Abstract
Esophageal adenocarcinoma carries a poor prognosis associated with a 5-year survival rate of 12.5–20%. Therefore, a new therapeutic modality is needed for this lethal tumor. Carnosol is a phenolic diterpene purified from the herbs such as rosemary and Mountain desert sage and has been shown to have anticancer activities in multiple cancers. In this study we examined the effect of carnosol on cell proliferation in esophageal adenocarcinoma cells. We found that carnosol dose-dependently decreased cell proliferation in FLO-1 esophageal adenocarcinoma cells and significantly increased caspase-3 protein, indicating that carnosol decreases cell proliferation and increases cell apoptosis in FLO-1 cells. Carnosol significantly increased H2O2 production and N-acetyl cysteine, a reactive oxygen species (ROS) scavenger, significantly inhibited carnosol-induced decrease in cell proliferation, indicating that ROS may mediate carnosol-induced decrease in cell proliferation. Carnosol-induced decrease in cell proliferation was partially reversed by NADPH oxidase inhibitor apocynin, suggesting that NADPH oxidases may be partially involved in carnosol’s effect. In addition, carnosol significantly downregulated SODD protein and mRNA expression and knockdown of SODD significantly inhibited the carnosol-induced reduction in cell proliferation, suggesting that downregulation of SODD may contribute to carnosol-induced reduction in cell proliferation. We conclude that carnosol dose-dependently decreased cell proliferation and significantly increased caspase-3 protein. Carnosol’s effect may be through the overproduction of ROS and the downregulation of SODD. Carnosol might be useful for the treatment of esophageal adenocarcinoma.
Similar content being viewed by others
Introduction
Esophageal adenocarcinoma has increased in incidence over the past several decades1,2,3. The major risk factor for esophageal adenocarcinoma is Barrett’s esophagus4, which carries nearly a 30–125-fold increased risk for the development of esophageal adenocarcinoma. The cancer incidence in Barrett’s esophagus is about 0.5–1.0% per year. The mechanisms of the progression from Barrett’s esophagus to adenocarcinoma are not fully understood. Many genetic and epigenetic alterations, chromosomal gains, and losses, and hypermethylation of gene promoters may be involved in this progression5,6. Esophageal adenocarcinoma has a poor prognosis associated with a median survival of less than 1 year7,8 and a 5-year survival rate of 12.5–20%9,10. Therefore, a new therapeutic modality is needed for this lethal tumor.
Carnosol is a phenolic diterpene purified from the herbs such as rosemary and Mountain desert sage, and has been shown to have anticancer activities in multiple cancers, including colon11, breast12, stomach13 and prostate14. It may inactivate STAT3 through the production of reactive oxygen species (ROS), thus inhibiting tumor migration and growth in breast cancer cells12. Carnosol may selectively inhibit the p300 histone acetyl transferase15 and triggers a ROS-dependent ER-stress response through activation of the three ER stress sensor pathways in breast cancer cells16. ROS dependent inactivation of STAT3 also mediates carnosol-induced apoptosis in human colon cancer HCT116 cells11. In gastric cancer, carnosol suppresses gastric cancer growth via inhibiting the RSK-CREB signaling pathway13. Whether carnosol has anticancer activity in esophageal adenocarcinoma is not known. The aim of this study is to examine the effect of carnosol on cell proliferation in esophageal adenocarcinoma cells and whether its effect is mediated by the silencer of death domains (SODD).
Results
Carnosol reduced cell proliferation and increased cell apoptosis in esophageal adenocarcinoma cells
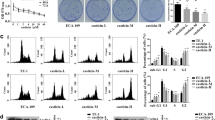
We first examined whether carnosol decreases cell proliferation by using WST-1 cell proliferation assay. We found that carnosol dose-dependently decreased cell proliferation in FLO-1 esophageal adenocarcinoma cells (Fig. 1, ANOVA, P < 0.001). To further confirm this result, we performed Western blot analyses by using caspase-3 antibody. We found that carnosol significantly increased caspase-3 protein (Fig. 2, t test, P < 0.05), indicating that carnosol increases cell apoptosis in esophageal adenocarcinoma cells. These data suggest that carnosol decreases cell proliferation and increases cell apoptosis in FLO-1 cells.
Effect of carnosol on caspase 3. (A) A typical image of Western blot analysis and (B) summarized data showed that carnosol (10–4 M, 24 h) significantly increased caspase-3 protein, indicating that carnosol increases cell apoptosis in FLO-1 cells. N = 4, * t-test, P < 0.05. Original blots are presented in Supplementary Figure 1.
Reactive oxygen species (ROS) may mediate carnosol’s effect
Intracellular reactive oxygen species may play an important role in cell apoptosis. Therefore, we examined the effect of ROS on carnosol-induced decrease in cell proliferation by using N-acetyl cysteine (NAC), a ROS scavenger, on cell proliferation. As shown in Fig. 3A, pre-treatment with NAC for 1 h significantly inhibited carnosol-induced decrease in cell proliferation (ANOVA, P < 0.001), indicating that ROS may mediate carnosol-induced decrease in cell proliferation.
Reactive oxygen species (ROS) may mediate carnosol’s effect. (A) Pre-treatment with N-acetyl cysteine (NAC, 10–6 M), a ROS scavenger, for 1 h significantly inhibited carnosol-induced decrease in cell proliferation, indicating that ROS may mediate carnosol (10–4 M, 24 h)-induced decrease in cell proliferation. N = 3, ANOVA, *P < 0.001, compared with vehicle control; **P < 0.001 compared with carnosol. (B) Hydrogen peroxide in the culture medium was measured. Carnosol (10–4 M, 3 h) significantly increased H2O2 production, suggesting that ROS may contribute at least in part to carnosol’s effect. N = 3, *t test, P < 0.05.
To confirm this result, we measured hydrogen peroxide in the culture medium. Cells were treated with vehicle or carnosol (10–4 M) for 3 h and then culture medium was collected for measurement. As shown in Fig. 3B, carnosol significantly increased H2O2 production (t test, P < 0.05), suggesting that ROS may contribute at least in part to carnosol’s effect.
NADPH oxidases may be involved in carnosol’s effect
We have shown that NADPH oxidases were present in FLO-1 cells17. Therefore, we examined the role of NADPH oxidases in carnosol-induced decrease in cell proliferation. Figure 4 showed that carnosol-induced decrease in cell proliferation was partially reversed by NADPH oxidase inhibitor apocynin (ANOVA, P < 0.0001), suggesting that NADPH oxidases may be partially involved in carnosol’s effect.
NADPH oxidases may be involved in carnosol’s effect. Carnosol (10–4 M, 24 h)-induced decrease in cell proliferation was partially reversed by NADPH oxidase inhibitor apocynin (10–6 M), suggesting that NADPH oxidases may be partially involved in carnosol’s effect. N = 12, ANOVA, *P < 0.0001, compared with vehicle control; **P < 0.0001 compared with carnosol.
SODD is involved in carnosol-induced decrease in cell proliferation in FLO-1 Cells.
We have previously showed that acid-induced increase in SODD expression depends on the activation of NOX5-S and NF-κB1 p50 in FLO EA cells18. Therefore, we examined whether carnosol-induced decrease in cell proliferation is through the reduction of SODD. Figure 5 showed that 24-h treatment with carnosol (10–4 M) significantly decreased SODD protein (Fig. 5A,B, t test, P < 0.05) and mRNA expression (Fig. 5C, t test, P = 0.001), supporting our hypothesis that SODD may be involved in carnosol induced reduction in cell proliferation.
SODD is involved in carnosol-induced decrease in cell proliferation in FLO-1 cells. (A) A typical image of Western blot analysis and (B) summarized data showed that carnosol (10–4 M, 24 h) significantly decreased SODD protein, indicating that carnosol downregulates SODD protein in FLO-1 cells. N = 4, * t-test, P < 0.05. Original blots are presented in Supplementary Figure 2. (C) 24-h treatment with carnosol (10–4 M) significantly decreased SODD mRNA expression in FLO-1 cells. #P = 0.001, t test, N = 8.
To further confirm this result, we used SODD siRNA to knock down SODD. We have shown that SODD siRNA successfully downregulated SODD protein expression18. Figure 6A showed that knockdown of SODD at basal condition significantly decreased cell proliferation (P < 0.02), indicating that SODD contributes to the cell proliferation at the basal condition in FLO-1 cells. In cells transfected with control siRNA, carnosol caused 68 ± 1% inhibition of cell proliferation (Fig. 6B). This inhibition was significantly reduced by knockdown of SODD (28.5 ± 4.8%, P < 0.02). The data suggest that downregulation of SODD may mediate carnosol-induced reduction in cell proliferation.
Role of SODD in carnosol-induced decrease in cell proliferation. (A) Knockdown of SODD at basal condition significantly decreased cell proliferation, indicating that SODD contributes to the cell proliferation at the basal condition in FLO-1 cells. Carnosol significantly decreased cell proliferation in cells transfected with control siRNA, a decrease which was partially reversed by knockdown of SODD. N = 3, ANOVA, ***P < 0.001, compared with control transfected with control siRNA, **P < 0.02, compared with control transfected with control siRNA; *P < 0.05, compared with carnosol transfected with control siRNA. B. In cells transfected with control siRNA, carnosol caused 68 ± 1% inhibition of cell proliferation. This inhibition was significantly reduced by knockdown of SODD (28.5 ± 4.8%). The data suggest that downregulation of SODD may mediate carnosol-induced reduction in cell proliferation. N = 3, t test, #P < 0.02.
Discussion
We found that carnosol inhibited cell proliferation and increased cell apoptosis in esophageal adenocarcinoma cells since carnosol dose-dependently decreased cell proliferation and significantly increased caspase-3 protein, suggesting that carnosol has anticancer activity in esophageal adenocarcinoma. The carnosol’s effect may be mediated by reactive oxygen species as supported by our results that N-acetyl cysteine (NAC) blocked carnosol-induced decrease in cell proliferation and carnosol significantly increased H2O2 production. This result is consistent with the results in breast cancer cells16 and human colon cancer HCT11611. In breast cancer cells16, carnosol activates ROS-dependent ER-stress responses, whereas in human colon cancer cells carnosol-induced apoptosis is mediated by inactivation of STAT3 through the production of ROS. Reactive oxygen species have dual effects. Low doses of ROS activate cell survival signaling pathway, whereas high doses of ROS induce cell apoptosis19. Therefore, carnosol might induce ROS production high enough to cause cell apoptosis.
ROS may be generated in mitochondria through the respiratory chains or by enzymes such as NADPH oxidases. Apocynin is a naturally occurring methoxy-substituted catechol and is an inhibitor of NADPH-oxidase20,21,22. It reduces the production of superoxide from activated neutrophils and macrophages, but the exact mechanism of its inhibition is not fully understood23. It is thought that apocynin inhibits reactive oxygen species (ROS) by inhibition of the assembly of NADPH-oxidase23. We found that apocynin partially reversed carnosol-induced decrease in cell proliferation, suggesting that NADPH oxidases might be partially involved in carnosol’s effect.
SODD is an anti-apoptotic protein and belongs to the BAG family24. SODD has been reported to be increased in cancer tissues; e.g. pancreatic cancers25, an increase of SODD expression which may decrease cell death and increase cell proliferation in various cancer cell lines25,26. SODD is associated with the ATPase domain of Hsc70/Hsp7024, the cytoplasmic domain of the tumor necrosis factor receptor 1 (TNFR1) and death receptor-3. TNFR1 signaling complex is activated by the release of SODD from TNFR1, which permits the recruitment of TNFR-associated death domain and TNFR-associated factor 226. SODD inhibits cell apoptosis through its binding to TNFR1 and preventing the ligand-independent oligomerization and spontaneous activation of TNFR127. We found that carnosol significantly decreased SODD protein and mRNA expression, indicating that carnosol may decrease cell proliferation via the downregulation of SODD. This result was further confirmed by the knockdown study showing that knockdown of SODD significantly blocked carnosol-induced inhibition of cell proliferation.
Carnosol might also play its anticancer role via epigenetic changes. For example, carnosol has been shown to inhibit p300 histone acetyl transferase activity by blocking the acetyl-CoA binding to the histone acetyl transferase catalytic domain since carnosol occupies the region where the pantetheine arm of the acetyl-CoA is bound15. It is well known that p300 is a coactivator of multiple transcription factors related to many biological processes28. High levels of p300 are associated with breast cancer progression29 and poor prognosis30. The inhibition of p300 histone acetyl transferase will decrease p300 acetylation, thus increasing p300 degradation. Whether this epigenetic modification of p300 is involved in carnosol’s anticancer effect in esophageal adenocarcinoma needs to be further explored.
In conclusion, carnosol dose-dependently decreased cell proliferation and significantly increased caspase-3 protein. Carnosol’s effect may be through the overproduction of ROS and the downregulation of SODD. Carnosol may be useful for the treatment of esophageal adenocarcinoma.
Materials and methods
Cell culture and treatment
The human Barrett’s adenocarcinoma cell line FLO-131 was obtained from Dr. David Beer (University of Michigan). The FLO-1 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum and antibiotics.
Cell proliferation assay
Cell proliferation was determined by using WST-1 Cell Proliferation Assay. Cell Proliferation Reagent WST-1 was purchased from MilliporeSigma. 2 × 104 FLO-1 cells were seeded in a 96-well plate. When cells reached 50–60% confluence, different concentrations of carnosol or vehicle controls were added to each well. After cells were cultured for 24 h, 10 μl WST-1 reagent was added to each well and cells were cultured for additional 1 h. Then the absorbance of each well was determined by using a BioTek Synergy H4 hybrid reader at 440 nm. Medium without cells was used as a blank. Carnosol was dissolved in ethanol and the same concentrations of ethanol were used in the control groups. The experiments were repeated three times for the experiments of siRNA, N-acetyl cysteine and carnosol’s effects on cell proliferation and twelve times for apocynin experiments.
Amplex® red hydrogen peroxide fluorescent assay
Levels of H2O2 in culture medium were measured by using Amplex® Red H2O2 Assay Kit (Thermo Fisher Scientific Inc., Waltham, MA) as we previously described32. The assay was performed in triplicates.
Small interfering RNA(SiRNA) transfection
FLO-1 cells were transfected with control siRNA or SODD siRNA by using Lipofectamine 2000 (Invitrogen, Grand Island, New York, USA) according to the manufacturer’s instruction and as we previously described18. 24 h after transfection, FLO-1 cells were treated with vehicle or carnosol (10–4 M) for additional 24 h.
Reverse-transcription PCR and quantitative real-time PCR
Reverse-transcription PCR and quantitative real-time PCR were performed as we previously described18. The primers used were as follows: SODD forward, 5′-GGGGTACCCAATGGTGCGATCTCGGCTCACTG-3′; SODD reverse, 5′-GAAGATCTCTCGAGGGGATCCGCTGCCCTGAAGCGCT-3′; 18S forward, 5′-CGGACAGGATTGACAGATTGATAGC-3′; and 18S reverse, 5′-TGCCAGAGTCTCGTTCGTTATCG -3′. The experiments were repeated eight times.
Western blot analysis
FLO-1 cells were treated with carnosol (10−4 M) or vehicle for 24 h and then collected for Western blot analysis. Western blot analysis was performed as described previously33. Primary antibodies used were as follows: SODD antibody (1:1000, Santa Cruz biotechnologies), caspase-3 antibody (1:2000, Upstate Biotechnology, Waltham, MA) and GAPDH antibody (1:2000, Santa Cruz biotechnologies). The experiments were performed in quadruplets.
Materials
Carnosol and N-acetyl cysteine (NAC) were purchased from Cayman Chemical Co. (Ann Arbor, MI, USA). Apocynin was purchased from Millipore Sigma (Burlington, MA).
Statistical analysis
All statistical analyses were performed by using GraphPad Prism software version 6 (GraphPad Software, Inc., Boston, MA). Data were expressed as mean ± S.E. Statistical differences between two groups were determined by Student’s t test. Differences among multiple groups were tested using analysis of variance (ANOVA) and checked for significance using Fisher’s protected least significant difference test.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information file.
References
Blot, W. J., Devesa, S. S., Kneller, R. W. & Fraumeni, J. F. Jr. Rising incidence of adenocarcinoma of the esophagus and gastric cardia. JAMA 265, 1287–1289 (1991).
Pennathur, A., Gibson, M. K., Jobe, B. A. & Luketich, J. D. Oesophageal carcinoma. Lancet 381, 400–412 (2013).
Thrift, A. P. & Whiteman, D. C. The incidence of esophageal adenocarcinoma continues to rise: analysis of period and birth cohort effects on recent trends. Ann. Oncol. 23, 3155–3162 (2012).
Lagergren, J., Bergstrom, R., Lindgren, A. & Nyren, O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N. Engl. J. Med. 340, 825–831 (1999).
Wild, C. P. & Hardie, L. J. Reflux, Barrett’s oesophagus and adenocarcinoma: Burning questions. Nat. Rev. Cancer 3, 676–684 (2003).
Souza, R. F., Morales, C. P. & Spechler, S. J. Review article: a conceptual approach to understanding the molecular mechanisms of cancer development in Barrett’s oesophagus. Aliment. Pharmacol. Ther. 15, 1087–1100 (2001).
Falk, G. W. Barrett’s esophagus. Gastroenterology 122, 1569–1591 (2002).
Crane, S. J. et al. Survival trends in patients with gastric and esophageal adenocarcinomas: a population-based study. Mayo Clin. Proc. 83, 1087–1094 (2008).
Sihvo, E. I., Luostarinen, M. E. & Salo, J. A. Fate of patients with adenocarcinoma of the esophagus and the esophagogastric junction: a population-based analysis. Am. J. Gastroenterol. 99, 419–424. https://doi.org/10.1111/j.1572-0241.2004.04094.x (2004).
Hur, C. et al. Trends in esophageal adenocarcinoma incidence and mortality. Cancer 119, 1149–1158. https://doi.org/10.1002/cncr.27834 (2013).
Park, K. W. et al. Carnosol induces apoptosis through generation of ROS and inactivation of STAT3 signaling in human colon cancer HCT116 cells. Int. J. Oncol. 44, 1309–1315. https://doi.org/10.3892/ijo.2014.2281 (2014).
Alsamri, H. et al. Carnosol, a natural polyphenol, inhibits migration, metastasis, and tumor growth of breast cancer via a ROS-dependent proteasome degradation of STAT3. Front. Oncol. 9, 743. https://doi.org/10.3389/fonc.2019.00743 (2019).
Wang, L. et al. Carnosol suppresses patient-derived gastric tumor growth by targeting RSK2. Oncotarget 9, 34200–34212. https://doi.org/10.18632/oncotarget.24409 (2018).
Johnson, J. J. Carnosol: A promising anti-cancer and anti-inflammatory agent. Cancer Lett. 305, 1–7. https://doi.org/10.1016/j.canlet.2011.02.005 (2011).
Alsamri, H. et al. Carnosol is a novel inhibitor of p300 acetyltransferase in breast cancer. Front. Oncol. 11, 664403. https://doi.org/10.3389/fonc.2021.664403 (2021).
Alsamri, H. et al. Carnosol induces p38-mediated ER stress response and autophagy in human breast cancer cells. Front. Oncol. 12, 911615. https://doi.org/10.3389/fonc.2022.911615 (2022).
Hong, J. et al. Role of Rac1 in regulation of NOX5-S function in Barrett’s esophageal adenocarcinoma cells. Am. J. Physiol. Cell Physiol. 301, 413–420 (2011).
Li, D., Hong, J. & Cao, W. Silencer-of-death domain mediates acid-induced decrease in cell apoptosis in Barrett’s associated esophageal adenocarcinoma cells. J. Pharmacol. Exp. Ther. 360, 14–22. https://doi.org/10.1124/jpet.116.236620 (2017).
Redza-Dutordoir, M. & Averill-Bates, D. A. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys. Acta 2977–2992, 2016. https://doi.org/10.1016/j.bbamcr.2016.09.012 (1863).
Lafeber, F. P. et al. Apocynin, a plant-derived, cartilage-saving drug, might be useful in the treatment of rheumatoid arthritis. Rheumatology (Oxford) 38, 1088–1093. https://doi.org/10.1093/rheumatology/38.11.1088 (1999).
Zhang, Y. et al. Apocynin but not allopurinol prevents and reverses adrenocorticotropic hormone-induced hypertension in the rat. Am. J. Hypertens. 18, 910–916. https://doi.org/10.1016/j.amjhyper.2005.02.017 (2005).
Stolk, J., Hiltermann, T. J., Dijkman, J. H. & Verhoeven, A. J. Characteristics of the inhibition of NADPH oxidase activation in neutrophils by apocynin, a methoxy-substituted catechol. Am. J. Respir. Cell Mol. Biol. 11, 95–102 (1994).
Stefanska, J. & Pawliczak, R. Apocynin: Molecular aptitudes. Mediators Inflamm 2008, 106507. https://doi.org/10.1155/2008/106507 (2008).
Takayama, S., Xie, Z. & Reed, J. C. An evolutionarily conserved family of Hsp70/Hsc70 molecular chaperone regulators. J. Biol. Chem. 274, 781–786 (1999).
Ozawa, F., Friess, H., Zimmermann, A., Kleeff, J. & Buchler, M. W. Enhanced expression of Silencer of death domains (SODD/BAG-4) in pancreatic cancer. Biochem. Biophys. Res. Commun. 271, 409–413 (2000).
Jiang, Y., Woronicz, J. D., Liu, W. & Goeddel, D. V. Prevention of constitutive TNF receptor 1 signaling by silencer of death domains. Science 283, 543–546 (1999).
Doong, H., Vrailas, A. & Kohn, E. C. What’s in the “BAG”?—A functional domain analysis of the BAG-family proteins. Cancer Lett. 188, 25–32 (2002).
Chen, J. & Li, Q. Life and death of transcriptional co-activator p300. Epigenetics 6, 957–961. https://doi.org/10.4161/epi.6.8.16065 (2011).
Fermento, M. E. et al. Inhibition of p300 suppresses growth of breast cancer. Role of p300 subcellular localization. Exp. Mol. Pathol. 97, 411–424. https://doi.org/10.1016/j.yexmp.2014.09.019 (2014).
Xiao, X. S. et al. High expression of p300 in human breast cancer correlates with tumor recurrence and predicts adverse prognosis. Chin. J. Cancer Res. 23, 201–207. https://doi.org/10.1007/s11670-011-0201-5 (2011).
Hughes, S. J. et al. Fas/APO-1 (CD95) is not translocated to the cell membrane in esophageal adenocarcinoma. Cancer Res. 57, 5571–5578 (1997).
Si, J. et al. NADPH oxidase NOX5-S mediates acid-induced cyclooxygenase-2 expression via activation of NF-kappaB in Barrett’s esophageal adenocarcinoma cells. J. Biol. Chem. 282, 16244–16255. https://doi.org/10.1074/jbc.M700297200 (2007).
Cao, W. et al. MAPK mediates PKC-dependent contraction of cat esophageal and lower esophageal sphincter circular smooth muscle. Am. J. Physiol. Gastrointest. Liver Physiol. 285, G86-95 (2003).
Acknowledgements
The work was supported in part by Chongqing Scientific Research Institute Performance Incentive Guidance Special Project [cstc2020jxjl130017] awarded to Dr. Aihua Li.
Author information
Authors and Affiliations
Contributions
A.L. performed the experiments, analyzed data and wrote the manuscript draft. W.C. designed the experiments, performed some experiments, analyzed data and wrote the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, A., Cao, W. Downregulation of SODD mediates carnosol-induced reduction in cell proliferation in esophageal adenocarcinoma cells. Sci Rep 13, 10580 (2023). https://doi.org/10.1038/s41598-023-37796-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37796-5
- Springer Nature Limited