Abstract
Data sources
Cochrane Oral Health Group Trials Register, Cochrane Central Register of Controlled Trials (CENTRAL), Medline, Embase, CANCERLIT, CINAHL, the US National Institutes of Health Trials Registry and the WHO Clinical Trials Registry Platform.
Study selection
Randomised controlled trials (RCTs) assessing the effects of oral cryotherapy in patients with cancer receiving treatment compared to usual care, no treatment or other interventions to prevent mucositis. The primary outcome was incidence of mucositis and its severity.
Data extraction and synthesis
Two reviewers carried out study assessment and data extraction independently. Treatment effect for continuous data was calculated using mean values and standard deviations and expressed as mean difference (MD) and 95% confidence interval. Risk ratio (RR) was calculated for dichotomous data. Meta-analysis was performed.
Results
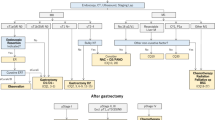
Fourteen studies with 1280 participants were included. Subgroup analysis was undertaken according to the main cancer treatment type. Cryotherapy reduced the risk of developing mucositis by 39% (RR = 0.61; 95%CI, 0.52 to 0.72) on patients treated with fluorouracil (5FU). For melphalan-based treatment the risk of developing mucositis was reduced by 41% (RR =0.59; 95%CI, 0.35 to 1.01). Oral cryotherapy was shown to be safe, with very low rates of minor adverse effects, such as headaches, chills, numbness/taste disturbance and tooth pain. This appears to contribute to the high rates of compliance seen in the included studies.
Conclusions
There is confidence that oral cryotherapy leads to a large reduction in oral mucositis in adults treated with 5FU. Although there is less certainty on the size of the reduction on patients treated with melphalan, it is certain there is reduction of severe mucositis.
Similar content being viewed by others
Commentary
Mucositis is a debilitating condition affecting a large number of patients receiving cancer treatment. According to the National Cancer Institute development of new technologies to prevent cancer therapy-induced complications, especially oral mucositis, could substantially reduce the risk of oral pain, oral and systemic infections and number of days in the hospital, and could improve quality of life and reduce health care costs. There are no universally effective agents or protocols to prevent toxicity.1 Mucositis develops in one to two weeks after treatment initiation and finally heals two to four weeks, if uncomplicated, after the treatment ends. Cryotherapy may help by reducing the amount of drug reaching the oral mucosa.
For this impeccable systematic review, the authors performed a comprehensive search until June of 2015 with no language restrictions. Two authors independently screened, assessed for eligibility, extracted data and assessed the risk of bias. A protocol for disagreement was in place.
They included fourteen RCTs with 1280 participants. All studies were judged as high risk of bias in the two blinding categories, but one can question whether masking the treatment for the patient and the researcher is even feasible for this intervention. Subgroup analysis was done according to type of cancer treatment. Clinical and statistical heterogeneity were calculated in the meta-analysis.
The authors presented the results for the main outcomes in a very user-friendly format summary, besides the forest plot. The applicability of the results is strengthened because the authors aligned the outcomes with the ones selected in COMET (Core Outcome Measures in Effectiveness Trials) Initiative's website.2 (www.cometinitiative.org)
In patients receiving 5FU, five RCTs with 444 participants showed that cryotherapy could reduce the risk of developing mucosits by 39%, the risk of developing moderate to severe by 48% and severe by 60%. For patients receiving melphalan-based therapy, five RCTs with 270 participants showed that cryotherapy could reduce the development of mucositis, severe and moderate mucositis and severe mucositis by 41%, 57% and 62% respectively, although the wide confidence intervals make those point estimates less reliable.
Such a simple, low cost intervention has the potential of benefiting many patients that otherwise may not have access to other more sophisticated, but not always better, approaches.
References
http://www.cancer.gov/about-cancer/treatment/side-effects/mouth-throat/oral-complications-hp-pdq#section/_13 [Accessed 19 August 2016]
http://www.cometinitiative.org [Accessed 19 August 2016]
Author information
Authors and Affiliations
Additional information
Address for correspondence: Luisa Fernandez Mauleffinch, Managing Editor, Cochrane Oral Health Group, School of Dentistry, The University of Manchester, JR Moore Building, Oxford Road, Manchester, M13 9PL, UK. E-mail: luisa.fernandez@manchester.ac.uk
Riley P, Glenny AM, Worthington HV, Littlewood A, Clarkson JE, McCabe MG. Interventions for preventing oral mucositis in patients with cancer receiving treatment: oral cryotherapy. Cochrane Database Syst Rev 2015; 12: Art. No.: CD011552. DOI: 10.1002/14651858.CD011552.pub2.
This paper is based on a Cochrane Review published in the Cochrane Library 2015, issue 12 (see http://www.thecochranelibrary.com for information). Cochrane Reviews are regularly updated as new evidence emerges and in response to feedback, and the Cochrane Library should be consulted for the most recent version of the review.
Rights and permissions
About this article
Cite this article
Spivakovsky, S. Oral cryotherapy reduced oral mucositis in patients having cancer treatments. Evid Based Dent 17, 80 (2016). https://doi.org/10.1038/sj.ebd.6401186
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ebd.6401186
- Springer Nature Limited
This article is cited by
-
Innovative intraoral cooling device better tolerated and equally effective as ice cooling
Cancer Chemotherapy and Pharmacology (2017)