); each orientation was presented 100 times per configuration.
The false alarm, miss, hit and correct rejection rates were recorded and analyzed to yield the sensitivity (d’ = z(Hit)-z(FA)) and the criterion (Cr = (z(Hit) + z(FA))/2) measures, with z defined as the inverse of the normal distribution function. This calculation was used in the previous studies9,47,59,60,61 and is based on MacMillian and Creelman’s equation98, which can be viewed as a deviation from the ideal observer’s decision criterion. The experiments were performed using the “Mix” procedure9,47,59,60. In the “Mix” procedure, the trials with different target–flanker configurations are presented in a random order. Each configuration was presented 100 times with the target present in about half of the trials (a probability of 0.5).
Additional Information
How to cite this article: Lev, M. and Polat, U. Temporal asynchrony and spatial perception. Sci. Rep. 6, 30413; doi: 10.1038/srep30413 (2016).
References
Polat, U. & Sagi, D. Lateral interactions between spatial channels: suppression and facilitation revealed by lateral masking experiments. Vision res 33, 993–999 (1993).
Adini, Y. & Sagi, D. Recurrent networks in human visual cortex: psychophysical evidence. Opt Soc Am 18, 2228–2236 (2001).
Adini, Y., Sagi, D. & Tsodyks, M. Excitatory-inhibitory network in the visual cortex: psychophysical evidence. P Nat Acad Sci USA 94, 10426–10431 (1997).
Bonneh, Y. & Sagi, D. Effects of spatial configuration on contrast detection. Vision res 38, 3541–3553 (1998).
Cass, J. & Alais, D. The mechanisms of collinear integration. J vis 6, 915–922 (2006).
Polat, U. & Sagi, D. Spatial interactions in human vision: from near to far via experience- dependent cascades of connections. P Nat Acad Sci USA 91, 1206–1209 (1994).
Polat, U. & Sagi, D. The architecture of perceptual spatial interactions. Vision res 34, 73–78 (1994).
Polat, U. & Sagi, D. Temporal asymmetry of collinear lateral interactions. Vision res 46, 953–960 (2006).
Polat, U. & Sagi, D. The relationship between the subjective and objective aspects of visual filling-in. Vision res 47, 2473–2481, doi: 10.1016/j.visres.2007.06.007 (2007).
Solomon, J. A. & Morgan, M. J. Facilitation from collinear flanks is cancelled by non-collinear flanks. Vision res 40, 279–286 (2000).
Woods, R. L., Nugent, A. K. & Peli, E. Lateral interactions: size does matter. Vision res 42, 733–745 (2002).
Levi, D. M., Hariharan, S. & Klein, S. A. Suppressive and facilitatory spatial interactions in amblyopic vision. Vision res 42, 1379–1394 (2002).
Cass, J. R. & Spehar, B. Dynamics of collinear contrast facilitation are consistent with long-range horizontal striate transmission. Vision res 45, 2728–2739 (2005).
Bair, W., Cavanaugh, J. R. & Movshon, J. A. Time course and time-distance relationships for surround suppression in macaque V1 neurons. J neurosci 23, 7690–7701 (2003).
Chen, C. C., Kasamatsu, T., Polat, U. & Norcia, A. M. Contrast response characteristics of long-range lateral interactions in cat striate cortex. Neuroreport 12, 655–661 (2001).
Kasamatsu, T., Miller, R., Zhu, Z., Chang, M. & Ishida, Y. Collinear facilitation is independent of receptive-field expansion at low contrast. Exp brain res 201, 453–465, doi: 10.1007/s00221-009-2057-1 (2010).
Weliky, M., Kandler, K., Fitzpatrick, D. & Katz, L. C. Patterns of excitation and inhibition evoked by horizontal connections in visual cortex share a common relationship to orientation columns. Neuron 15, 541–552 (1995).
Kisvarday, Z. F., Toth, E., Rausch, M. & Eysel, U. T. Orientation-specific relationship between populations of excitatory and inhibitory lateral connections in the visual cortex of the cat. Cereb Cortex 7, 605–618 (1997).
Mizobe, K., Polat, U., Pettet, M. W. & Kasamatsu, T. Facilitation and suppression of single striate-cell activity by spatially discrete pattern stimuli presented beyond the receptive field. Visual neurosci 18, 377–391. (2001).
Fitzpatrick, D. Seeing beyond the receptive field in primary visual cortex. Curr opin neurobiol 10, 438–443 (2000).
Levitt, J. B. & Lund, J. S. Contrast dependence of contextual effects in primate visual cortex. Nature 387, 73–76 (1997).
Gerard-Mercier, F., Carelli, P. V., Pananceau, M., Troncoso, X. G. & Fregnac, Y. Synaptic Correlates of Low-Level Perception in V1. J neurosci 36, 3925–3942, doi: 10.1523/JNEUROSCI.4492-15.2016 (2016).
Troncoso, X. G. et al. Spatio-temporal synergy requirements for binding feedforward and horizontal waves in V1. SFN Annual Meeting, 331, 304/P342, http://www.abstractsonline.com/plan/ViewAbstract.aspx?cKey=6c7d4543-3741-4da2-a6ea-8637dd78927e&mID=3744&mKey=d0ff4555-8574-4fbb-b9d4-04eec8ba0c84&sKey=3f920ffc-ee5a-49dc-90da-070e281e26d0 (2015).
Haider, B., Duque, A., Hasenstaub, A. R. & McCormick, D. A. Neocortical network activity in vivo is generated through a dynamic balance of excitation and inhibition. J neurosci 26, 4535–4545, doi: 10.1523/JNEUROSCI.5297-05.2006 (2006).
Shu, Y., Hasenstaub, A. & McCormick, D. A. Turning on and off recurrent balanced cortical activity. Nature 423, 288–293 (2003).
Bolz, J. & Gilbert, C. D. The role of horizontal connections in generating long receptive fields in the cat visual cortex. Euro J Neurosc 1, 263–268 (1989).
Gilbert, C. D. & Wiesel, T. N. Columnar specificity of intrinsic horizontal and corticocortical connections in cat visual cortex. J Neurosci 9, 2432–2442 (1989).
Hirsch, J. A. & Gilbert, C. D. Synaptic physiology of horizontal connections in the cat’s visual cortex. J Neurosci 11, 1800–1809 (1991).
Yousef, T. et al. Orientation topography of layer 4 lateral networks revealed by optical imaging in cat visual cortex (area 18). The Euro J Neurosc 11, 4291–4308 (1999).
Gilbert, C. D. & Li, W. Top-down influences on visual processing. Nat Rev Neurosci 14, 350–363, doi: 10.1038/nrn3476 (2013).
Kapadia, M. K., Ito, M., Gilbert, C. D. & Westheimer, G. Improvement in visual sensitivity by changes in local context: parallel studies in human observers and in V1 of alert monkeys. Neuron 15, 843–856 (1995).
Polat, U. & Norcia, A. M. Neurophysiological evidence for contrast dependent long-range facilitation and suppression in the human visual cortex. Vision res 36, 2099–2109 (1996).
Polat, U., Mizobe, K., Pettet, M. W., Kasamatsu, T. & Norcia, A. M. Collinear stimuli regulate visual responses depending on cell’s contrast threshold. Nature 391, 580–584 (1998).
Chavane, F. et al. The visual cortical association field: a Gestalt concept or a psychophysiological entity? J physiology-Paris 94, 333–342 (2000).
Polat, U. Functional architecture of long-range perceptual interactions. Spatial vision 12, 143–162 (1999).
Mizobe, K., Polat, U., Kasamatsu, T. & Norcia, A. M. Lateral masking reveals facilitation and suppression from the same single cells in cat area 17. Invest Ophthal Visl Sci 37, S483 (1996).
Shu, Y., Hasenstaub, A. & McCormick, D. A. Turning on and off recurrent balanced cortical activity. Nature 423, 288–293 (2003).
Bringuier, V., Chavane, F., Glaeser, L. & Fregnac, Y. Horizontal propagation of visual activity in the synaptic integration field of area 17 neurons. Science 283, 695–699 (1999).
Borg-Graham, L. J., Monier, C. & Fregnac, Y. Visual input evokes transient and strong shunting inhibition in visual cortical neurons. Nature 393, 369–373 (1998).
Meirovithz, E. et al. Population response to contextual influences in the primary visual cortex. Cereb Cortex 20, 1293–1304, doi: 10.1093/cercor/bhp191 (2010).
Malach, R., Amir, Y., Harel, M. & Grinvald, A. Relationship between intrinsic connections and functional architecture revealed by optical imaging and in vivo targeted biocytin injections in primate striate cortex. P Nat Acad Sci USA 90, 10469–10473 (1993).
Haider, B. et al. Synaptic and network mechanisms of sparse and reliable visual cortical activity during nonclassical receptive field stimulation. Neuron 65, 107–121, doi: 10.1016/j.neuron.2009.12.005 (2010).
Chavane, F. et al. Lateral Spread of Orientation Selectivity in V1 is Controlled by Intracortical Cooperativity. Front syst neurosci 5, 4, doi: 10.3389/fnsys.2011.00004 (2011).
Jancke, D., Chavane, F., Naaman, S. & Grinvald, A. Imaging cortical correlates of illusion in early visual cortex. Nature 428, 423–426, doi: 10.1038/nature02396 (2004).
Ozeki, H. et al. Relationship between Excitation and Inhibition Underlying Size Tuning and Contextual Response Modulation in the Cat Primary Visual Cortex. J Neurosci 24, 1428–1438, doi: 10.1523/jneurosci.3852-03.2004 (2004).
Ichida, J. M., Schwabe, L., Bressloff, P. C. & Angelucci, A. Response facilitation from the “suppressive” receptive field surround of macaque V1 neurons. J Neurophysiology 98, 2168–2181, doi: 10.1152/jn.00298.2007 (2007).
Lev, M. & Polat, U. Space and time in masking and crowding. J vis 15, 10, doi: 10.1167/15.13.10 (2015).
Polat, U., Sterkin, A. & Yehezkel, O. Spatio-temporal low-level neural networks account for visual masking. Adv Cogn Psychol 3, 153–165, doi: 10.2478/v10053-008-0021-4 (2007).
Kim, K., Kim, T., Yoon, T. & Lee, C. Covariation between Spike and LFP Modulations Revealed with Focal and Asynchronous Stimulation of Receptive Field Surround in Monkey Primary Visual Cortex. PloS one 10, e0144929, doi: 10.1371/journal.pone.0144929 (2015).
Georges, S., Series, P., Fregnac, Y. & Lorenceau, J. Orientation dependent modulation of apparent speed: psychophysical evidence. Vision res 42, 2757–2772 (2002).
Series, P., Georges, S., Lorenceau, J. & Fregnac, Y. Orientation dependent modulation of apparent speed: a model based on the dynamics of feed-forward and horizontal connectivity in V1 cortex. Vision res 42, 2781–2797 (2002).
Paradis, A.-L., Morel, S., Seriès, P. & Lorenceau, J. Speeding up the brain: when spatial facilitation translates into latency shortening. Frontiers Human neurosci 6, doi: 10.3389/fnhum.2012.00330 (2012).
Polat, U., Ma-Naim, T., Belkin, M. & Sagi, D. Improving vision in adult amblyopia by perceptual learning. P Nat Acad Sci USA 101, 6692–6697 (2004).
Polat, U. Restoration of underdeveloped cortical functions: evidence from treatment of adult amblyopia. Restor Neurol Neurosc 26, 413–424 (2008).
Liu, Y. J., Hashemi-Nezhad, M. & Lyon, D. C. Dynamics of extraclassical surround modulation in three types of V1 neurons. J Neurophysiology 105, 1306–1317, doi: 10.1152/jn.00692.2010 (2011).
McGuire, B. A., Gilbert, C. D., Rivlin, P. K. & Wiesel, T. N. Targets of horizontal connections in macaque primary visual cortex. J Comp Neurol 305, 370–392 (1991).
Solomon, J. A., Watson, A. B. & Morgan, M. J. Transducer model produces facilitation from opposite-sign flanks. Vision res 39, 987–992 (1999).
Ejima, Y. & Miura, K. Y. Change in detection threshold caused by peripheral gratings: dependence on contrast and separation. Vision res 24, 367–372 (1984).
Zomet, A., Amiaz, R., Grunhaus, L. & Polat, U. Major Depression Affects Perceptual Filling-In. Biol Psychiat (2008).
Lev, M. & Polat, U. Collinear facilitation and suppression at the periphery. Vision res 51, 2488–2498, doi: 10.1016/j.visres.2011.10.008 (2011).
Amiaz, R., Zomet, A. & Polat, U. Excitatory repetitive transcranial magnetic stimulation over the dorsolateral prefrontal cortex does not affect perceptual filling-in in healthy volunteers. Vision res 51, 2071–2076, doi: 10.1016/j.visres.2011.08.003 (2011).
Zomet, A., Polat, U. & Levi, D. M. Noise and the Perceptual Filling-in effect. Sci Rep, 6, 24938. doi: 10.1038/srep24938 (2016).
Levi, D. M. Crowding–an essential bottleneck for object recognition: a mini-review. Vision res 48, 635–654, doi: 10.1016/j.visres.2007.12.009 (2008).
Whitney, D. & Levi, D. M. Visual crowding: a fundamental limit on conscious perception and object recognition. Trends Gogn Sci 15, 160–168, doi: 10.1016/j.tics.2011.02.005 (2011).
Levi, D. M. & Carney, T. Crowding in peripheral vision: why bigger is better. Curr biol 19, 1988–1993, doi: 10.1016/j.cub.2009.09.056 (2009).
Levi, D. M. & Carney, T. The effect of flankers on three tasks in central, peripheral and amblyopic vision. J vis 11, 10, doi: 10.1167/11.1.10 (2011).
Fahle, M. Figure-ground discrimination from temporal information. P Royal Soc Lon B Bio 254, 199–203, doi: 10.1098/rspb.1993.0146 (1993).
Leonards, U., Singer, W. & Fahle, M. The influence of temporal phase differences on texture segmentation. Vision res 36, 2689–2697 (1996).
Engel, A. K., Konig, P., Kreiter, A. K., Schillen, T. B. & Singer, W. Temporal coding in the visual cortex: new vistas on integration in the nervous system. Trends Neurosci 15, 218–226 (1992).
Engel, A. K., Konig, P. & Singer, W. Direct physiological evidence for scene segmentation by temporal coding. P Nat Acad Sci USA 88, 9136–9140 (1991).
Singer, W. & Gray, C. M. Visual feature integration and the temporal correlation hypothesis. Annu Rev Nneurosci 18, 555–586 (1995).
Cheadle, S. et al. Spatial structure affects temporal judgments: evidence for a synchrony binding code. J vis 8, 12 11–12, doi: 10.1167/8.7.12 (2008).
Usher, M. & Donnelly, N. Visual synchrony affects binding and segmentation in perception. Nature 394, 179–182 (1998).
Polat, U. & Norcia, A. M. Elongated physiological summation pools in the human visual cortex. Vision res 38, 3735–3741 (1998).
Kovacs, I. & Julesz, B. A closed curve is much more than an incomplete one: effect of closure in figure-ground segmentation. P Nat Acad Sci USA 90, 7495–7497 (1993).
Kovacs, I. Gestalten of today: early processing of visual contours and surfaces. Behav Brain Res 82, 1–11 (1996).
Li, W. & Gilbert, C. D. Global contour saliency and local colinear interactions. J Neurophysiol 88, 2846–2856 (2002).
Li, W., Piech, V. & Gilbert, C. D. Contour saliency in primary visual cortex. Neuron 50, 951–962 (2006).
Kasamatsu, T., Polat, U., Pettet, M. W. & Norcia, A. M. Colinear facilitation promotes reliability of single-cell responses in cat striate cortex. Exp brain res 138, 163–172 (2001).
Sterkin, A. & Polat, U. Response similarity as a basis for perceptual binding. Journal of vision 8, 17 11–12, doi: 10.1167/8.7.17 (2008).
Chen, C. C. & Tyler, C. W. Lateral sensitivity modulation explains the flanker effect in contrast discrimination. P Royal Soc Lon B Bio 268, 509–516, doi: 10.1098/rspb.2000.1387 (2001).
Chen, C. C. & Tyler, C. W. Lateral modulation of contrast discrimination: flanker orientation effects. J vis 2, 520–530, doi: 10:1167/2.6.8 (2002).
Chen, C. C. & Tyler, C. W. Excitatory and inhibitory interaction fields of flankers revealed by contrast-masking functions. J vis 8, 10 11–14, doi: 10.1167/8.4.10 (2008).
Lev, M. & Polat, U. When simultaneous presentation results in backward masking. J vis 10, 1365, doi: 10.1167/10.7.1365 (2010).
Polat, U. & Tyler, C. W. What pattern the eye sees best. Vision research 39, 887–895 (1999).
Grinvald, A., Lieke, E. E., Frostig, R. D. & Hildesheim, R. Cortical point-spread function and long-range lateral interactions revealed by real-time optical imaging of macaque monkey primary visual cortex. J Neurosci 14, 2545–2568 (1994).
Kapadia, M. K., Westheimer, G. & Gilbert, C. D. Dynamics of spatial summation in primary visual cortex of alert monkeys. P Nat Acad Sci USA 96, 12073–12078 (1999).
Sterkin, A., Yehezkel, O. & Polat, U. Learning to be fast: Gain accuracy with speed. Vision res 61, 115–124, doi: 10.1016/j.visres.2011.09.015 (2012).
Sterkin, A., Yehezkel, O., Bonneh, Y. S., Norcia, A. & Polat, U. Backward masking suppresses collinear facilitation in the visual cortex. Vision res 49, 1784–1794, doi: 10.1016/j.visres.2009.04.013 (2009).
Breitmeyer, B. G. Visual masking: an integrative approach. Vol. 4 (Oxford University Press, 1984).
Pelli, D. G., Palomares, M. & Majaj, N. J. Crowding is unlike ordinary masking: distinguishing feature integration from detection. J vis 4, 1136–1169 (2004).
Livne, T. & Sagi, D. Configuration influence on crowding. Journal of vision 7, 4 1–12 (2007).
Livne, T. & Sagi, D. How do flankers’ relations affect crowding? J vis 10, 1 1–14, doi: 10.1167/10.3.1 (2010).
Saarela, T. P., Sayim, B., Westheimer, G. & Herzog, M. H. Global stimulus configuration modulates crowding. J vis 9, 5 1–11, doi: 10.1167/9.2.5 (2009).
Ramachandran, V. S., Ruskin, D., Cobb, S., Rogers-Ramachandran, D. & Tyler, C. W. On the perception of illusory contours. Vision res 34, 3145–3152 (1994).
Dresp, B. & Bonnet, C. Psychophysical evidence for low-level processing of illusory contours and surfaces in the Kanizsa square. Vision res 31, 1813–1817 (1991).
Geisler, W. S., Perry, J. S., Super, B. J. & Gallogly, D. P. Edge co-occurrence in natural images predicts contour grouping performance. Vision res 41, 711–724 (2001).
Macmillan, N. A. & Creelman, C. D. Detection Theory. (Lawrence Erlbaum Associates, Inc., 2005).
Acknowledgements
This work was performed in partial fulfillment of the requirements for a Ph.D. degree by Maria Lev at the Sackler Faculty of Medicine, Tel Aviv University, Ramat Aviv, Israel. This study was supported by grants from the Israel Science Foundations (ISF188/2010). We thank Dr. Sharon Gilaie-Dotan for her help in writing the manuscript.
Author information
Authors and Affiliations
Contributions
M.L. and U.P. were involved in designing the study, as well as in writing, editing and reviewing the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lev, M., Polat, U. Temporal asynchrony and spatial perception. Sci Rep 6, 30413 (2016). https://doi.org/10.1038/srep30413
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep30413
- Springer Nature Limited
This article is cited by
-
Binocular summation is affected by crowding and tagging
Scientific Reports (2021)