Abstract
Spinocerebellar ataxia type 7 (SCA7) is a progressive neurodegenerative disorder resulting from abnormal expansion of an uninterrupted polyglutamine (polyQ) repeat in its disease protein, ataxin-7 (ATXN7). ATXN7 is part of Spt-Ada-Gcn5 acetyltransferase (SAGA), an evolutionarily conserved transcriptional coactivation complex with critical roles in chromatin remodeling, cell signaling, neurodifferentiation, mitochondrial health and autophagy. SCA7 is dominantly inherited and characterized by genetic anticipation and high repeat-length instability. Patients with SCA7 experience progressive ataxia, atrophy, spasticity, and blindness. There is currently no cure for SCA7, and therapies are aimed at alleviating symptoms to increase quality of life. Here, we report novel Drosophila lines of SCA7 with polyQ repeats in wild-type and human disease patient range. We find that ATXN7 expression has age- and polyQ repeat length-dependent reduction in fruit fly survival and retinal instability, concomitant with increased ATXN7 protein aggregation. These new lines will provide important insight on disease progression that can be used in the future to identify therapeutic targets for SCA7 patients.
Similar content being viewed by others
Introduction
Spinocerebellar ataxia type 7 (SCA7) is an inherited, autosomal dominant disease belonging to the family of polyglutamine (polyQ) neurodegenerative disorders1,2. The etiology of the polyQ family of diseases is in part, shared: toxic gain-of-function mutations—abnormal expansion of the triplet repeat CAG/A—yield longer than normal tracts of glutamine (Q)3,4,5,6,7,8. These elongated tracts cause protein misfolding and aggregation, particularly harmful to highly specialized neural cells. Although the expanded polyQ region is a shared feature in this neurodegenerative family, each causative disease protein is unique. Importantly, despite broad expression of disease genes, individual polyQ disorders affect different CNS regions and present distinct symptomology9.
Like other members of the polyQ family of disorders, SCA7 patients experience progressive neuronal loss in the cerebellum and brainstem that yield ataxia and early mortality, while unusually severe intergenerational repeat length instability, retinal degeneration, and blindness are features that are unique to SCA710. SCA7 results from abnormal CAG expansion in the ATXN7 gene, whose protein product is part of Spt-Ada-Gcn5 acetyltransferase (SAGA), a highly evolutionarily conserved transcriptional coactivation complex1,2. SAGA has chromatin remodeling activities essential for RNA polymerase II transcription, and also modifies autophagy, cell–cell interactions, mitochondrial homeostasis, and neuronal differentiation10,11. Additional work has implicated changes in gene expression, transcriptional regulation, and epigenetics in SCA7 pathogenesis, but the molecular mechanisms that underly its pathogenesis are not fully understood12,13,14,15.
Here, we report the generation and characterization of new Drosophila melanogaster models of SCA7 that ectopically express full-length, Myc-tagged, human ataxin-7 (ATXN7) protein with normal (Q10) or expanded (Q92) polyQ, within patient range. Both Q10 and Q92 proteins had variable, tissue-specific toxicity, with 92Q conferring stronger progressive effects on retinal cell stability and longevity. The advantages of Drosophila models of SCA7 include their flexible and precisely controlled genetics, allowing for rapid, cost-effective, longitudinal investigations aimed at understanding pathogenesis and discovering therapeutic opportunities.
Results
Wild-type and polyQ-expanded models of SCA7
Techniques we successfully used in the past were used to generate new Gal4-UAS Drosophila lines that express Myc-tagged human ATXN7 with either Q10 or Q9216,17,18. Normal polyQ tracts range from 7–27 CAG repeats in ATXN7 whereas full pathogenic penetrance occurs at 37–460 repeats19. A 5′ Myc tag was added to the full-length ATXN7 gene with either a 10- or 92-triplet CAG repeat (Fig. 1A) and sub-cloned into the pWALIUM10.moe vector (Fig. 1B). This cloning strategy yields a single-copy, phiC31-dependent insertion into the attP2 site on the third Drosophila chromosome20, in the same integration site and the same orientation as previously published polyQ family disease model flies16,17,18. We utilize this expression system and vector for its ability to easily modify protein expression, allowing us to both control the amount of disease protein expressed and compare across Drosophila polyQ models with similar protein expression levels17,21,22.
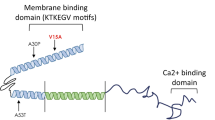
Generation of Drosophila SCA7 models. (A) Schematic of Myc-tagged ATXN7 Q10 and Q92 including construct-specific polyQ insertions. ATXN7 amino acid sequence, with 5X Myc tag (underlined) and polyQ repeat location (red) is also shown. (B) Top: diagrammatic representation of the cloning strategy used to insert ATXN7 cDNA into attP2 site. Bottom: PCR reactions from genomic DNA indicating that the transgene was integrated into the correct site in the proper orientation. (C–F) Western blots from control, Q10, and Q92 flies in (C) fly eyes, (D) pan-neuronally, (E) glia, and (F) all tissues. Quantifications are on the right of each blot. Q10 (105 kD) and Q92 (115 kD) indicated by blue arrows. Potential proteolysis products indicated with black brackets. The entire ataxin-7-positive signal was quantified in each case. qRT-PCR quantifications are found directly below Western blot quantifications. 10 fly heads (GMR-Gal4) or 5 whole flies (sqh-Gal4, elav-Gal4, repo-Gal4) were homogenized, depending on experiment. At least 3 biological replicates were used; ‘n’ indicated above panels. Significance determined by student t-test. *p > 0.05, ****p < 0.0001.
We assessed ATXN7 protein and mRNA levels in one-day-old flies when UAS expression was driven in fly eyes (GMR-Gal4, Fig. 1C), all neurons (elav-Gal4, Fig. 1D), all glia (repo-Gal4, Fig. 1E) or ubiquitously (sqh-Gal4, Fig. 1F). When ATXN7 expression was induced in either fly eyes (Fig. 1C) or ubiquitously (Fig. 1F) higher levels of Q92 mRNA and protein were observed in comparison to age-matched Q10 flies. ATXN7 mRNA and protein levels were similar, regardless of repeat length, when UAS expression was driven pan-neuronally (Fig. 1D). We also examined ATXN7 mRNA and protein levels when UAS expression was confined to glial cells: although we observed lower levels of Q92 mRNA when compared to Q10, protein levels were higher with the Q92 variant (Fig. 1E). Western blotting detected the presence of probable proteolysis products independent of driver (black brackets), suggesting high proteolytic activity of ATXN7 protein in all tissues tested, regardless of the polyQ length; cleavage of ATXN7 by caspases has been reported before23. When we immunoprecipitated ATXN7 using its N-terminal Myc tag, we observed that these species consist of at least some N-terminal fragments of this protein (supplemental Fig. 1).
As noted in the introduction, ATXN7 is part of the SAGA complex. We examined if the human protein that we introduced in the fruit fly retains some of its functions in relation to SAGA, including the ubiquitination status of histone H2B. As shown in supplemental Fig. 2, over-expression of ATXN7 Q10 and Q92 led to changes in the levels of H2B of the higher-molecular weight species that are consistent with its ubiquitinated forms. Additionally, the precise pattern of the migration of the H2B species identified differs between Q10 and Q92 ATXN7. These blots indicate that human ATXN7 is functionally competent in the fly, and suggest that its polyQ expansion may alter this functionality.
Effects of ataxin-7 expression on development and survival. A summary of experiments performed can be found in (A). Ubiquitous expression of Q10 reduced longevity in female flies but not males, and Q92 expression decreased female longevity (B,E). Pan-neuronal expression of Q92 decreased female longevity (C) and both Q10 and Q92 male flies had reduced survival compared to controls (F). Glial expression of ATXN7 Q10 reduced longevity in females and males, and glial Q92 expression did not (D,G). n > 200 flies for all groups, analyzed by log-rank.
As shown in Fig. 1, while there is general congruence between mRNA and protein levels of ATXN7 Q10 and Q92 when expressed in different tissues (Fig. 1C,D,F), this is not always the case (Fig. 1E), suggesting that different tissues handle the message and protein of ATXN7 differently. The transgene integration and binary expression system we utilized lead to similar levels of target expression (e.g., ref16,21,22,24,25,26,27,28,29,30,31,32). The difference in mRNA levels between the two different variants of ATXN7 in different tissues most likely represents differential message stability.
Toxicity in SCA7 model flies
We have previously shown that ectopic expression of human, polyQ-expanded disease proteins reduces survival in multiple fly models of SCA neurodegeneration16,17,18,25,26,28,33,34,35. To determine any potential detrimental effects of ATXN7, we first tracked longevity in sex-separated, male and female flies expressing ATXN7 harboring either Q10 or Q92 in all tissues, or specifically in neurons, or glial cells (Fig. 2).
Male and female controls flies developed normally and did not show reduced lifespan. Female flies ubiquitously expressing ATXN7 Q10 had shorter lifespan than background controls (heterozygous Gal4 flies backcrossed once into appropriate wild-type flies), and Q92 expression reduced lifespan further (Fig. 2B). In male flies, ubiquitous expression of either Q10 or Q92 had less pronounced lifespan effects, although Q10 expression conferred modest longevity protection, particularly at mid-life (Fig. 2E). Pan-neuronal expression of Q10 did not affect lifespan in female flies, but Q92 expression in female neurons decreased early-life longevity (Fig. 2C). In contrast, male flies expressing ATXN7 Q10 or Q92 in all neural cells had shorter lifespan than age-matched controls (Fig. 2F). Expressing two copies of each transgene in all adult neurons did not lead to markedly increased toxicity from the expression of either variant of ATXN7 (supplemental Fig. 3). When ATXN7 was restricted to glia, Q10 expression reduced longevity in both females and males, while Q92 expression was less toxic and did not negatively impact longevity (Fig. 2D,G). Taken together, these results suggest that ATXN7 expression has sexually dimorphic, tissue-dependent effects on survival in Drosophila, and that expression of wild-type, human ATXN7 Q10 protein can have toxic effects in flies even in the absence of SCA7-associated polyQ expansions.
Effects of ataxin-7 expression on motility. (A) Both Q10 and Q92 expression reduced mobility in female flies. (B) Female flies expressing Q10 in neurons have slightly better climbing speed than background control flies. (C) Glial expression of Q92 reduces female climbing speed across ages, and Q10 expression impairs climbing further. (D–F) Male flies expressing Q10 or Q92 have lower climbing speed than age-matched controls at days 5 and 10 whether ATXN7 is expressed in all tissues (D), neurons (E), or glia (F). n ≥ 100 per genotype, per sex, analyzed by linear regression.
Expression of polyQ disease proteins in flies causes reduced motility, as we and others have shown before16,24,30,32,34,36,37,38,39,40,41,42,43,44,45. To test if ATXN7 expression negatively impacts mobility, we longitudinally measured 3 weeks of climbing speed in male and female flies expressing either Q10 or Q92 in the same cell populations as our longevity assessments: ubiquitously, neurons, and glia. In female flies, ubiquitous or glial expression of either Q10 or Q92 reduced climbing speed across ages (Fig. 3A,C), and glial Q10 expression reduced climbing speed more than Q92 (Fig. 3C). In contrast, neuronal ATXN7 expression did not negatively impact female climbing speed, and flies expressing Q10 in neurons had slightly better mobility than both control and Q92 flies (Fig. 3B). In male flies, both Q10 and Q92 expression impaired mobility during the first week of adulthood whether ATXN7 expression was ubiquitous or specific to neurons or glia (Fig. 3D–F). These results further support sexually dimorphic toxic effects of ATXN7 expression that are not fully dependent on polyQ repeat length.
As noted above, in some tissues we observed differences in protein levels between Q10 and Q92 ATXN7. In the case of glial expression, higher abundance of the pathogenic form did not correlate with increased toxicity (Figs. 1, 2 and 3). In the case of ubiquitous expression, however, there was a correlation between higher levels of ATXN7 Q92 and increased toxicity from this species compared to the wild type counterpart (Figs. 1, 2 and 3). Lastly, in the case of pan-neuronal expression, where the protein levels of Q10 and Q92 ATXN7 were comparable, Q92 had a mildly more toxic effect. We used this expression pattern to evaluate a potentially direct correlation between amount of ATXN7 protein present versus its toxicity. As shown in supplemental Fig. 4, increasing the dosage of Q10 and Q92 levels by doubling transgene copies did not confer increased toxicity. Collectively, these findings indicate yet again tissue-specific effects of ATXN7 in this model of SCA7, and suggest that there is not a direct correlation between protein levels and its toxicity from human ATXN7 in the fruit fly. Next, we turned our attention to the fly eye for additional studies.
Effects of fly-eye specific ataxin-7 expression. (A) Pigmentation is normal in control and Q10 flies across ages. Q92 flies have pseudopupil (dotted circles) loss by adult day 56, n ≥ 10 per group. (B) Ability of ATXN7 Q10 and Q92 over-expression to counteract the effects of knockdown of endogenous Atxn7 and Atxn7-Like in fly eyes on days 1 and 30; n ≥ 10 per group. (C) Western blots from the groups in panel (B).
The Drosophila eye has been a stalwart model of neurogenerative diseases and genetic screens for many years43,44,45,46,47. We therefore used a fly eye specific SCA7 model to observe phenotypic neurodegeneration that may occur when ATXN7 is expressed, and to determine whether or not polyQ length or protein levels dictate degenerative phenotypes16,27,48. We have successfully used these tools in the past to detect changes that may be missed when disease protein expression is driven in other tissues16,27,48. When ATXN7 Q92 was selectively expressed in fly eyes (GMR-Gal4), pseudopupil loss, an indicator of disorganization of underlying structures16, was present by adult day 56 (Fig. 4A). Higher levels of expression of pathogenic ATXN7, but not of the normal version of this protein, coincided with earlier onset of the loss of the pseudopupil effect (supplemental Fig. 4).
We additionally examined the functionality of the human ATXN7 proteins in the fly by testing their ability to complement loss of endogenous Atxn7. As shown in Fig. 4B, knockdown of endogenous Atxn7 (but not of the closely related, Atxn7-like) led to loss of the pseudopupil by day 30. This was reversed by over-expression of either Q10 or Q92, human ATXN7, indicating functional human ATXN7 in the fly, similar to what we observed with H2B in supplemental Fig. 1. Interestingly, Western blotting from these eyes indicated a potential effect from endogenous Atxn7, and less so from Atxn7-like, on the protein levels of Q10 and Q92 (Fig. 4C). Altogether, we conclude that human ATXN7 retains function in the fly eye, and that the expanded version of this protein is mildly toxic to the fly eye. The Western blotting data that suggest a connection between endogenous and human ATXN7 in the fly eye require future attention to elucidate their interaction functionally and physically.
To further dissect the timeline of retinal degeneration, we quantified fluorescent intensity of membrane-targeted GFP (CD8-GFP), an indirect measurement of retinal integrity49. In both male and female flies, GFP expression was already lower in one-day-old flies expressing either ATXN7 Q10 or Q92 when compared to background controls expressing CD8-GFP without ATXN7 (Fig. 5A, top row and quantification on the right). By adult day 20, GFP expression was further reduced in flies expressing ATXN7 Q10, and Q92 expression in fly eyes exacerbated this phenotype, with more pronounced retinal cell loss observed by day 40 (Fig. 5A, middle and bottom rows, respectively). These results suggest progressive, polyQ-length dependent retinal degeneration, detected earlier and with more sensitivity in this model than observation of external eye structure with light microscopy.
Time course of structural and biochemical profile of ATXN7 effects in eyes. (A) CD8-GFP expression declines slightly with age in control flies (left panels, quantified at right; black histograms). Both Q10 (middle panels, green histograms) and Q92 (right panels, magenta histograms) have lower GFP fluorescence than age-matched control flies, and Q92 flies have lower GFP fluorescence than both control and Q10 flies by day 20; n ≥ 29. (B) 5 μM histological sections of control (left), Q10 (middle) and Q92 (right) flies at adult day 1 (upper panels). At adult day 56 (lower panels) Q92 flies have darkly stained aggregates, corneal disruption, loss of ommatidial boundaries (red bracket) and retinal disorganization. n ≥ 6. (C) Western blots of ATXN7 in fly eyes over time. SDS-resistant species are present in the upper portions of the blot in Q92 flies by adult day 35. Lysates from 20 fly heads and at least 2 biological replicates performed per group. (D) Western blots from flies expressing ATXN7 Q10 or Q92 in all tissues for the time course indicated in the panel.
Histological examination of fly eyes supports these findings, revealing progressive degeneration that was more prominent with age and in Q92 model flies (Fig. 5B). On adult day 1, eye morphology was unaffected regardless of genotype (Fig. 5B, top row). In contrast, Q92 flies develop darkly stained aggregation products, loss of ommatidial boundaries (red bracket) and prominent retinal disorganization by adult day 35, suggesting age- and polyQ-length dependent neurodegeneration (Fig. 5B, bottom row).
We next examined ATXN7 protein migration patterns in the fly eye-specific SCA7 model, a strong molecular indicator of polyQ family disease progression9,50,51. We have previously shown that aggregation of polyQ proteins precedes toxicity, and that aggregation levels of these proteins are an indicator of pathogenesis16,17,18,25,26,27. Eye-specific ATXN7 expression was visible in dissected Q10 and Q92 fly heads as early as adult day 1 (Fig. 5C). SDS-resistant smears, indicative of aggregated species that migrate more slowly through SDS-PAGE gels, were apparent by day 35 and still present on day 56 in ATXN7 Q92 flies only. The SDS-resistant species were concomitant with loss of the primary SDS-soluble band of ATXN7 protein in the Q92 version (Fig. 5C).
Considering the formation of SDS-resistant species from expression of ATXN7 Q92 over time in the fly eyes, we also tested whether a similar pattern is observed when human ATXN7 is expressed everywhere. As shown in Fig. 5D, the pathogenic variant of human ATXN7 also aggregates over time when expressed ubiquitously. Here, we also noticed a reduction in the levels of ATXN7 Q10 over time (Fig. 5D; a similar pattern is observed with pan-neuronal and pan-glial expression, as shown in supplemental Fig. 5). We wondered if this could be due to aggregative properties of the normal version of the protein that may not appear as SDS-resistant species. We examined this possibility by conducting soluble/pellet fractionations of ATXN7. Both Q10 and Q92 ATXN7 separate into soluble and pellet fractions at all three ages examined (supplemental Fig. 6). Taken together, these results indicate progressive, polyQ-dependent toxicity that correlates with higher Q92 protein levels in these new SCA7 model flies, suggesting that ATXN7 toxicity coincides with both polyQ expansion and protein aggregation. They also suggest aggregative properties of normal ATXN7 in this model, which may be additionally helpful to understand the biology of this protein in normal conditions and in disease.
Discussion
We described here new Drosophila lines of SCA7 that rely on the expression of full-length, human ATXN7 protein with a polyQ repeat length in either wild-type (Q10) or human SCA7 disease range (Q92). Compared to control flies, both Q10 and Q92 expression in neurons, fly eyes, or ubiquitously had variable, progressive degenerative effects; Q92 expression was deleterious to survival and in fly eyes, concomitant with increased aggregation over time. Our initial characterization of these new models opens the door for the generation of new isogenic lines that precisely dissect the molecular mechanisms driving disease progression in SCA7.
Fly eyes have been a valuable tool to aid our understanding of proteotoxic neurodegenerative disease progression17,18,28,33,49,52,53,54,55. We observe human ATXN7-dependent eye degeneration in an age- and polyQ length-dependent manner. This finding is especially important in the context of SCA7, as progressive blindness is a clinical presentation that is unique to this disorder of the polyQ family6,19. SCA7 is broadly classified into three categories: adult, young-adult, and early-childhood—classifications based on clinical presentation, age of onset, and polyQ repeat length56. Although common in earlier-onset diseases, patients who present in the fifth decade of life and later do not typically experience visual impairment19,56. With our fly models, we observe age- and polyQ repeat length-dependent decreases in GFP fluorescence when Q10 or Q92 proteins are expressed in the fly eye; histological sections reveal progressive retinal deterioration in Q92 model flies, without clear impact from the Q10 counterpart. These new tools will provide a valuable platform by which to determine the timeline of retinal phenotypes as well as potential genetic or pharmacological modifiers of blindness in SCA7 patients.
In contrast, when ATXN7 is expressed in all tissues, neurons, or glial cells, toxic effects on survival and motility are sex- and tissue-specific and do not fully depend on polyQ repeat length. Furthermore, we report a correlation between protein levels and toxic phenotypes, particularly when ATXN7 was expressed in fly eyes or ubiquitously. We and others have observed detrimental effects from overexpression of wild-type, human polyQ proteins in Drosophila17,57; the mechanisms underlying their pathogenicity are unclear. In Drosophila, loss of endogenous Atxn7 yields neurodegenerative phenotypes that are similar to overexpression of polyQ-expanded human ATXN758,59. In this study, we observed that both normal and pathogenic, human ATXN7 reverse structural defects due to knockdown of endogenous Atxn7. Atxn7 mutation uncouples SAGA’s normal function as a transcriptional coactivator from its protein quality control function, resulting in aberrant gene expression that yields reduced lifespan, impaired motility, and retinal degeneration58,59,60. It is possible that toxicity from Q10 expression results from disrupted endogenous Atxn7 function, while both loss-of-function and aggregation-related gain-of-function contribute to Q92 toxicity. Using these new lines, future work may differentiate the relative contribution of protein level, aggregation, and loss of endogenous ATXN7 function to SCA7 pathogenesis.
We report here sex-specific effects of ATXN7 Q10 and Q92 expression on both survival and motility. Clinically, SCA7 is dominantly inherited and does not present in a gender-specific way61. This difference between humans and our fly models of SCA7 can be leveraged toward the identification of therapeutic targets, looking for unique, specific factors that are protective. Sex determination in Drosophila is cell autonomous and reversible, controlled through alternative splicing of a single gene62. We and others have taken advantage of this genetic flexibility to identify the cellular source of variation between male and female flies62,63,64,65,66,67,68,69. As described above, male SCA7 model flies are protected from early mortality conferred by ubiquitous ATXN7 Q10 expression, but more sensitive to neurodegeneration when expression is confined to neurons. Follow-up investigations could be geared toward identifying molecular targets that are protective in particular sexes, and in particular tissues, with the goal of modulating those pathways to prevent neurodegeneration across sexes and age.
Finally, examination of protein levels in our Q10 and Q92 flies indicates the presence of proteolytic fragments that differ depending on age, polyQ length, and the tissue in which they are expressed (see Figs. 1,4, and 5). Proteolytic fragmentation of disease proteins is not uncommon in polyQ family disease models and is thought to modify aggregation, function, localization, and toxicity of disease proteins70,71,72,73,74. In SCA7, toxic protein fragments cause increased nuclear retention of ATXN7 and impair its normal functional interaction with SAGA70, and may yield post-translational modifications that prevent normal degradation75. Our results support these findings. Here, we observe gradually accumulating proteolytic cleavage products of Q10 expression in fly eyes, whereas Q92 fragments shift over time, a finding that correlates with more severe retinal degeneration. These models can be leveraged to precisely identify how proteolytic cleavage and posttranslational modifications define aggregation susceptibility and proteotoxicity in SCA7.
Together, these new Drosophila models provide a versatile, cost-effective system under precise genetic control that will enable expedient and targeted examination of the molecular pathways that drive SCA7 neurodegeneration. These tools will be used in the future to guide the generation of new therapeutics for SCA7 patients and their families.
Methods
Fly stocks and maintenance
elav-Gal4 (#458) and GMR-Gal4 (#8121) were obtained from the Bloomington Drosophila Stock Center. The RNAi lines of Atxn7(#102078) and Atxn7L3 (#100581) were obtained from Vienna Drosophila RNAi Center (VDRC). w−; UAS-CD8-GFP; GMR-Gal4 was described previously49. Gifted stocks used in this study were sqh-Gal4 (Daniel Kiehart, Duke University), repo-Gal4 (Vanessa Auld, University of British Columbia), y, w; +; attP2 (Jamie Roebuck, Duke University) and w1118 (Russ Finley, Wayne State University). To generate transgenic lines that express ATXN7Q10 (Q10) and ATXN7Q92 (Q92) through the Gal4-UAS system29, the DNA sequence of full length, human ATXN7 was inserted into pWALIUM10-moe plasmid (DNA Resource Core at Harvard Medical School, MA, USA) with a 5’ Myc tag and a construct-specific (Q10 or Q92) polyQ expansion. Constructs were injected by the Duke University Model System Injection Service into y, w; +; attP2. For transgene verification, genomic DNA were extracted from different founder lines using DNAzol (ThermoFisher, Waltham, MA USA) and PCR-amplified using the below primers.
white-end-F: 5′-TTCAATGATATCCAGTGCAGTAAAA-3′
attP2-3L-R: 5′-CTCTTTGCAAGGCATTACATCTG-3′.
Flies were housed in a 25 °C incubator on a 12:12 h light:dark cycle at 40% relative humidity. Control flies for all Gal4-UAS experiments consisted of heterozygous Gal4 lines backcrossed into y, w; +; attP2 and/or w1118 flies, depending on experiment. Adult progeny were synchronized by collecting within 12 h of eclosion over a 24 h time period. Groups of 10–20 age- and sex-matched flies were immediately transferred into narrow polypropylene vials containing 5 mL of standard 2% agar, 10% sucrose, 10% yeast with appropriate preservatives. Food vials were changed every second to third day.
Western blots
Five whole flies or fifteen fly heads per biological replicate, depending on experiment, were homogenized in boiling lysis buffer (50 mM Tris pH 6.8, 2% SDS, 10% glycerol, 100 mM dithiothreitol), sonicated, boiled for 10 min, and centrifuged at 13,300xg at room temperature for 10 min. Western blots were developed ChemiDoc (Bio-Rad, Hercules, CA, USA) and quantified with ImageLab (Bio-Rad, Hercules, CA, USA). For ATXN7, protein levels were measured by quantifying the entire lane (Anti-Myc). Q92 expression was normalized to Q10 expression, taking into account total protein for each genotype using direct blue. Primary antibody used was Mouse anti-Myc (9B11) (1:1000, Cell signaling, Danvers, MA, USA); Secondary antibody: peroxidase-conjugated anti-mouse (1:5000, Jackson Immunoresearch, West Grove, PA, USA). For direct blue staining, PVDF membranes were submerged for 10 min in 0.008% Direct Blue 71 (Sigma-Aldrich, St. Louis, MO, USA) in 40% ethanol and 10% acetic acid, rinsed in 40% ethanol/10% acetic acid, air dried, and imaged. Western blots were performed using at least 3 biological replicates, and statistical analysis (student t-test) was performed in GraphPad Prism (San Diego, CA, USA). Supplemental table 1 lists the antibodies used in this work.
Immunoprecipitation
Ten flies were homogenized in 800 µL of buffer consisting of NETN lysis buffer (50 mM Tris, pH 7.5, 150 mM NaCl, 0.5% Nonidet P-40) supplemented with protease inhibitor cocktail (PI; Sigma-Aldrich, St. Louis, MO, USA). Homogenates were centrifuged for 10 min at 10,000×g, 4 °C. Supernatant was then combined with bead-bound anti-Myc antibody and tumbled at 4 °C for 2 h. Beads were rinsed 3X with NETN, then bead-bound complexes were eluted through Laemmli buffer (Bio-Rad, Hercules, CA, USA) and boiling.
Soluble/pellet preparation
Five flies per sample were homogenized in 100 µL NETN lysis buffer with PI. The homogenates were centrifuged at 20,000×g at 4 °C for 30 min. The supernatant was removed and supplemented with 20 µL of 6% SDS buffer. The pellet was resuspended in 100 µL of Laemmli buffer (Bio-Rad, Hercules, CA, USA) and supplemented with 20 µL SDS lysis buffer. Samples were then boiled for 10 min. Equal volumes were loaded on 4–20% gradient SDS-PAGE gels for Western blotting.
qRT-PCR
Total RNA was extracted by using TRIzol reagent (Invitrogen, Waltham, MA, USA). Extracted RNA was treated with TURBO DNAse (Invitrogen, Waltham, MA, USA) to remove contaminating DNA. Reverse transcription was performed with the High-Capacity cDNA Reverse Transcription Kit (ABI, Waltham, MA, USA). Messenger RNA levels were quantified by using the StepOnePlus Real-Time PCR System with Fast SYBR Green Master Mix (ABI, Waltham, MA, USA). rp49 was used as internal control. At least three biological replicates were used, and statistical analysis (student t-test) was performed in GraphPad Prism (San Diego, CA, USA).
Primers:
ATXN7-F: 5′- CTGCTCTCATCTACCTGCATCTC -3′;
ATXN7-R: 5′- TAGTGCTGTTACCAGAAGACTCCTT -3′;
rp49-F: 5′- AGATCGTGAAGAAGCGCACCAAG-3′;
rp49-R: 5′- CACCAGGAACTTCTTGAATCCGG-3′.
Lifespan
At least 200 adults were age-matched and separated by sex within 12 h of eclosion. Flies were transferred to narrow polypropylene vials containing 5 mL of standard 2% agar, 10% sucrose, 10% yeast food. Flies were transferred and scored for death events every 2–3 days until no flies remained. Survival curves were analyzed by log-rank in GraphPad Prism (San Diego, CA, USA).
Climbing speed
Negative geotaxis was assessed using a modified Rapid Iterative Negative Geotaxis (RING) assay in groups of at least 100 flies, as described32,42,76. Briefly, vials of 20 flies were briskly tapped down, then measured for climbing distance after 2 s of inducing the negative geotaxis instinct. For each group of vials, an average of 5 consecutive trials was calculated and batch processed using ImageJ (Bethesda, MD). Flies were longitudinally tested 2–3 times per week for 3 weeks. Between assessments, flies were returned to food vials and housed normally as described above. Negative geotaxis results were analyzed using linear regression (slope, y-intercept) in GraphPad Prism (San Diego, CA, USA). All negative geotaxis experiments were performed in duplicate, with one complete trial shown in each graph.
Fluorescence measurements
All fluorescence images were taken with an Olympus BX53 microscope and CellSens software (Olympus, Waltham, MA, USA). The same objective (4X) and camera settings (ISO 200, exposure time 500 ms capture time for each sample) were used for all images. Control and experimental flies were all imaged on the same day. ImageJ’s freehand tool was used to outline and to quantify fluorescence readings from each fly eye. GFP expression was statistically analyzed by ANOVA in GraphPad Prism (San Diego, CA, USA). n ≥ 29 flies for all groups.
Histology
Adult flies whose proboscises and wings were removed were fixed in 2% glutaraldehyde/2% paraformaldehyde and 0.05% Triton X-100 in phosphate buffered saline overnight at 4 °C. Fixed flies were later dehydrated in a series of 30, 50, 75 and 100% ethanol and propylene oxide, embedded in Poly/Bed812 (Polysciences, Warrington, PA, USA), sectioned at 5 μm and then stained with Toluidine Blue. n ≥ 6 flies for all groups.
Data availability
Fly lines, plasmids, and source data are available upon request. Each gel and blot presented in every figure has a full-length (uncropped) image included in supplemental Fig 7. The authors affirm that all data necessary for confirming the conclusions of the article are present within the article and figures. To request data from this study, please contact W-L. T at wtsou@wayne.edu.
References
Helmlinger, D. et al. Ataxin-7 is a subunit of GCN5 histone acetyltransferase-containing complexes. Hum. Mol. Genet. 13, 1257–1265. https://doi.org/10.1093/hmg/ddh139 (2004).
Garden, G. A. & La Spada, A. R. Molecular pathogenesis and cellular pathology of spinocerebellar ataxia type 7 neurodegeneration. Cerebellum 7, 138–149. https://doi.org/10.1007/s12311-008-0027-y (2008).
Zoghbi, H. Y. & Orr, H. T. Glutamine repeats and neurodegeneration. Annu. Rev. Neurosci. 23, 217–247. https://doi.org/10.1146/annurev.neuro.23.1.217 (2000).
La Spada, A. R. & Taylor, J. P. Polyglutamines placed into context. Neuron 38, 681–684. https://doi.org/10.1016/s0896-6273(03)00328-3 (2003).
Todi, S. V., Williams, A. & Paulson, H. in Molecular Neurology (ed S. G. Waxman) Ch. 17, 257–276 (Academic Press, 2007).
Paulson, H. L., Shakkottai, V. G., Clark, H. B. & Orr, H. T. Polyglutamine spinocerebellar ataxias - from genes to potential treatments. Nat. Rev. Neurosci. 18, 613–626. https://doi.org/10.1038/nrn.2017.92 (2017).
Lieberman, A. P., Shakkottai, V. G. & Albin, R. L. Polyglutamine repeats in neurodegenerative diseases. Annu. Rev. Pathol. https://doi.org/10.1146/annurev-pathmechdis-012418-012857 (2018).
Buijsen, R. A. M., Toonen, L. J. A., Gardiner, S. L. & van Roon-Mom, W. M. C. Genetics, mechanisms, and therapeutic progress in polyglutamine spinocerebellar ataxias. Neurotherapeutics https://doi.org/10.1007/s13311-018-00696-y (2019).
Klockgether, T., Mariotti, C. & Paulson, H. L. Spinocerebellar ataxia. Nat. Rev. Dis. Primers 5, 24. https://doi.org/10.1038/s41572-019-0074-3 (2019).
Niewiadomska-Cimicka, A. & Trottier, Y. Molecular targets and therapeutic strategies in spinocerebellar ataxia type 7. Neurotherapeutics 16, 1074–1096. https://doi.org/10.1007/s13311-019-00778-5 (2019).
Bonnet, J. et al. The SAGA coactivator complex acts on the whole transcribed genome and is required for RNA polymerase II transcription. Gene Dev. 28, 1999–2012. https://doi.org/10.1101/gad.250225.114 (2014).
Abou-Sleymane, G. et al. Polyglutamine expansion causes neurodegeneration by altering the neuronal differentiation program. Hum. Mol. Genet. 15, 691–703. https://doi.org/10.1093/hmg/ddi483 (2006).
Kizilyaprak, C., Spehner, D., Devys, D. & Schultz, P. The linker histone H1C contributes to the SCA7 nuclear phenotype. Nucleus 2, 444–454. https://doi.org/10.4161/nucl.2.5.17843 (2011).
Tan, J. Y. et al. Cross-talking noncoding RNAs contribute to cell-specific neurodegeneration in SCA7. Nat. Struct. Mol. Biol. 21, 955–961. https://doi.org/10.1038/nsmb.2902 (2014).
Yoo, S. Y. et al. SCA7 knockin mice model human SCA7 and reveal gradual accumulation of mutant ataxin-7 in neurons and abnormalities in short-term plasticity. Neuron 37, 383–401. https://doi.org/10.1016/s0896-6273(02)01190-x (2003).
Johnson, S. L. et al. Differential toxicity of ataxin-3 isoforms in Drosophila models of spinocerebellar ataxia type 3. Neurobiol. Dis. 132, 104535. https://doi.org/10.1016/j.nbd.2019.104535 (2019).
Tsou, W. L., Qiblawi, S. H., Hosking, R. R., Gomez, C. M. & Todi, S. V. Polyglutamine length-dependent toxicity from alpha1ACT in Drosophila models of spinocerebellar ataxia type 6. Biol. Open 5, 1770–1775. https://doi.org/10.1242/bio.021667 (2016).
Tsou, W. L. et al. DnaJ-1 and karyopherin alpha3 suppress degeneration in a new Drosophila model of spinocerebellar ataxia type 6. Hum. Mol. Genet. 24, 4385–4396. https://doi.org/10.1093/hmg/ddv174 (2015).
La Spada, A. R. in GeneReviews((R)) (eds M. P. Adam et al.) (1993).
Groth, A. C., Fish, M., Nusse, R. & Calos, M. P. Construction of transgenic Drosophila by using the site-specific integrase from phage phi C31. Genetics 166, 1775–1782. https://doi.org/10.1534/genetics.166.4.1775 (2004).
Markstein, M., Pitsouli, C., Villalta, C., Celniker, S. E. & Perrimon, N. Exploiting position effects and the gypsy retrovirus insulator to engineer precisely expressed transgenes. Nat. Genet. 40, 476–483. https://doi.org/10.1038/ng.101 (2008).
Ni, J. Q. et al. Vector and parameters for targeted transgenic RNA interference in Drosophila melanogaster. Nat. Methods 5, 49–51. https://doi.org/10.1038/nmeth1146 (2008).
Johnson, S. L., Tsou, W. L., Prifti, M. V., Harris, A. L. & Todi, S. V. A survey of protein interactions and posttranslational modifications that influence the polyglutamine diseases. Front. Mol. Neurosci. 15, 974167. https://doi.org/10.3389/fnmol.2022.974167 (2022).
Tsou, W. L. et al. DnaJ-1 and karyopherin alpha-3 suppress degeneration in a new Drosophila model of spinocerebellar ataxia type 6. Hum. Mol. Genet. https://doi.org/10.1093/hmg/ddv174 (2015).
Sutton, J. R. et al. Interaction of the polyglutamine protein ataxin-3 with Rad23 regulates toxicity in Drosophila models of spinocerebellar ataxia type 3. Hum. Mol. Genet. 26, 1419–1431. https://doi.org/10.1093/hmg/ddx039 (2017).
Ristic, G., Sutton, J. R., Libohova, K. & Todi, S. V. Toxicity and aggregation of the polyglutamine disease protein, ataxin-3 is regulated by its binding to VCP/p97 in Drosophila melanogaster. Neurobiol. Disease 116, 78–92. https://doi.org/10.1016/j.nbd.2018.04.013 (2018).
Johnson, S. L., Ranxhi, B., Libohova, K., Tsou, W. L. & Todi, S. V. Ubiquitin-interacting motifs of ataxin-3 regulate its polyglutamine toxicity through Hsc70–4-dependent aggregation. Elife. https://doi.org/10.7554/eLife.60742 (2020).
Johnson, S. L. et al. Drosophila as a model of unconventional translation in spinocerebellar ataxia type 3. Cells-Basel. https://doi.org/10.3390/cells11071223 (2022).
Casci, I. & Pandey, U. B. A fruitful endeavor: Modeling ALS in the fruit fly. Brain Res. 1607, 47–74. https://doi.org/10.1016/j.brainres.2014.09.064 (2015).
Pandey, M. & Rajamma, U. Huntington’s disease: The coming of age. J. Genet. 97, 649–664 (2018).
Brand, A. H. & Perrimon, N. Targeted gene-expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993).
Perrimon, N., Bonini, N. M. & Dhillon, P. Fruit flies on the front line: The translational impact of Drosophila. Dis. Model Mech. 9, 229–231. https://doi.org/10.1242/dmm.024810 (2016).
Blount, J. R., Johnson, S. L., Libohova, K., Todi, S. V. & Tsou, W. L. Degron capability of the hydrophobic C-terminus of the polyglutamine disease protein, ataxin-3. J. Neurosci. Res. 98, 2096–2108. https://doi.org/10.1002/jnr.24684 (2020).
Sujkowski, A., Richardson, K., Prifti, M. V., Wessells, R. J. & Todi, S. V. Endurance exercise ameliorates phenotypes in Drosophila models of spinocerebellar ataxias. Elife https://doi.org/10.7554/eLife.75389 (2022).
Blount, J. R. et al. Ubiquitin-binding site 2 of ataxin-3 prevents its proteasomal degradation by interacting with Rad23. Nat. Commun. https://doi.org/10.1038/ncomms5638 (2014).
Jackson, G. R. et al. Polyglutamine-expanded human huntingtin transgenes induce degeneration of Drosophila photoreceptor neurons. Neuron 21, 633–642 (1998).
Fernandez-Funez, P. et al. Identification of genes that modify ataxin-1-induced neurodegeneration. Nature 408, 101–106 (2000).
Bonini, N. M. & Fortini, M. E. Human neurodegenerative disease modeling using Drosophila. Annu. Rev. Neurosci. 26, 627–656 (2003).
Romero, E. et al. Suppression of neurodegeneration and increased neurotransmission caused by expanded full-length huntingtin accumulating in the cytoplasm. Neuron 57, 27–40. https://doi.org/10.1016/j.neuron.2007.11.025 (2008).
Hsu, T. C. et al. Deactivation of TBP contributes to SCA17 pathogenesis. Hum. Mol. Genet. 23, 6878–6893. https://doi.org/10.1093/hmg/ddu410 (2014).
Sutton, J. R. et al. Interaction of the polyglutamine protein ataxin-3 with Rad23 regulates toxicity in Drosophila models of spinocerebellar ataxia type 3. Hum. Mol. Genet. https://doi.org/10.1093/hmg/ddx039 (2017).
Ristic, G., Sutton, J. R., Libohova, K. & Todi, S. V. Toxicity and aggregation of the polyglutamine disease protein, ataxin-3 is regulated by its binding to VCP/p97 in Drosophila melanogaster. Neurobiol. Dis. 116, 78–92. https://doi.org/10.1016/j.nbd.2018.04.013 (2018).
Rosas-Arellano, A., Estrada-Mondragon, A., Pina, R., Mantellero, C. A. & Castro, M. A. The tiny Drosophila melanogaster for the biggest answers in Huntington’s disease. Int. J. Mol. Sci. https://doi.org/10.3390/ijms19082398 (2018).
Ueyama, M. & Nagai, Y. Repeat expansion disease models. Adv. Exp. Med. Biol. 1076, 63–78. https://doi.org/10.1007/978-981-13-0529-0_5 (2018).
Patel, N. et al. Phenotypic defects from the expression of wild-type and pathogenic TATA-Binding Proteins in new Drosophila models of Spinocerebellar Ataxia Type 17. G3 (Bethesda), https://doi.org/10.1093/g3journal/jkad180 (2023).
Matsumoto, T., Takeyama, K., Sato, T. & Kato, S. Androgen receptor functions from reverse genetic models. J. Steroid Biochem. Mol. Biol. 85, 95–99. https://doi.org/10.1016/s0960-0760(03)00231-0 (2003).
Mishra, M. & Knust, E. Analysis of the drosophila compound eye with light and electron microscopy. Methods Mol. Biol. 345–364, 2019. https://doi.org/10.1007/978-1-4939-8669-9_22 (1834).
Passarella, D. & Goedert, M. Beta-sheet assembly of Tau and neurodegeneration in Drosophila melanogaster. Neurobiol. Aging 72, 98–105. https://doi.org/10.1016/j.neurobiolaging.2018.07.022 (2018).
Smylla, T. K., Wagner, K. & Huber, A. Application of fluorescent proteins for functional dissection of the drosophila visual system. Int. J. Mol. Sci. https://doi.org/10.3390/ijms22168930 (2021).
St Johnston, D. The art and design of genetic screens: Drosophila melanogaster. Nat. Rev. Genet. 3, 176–188. https://doi.org/10.1038/nrg751 (2002).
Ashraf, N. S. et al. Druggable genome screen identifies new regulators of the abundance and toxicity of ATXN3, the Spinocerebellar Ataxia type 3 disease protein. Neurobiol. Dis. 137, 104697. https://doi.org/10.1016/j.nbd.2019.104697 (2020).
Burr, A. A., Tsou, W. L., Ristic, G. & Todi, S. V. Using membrane-targeted green fluorescent protein to monitor neurotoxic protein-dependent degeneration of drosophila eyes. J. Neurosci. Res. 92, 1100–1109. https://doi.org/10.1002/jnr.23395 (2014).
Costa Mdo, C. & Paulson, H. L. Toward understanding Machado-Joseph disease. Prog. Neurobiol. 97, 239–257. https://doi.org/10.1016/j.pneurobio.2011.11.006 (2012).
Lieberman, A. P., Shakkottai, V. G. & Albin, R. L. Polyglutamine repeats in neurodegenerative diseases. Annu. Rev. Pathol. 14, 1–27. https://doi.org/10.1146/annurev-pathmechdis-012418-012857 (2019).
Bonini, N. M. in Molecular Biology of the Cell Vol. 10 350a (1999).
Bonini, N. M. & Fortini, M. E. Human neurodegenerative disease modeling using Drosophila. Annu. Rev. Neurosci. 26, 627–656. https://doi.org/10.1146/annurev.neuro.26.041002.131425 (2003).
Tsou, W. L. et al. Ubiquitination regulates the neuroprotective function of the deubiquitinase ataxin-3 in vivo. J. Biol. Chem. 288, 34460–34469. https://doi.org/10.1074/jbc.M113.513903 (2013).
McGurk, L. & Bonini, N. M. Protein interacting with C kinase (PICK1) is a suppressor of spinocerebellar ataxia 3-associated neurodegeneration in Drosophila. Hum. Mol. Genet. 21, 76–84. https://doi.org/10.1093/hmg/ddr439 (2012).
Goswami, R. et al. The molecular basis of spinocerebellar ataxia type 7. Front. Neurosci. 16, 818757. https://doi.org/10.3389/fnins.2022.818757 (2022).
Fernandez-Funez, P. et al. Identification of genes that modify ataxin-1-induced neurodegeneration. Nature 408, 101–106. https://doi.org/10.1038/35040584 (2000).
Latouche, M. et al. A conditional pan-neuronal Drosophila model of spinocerebellar ataxia 7 with a reversible adult phenotype suitable for identifying modifier genes. J. Neurosci. 27, 2483–2492. https://doi.org/10.1523/JNEUROSCI.5453-06.2007 (2007).
Mohan, R. D. et al. Loss of Drosophila ataxin-7, a SAGA subunit, reduces H2B ubiquitination and leads to neural and retinal degeneration. Genes Dev 28, 259–272. https://doi.org/10.1101/gad.225151.113 (2014).
Cloud, V. et al. Ataxin-7 and non-stop coordinate SCAR protein levels, subcellular localization, and actin cytoskeleton organization. Elife https://doi.org/10.7554/eLife.49677 (2019).
Gardiner, S. L. et al. Prevalence of carriers of intermediate and pathological polyglutamine disease-associated alleles among large population-based cohorts. JAMA Neurol. 76, 650–656. https://doi.org/10.1001/jamaneurol.2019.0423 (2019).
Verhulst, E. C., van de Zande, L. & Beukeboom, L. W. Insect sex determination: It all evolves around transformer. Curr. Opin. Genet. Dev. 20, 376–383. https://doi.org/10.1016/j.gde.2010.05.001 (2010).
Sujkowski, A., Ramesh, D., Brockmann, A. & Wessells, R. Octopamine drives endurance exercise adaptations in Drosophila. Cell Rep. 21, 1809–1823. https://doi.org/10.1016/j.celrep.2017.10.065 (2017).
Sujkowski, A. & Wessells, R. Using Drosophila to understand biochemical and behavioral responses to exercise. Exercise Sport Sci. Rev. 46, 112–120. https://doi.org/10.1249/JES.0000000000000139 (2018).
Chan, Y. B. & Kravitz, E. A. Specific subgroups of FruM neurons control sexually dimorphic patterns of aggression in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 104, 19577–19582. https://doi.org/10.1073/pnas.0709803104 (2007).
Dauwalder, B., Tsujimoto, S., Moss, J. & Mattox, W. The Drosophila takeout gene is regulated by the somatic sex-determination pathway and affects male courtship behavior. Gene Dev. 16, 2879–2892. https://doi.org/10.1101/gad.1010302 (2002).
Ferveur, J. F., Stortkuhl, K. F., Stocker, R. F. & Greenspan, R. J. Genetic feminization of brain structures and changed sexual orientation in male Drosophila. Science 267, 902–905. https://doi.org/10.1126/science.7846534 (1995).
Mundiyanapurath, S., Chan, Y. B., Leung, A. K. & Kravitz, E. A. Feminizing cholinergic neurons in a male Drosophila nervous system enhances aggression. Fly (Austin) 3, 179–184 (2009).
Steinmann-Zwicky, M. Sex determination of the Drosophila germ line: tra and dsx control somatic inductive signals. Development 120, 707–716 (1994).
Mookerjee, S. et al. Posttranslational modification of ataxin-7 at lysine 257 prevents autophagy-mediated turnover of an N-terminal caspase-7 cleavage fragment. J. Neurosci. 29, 15134–15144. https://doi.org/10.1523/Jneurosci.4720-09.2009 (2009).
Zhou, H. et al. Huntingtin forms toxic NH2-terminal fragment complexes that are promoted by the age-dependent decrease in proteasome activity. J. Cell. Biol. 163, 109–118. https://doi.org/10.1083/jcb.200306038 (2003).
Harmuth, T. et al. Mitochondrial morphology, function and homeostasis are impaired by expression of an N-terminal calpain cleavage fragment of ataxin-3. Front. Mol. Neurosci. 11, 368. https://doi.org/10.3389/fnmol.2018.00368 (2018).
Ishiguro, T. et al. The carboxy-terminal fragment of alpha(1A) calcium channel preferentially aggregates in the cytoplasm of human spinocerebellar ataxia type 6 Purkinje cells. Acta Neuropathol. 119, 447–464. https://doi.org/10.1007/s00401-009-0630-0 (2010).
Kim, Y. J. et al. Caspase 3-cleaved N-terminal fragments of wild-type and mutant huntingtin are present in normal and Huntington’s disease brains, associate with membranes, and undergo calpain-dependent proteolysis. Proc. Natl. Acad. Sci. USA 98, 12784–12789. https://doi.org/10.1073/pnas.221451398 (2001).
Young, J. E. et al. Proteolytic cleavage of ataxin-7 by caspase-7 modulates cellular toxicity and transcriptional dysregulation. J. Biol. Chem. 282, 30150–30160. https://doi.org/10.1074/jbc.M705265200 (2007).
Damschroder, D., Cobb, T., Sujkowski, A. & Wessells, R. Drosophila endurance training and assessment and its effects on systemic adaptations. BioProtocols https://doi.org/10.21769/BioProtoc.3037 (2017).
Acknowledgements
We extend our gratitude to Pegi Laci and Racquel Harrison for help with conducting lifespan and mobility assays.
Funding
This study was supported in part by NINDS R01NS086778 (SVT).
Author information
Authors and Affiliations
Contributions
A.S.: conceptualization, data curation, software, formal analysis, validation, investigation, visualization, methodology, and writing and editing. B.R.: data curation, validation, investigation, methodology. Z.R.B.: data curation, validation, investigation, methodology. Z.M.C.: data curation, validation, investigation, methodology. M.V.P.: data curation, validation, investigation, methodology. Z.Q.: data curation, validation, investigation, methodology. N.A.: data curation, validation, investigation, methodology. S.V.T.: conceptualization, data curation, funding acquisition, software, formal analysis, validation, investigation, visualization, methodology, and writing and editing. W-L.T.: conceptualization, data curation, software, formal analysis, validation, investigation, visualization, methodology, and writing and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sujkowski, A., Ranxhi, B., Bangash, Z.R. et al. Progressive degeneration in a new Drosophila model of spinocerebellar ataxia type 7. Sci Rep 14, 14332 (2024). https://doi.org/10.1038/s41598-024-65172-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-65172-4
- Springer Nature Limited