Abstract
To assess the clinical value of mechanical thrombectomy (MT) combined with intravenous thrombolysis (IVT) in acute ischemic stroke (AIS) by comparing it with the MT alone. In this study, we conducted a comprehensive meta-analysis of both observational and randomized controlled studies (RCTs) to investigate various outcomes. Our search for relevant studies was conducted between January 2011 and June 2022 in four major databases: PubMed, Embase, WOS, and Cochrane Library. We collected data on several outcomes, including functional independence (FI; defined as modified Rankin Scale score of 0 to 2), excellent outcomes (mRS 0–1), successful recanalization (SR), symptomatic intracerebral hemorrhage (sICH), any intracerebral hemorrhage (aICH), and mortality at three months or discharge. The primary efficacy outcome and safety outcome were FI and sICH, respectively, whereas excellent outcomes and SR were considered secondary efficacy outcomes. Additionally, mortality and aICH were analyzed as secondary safety outcomes. We employed the Mantel–Haenszel fixed-effects model for RCTs when I2 < 50%, otherwise the random-effects model was utilized. For observational studies and subgroup analyses, we used the random-effects model to minimize potential bias. A total of 55 eligible studies (nine RCTs and 46 observational studies) were included. For RCTs, the MT + IVT group was superior in FI (OR: 1.27, 95% CI: 1.11–1.46), excellent outcomes (OR: 1.21, 95% CI: 1.03–1.43), SR (OR: 1.23, 95% CI: 1.05–1.45), mortality (OR: 0.72, 95% CI: 0.54–0.97) in crude analyses. In adjusted analyses, the MT + IVT group reduced the risk of mortality (OR: 0.65, 95% CI: 0.49–0.88). However, the difference in FI between the MT + IVT group and the MT alone group was not significant (OR: 1.17, 95% CI: 0.99–1.38, Fig. 3a). For observational studies, the results of FI (OR: 1.34, 95% CI: 1.16–1.33), excellent outcomes (OR: 1.30, 95% CI: 1.09–1.54), SR (OR: 1.23, 95% CI: 1.05–1.44), mortality (OR: 0.70, 95% CI: 0.64–0.77) in the MT + IVT group were better. Additionally, the MT + IVT group increased the risk of hemorrhagic transformation (HT) including sICH (OR: 1.16, 95% CI: 1.11–1.21) and aICH (OR: 1.24, 95% CI: 1.05–1.46) in crude analyses. In adjusted analyses, significant better outcomes were seen in the MT + IVT group on FI (OR: 1.36, 95% CI: 1.21–1.52), excellent outcomes (OR: 1.49, 95% CI: 1.26–1.75), and mortality (OR: 0.73, 95% CI: 0.56–0.94). The MT + IVT therapy did improve the prognosis for AIS patients and did not increase the risk of HT compared with MT alone therapy.
Similar content being viewed by others
Introduction
Stroke is the second greatest cause of mortality and the leading causes of disability worldwide. According to the Global Burden of Disease Study 2019, the burden of stroke is steadily rising, especially in low- and middle-income nations1. Ischemic and hemorrhagic strokes are the two main subtypes, with ischemic strokes accounting for around 85% of instances2. Intravenous thrombolysis (IVT) and mechanical thrombectomy (MT) are routinely performed in acute ischemic stroke (AIS) patients with occlusion of anterior circulation. According to the latest guidelines, the treatment window for MT was expanded up to 16–24 h, and IVT with alteplase was approved for patients within 4.5 hours3.
The prognosis of AIS was greatly improved when comparing MT with routine medical care4. However, there has been controversy regarding the effectiveness of IVT before MT. Most studies indicated that bridging treatment can encourage successful recanalization (SR)5,6,7,8,9. IVT, however, raised potential complications, especially intracranial hemorrhage and distal embolization. IVT-induced thrombus fragmentation would make subsequent MT more difficult10,11. These conflicting results highlighted the challenges of clinical operation selection.
Currently, several systematic and meta-analysis have compared the MT alone and bridging treatment (MT + IVT)12,13,14. Katsanos et al. indicated that AIS patients with MT + IVT treatment, compared to MT alone treatment, improved functional independence (FI), SR, and three-month mortality results12. In direct contrast, one study showed no statistically significant difference between the two treatment13. We also found either an assessment limited to observational studies or just randomized controlled trials (RCTs)12,13,14. Given the increasing number of clinical trials in this field, a comprehensive systematic review and meta-analysis should be conducted once more. The evaluations of therapeutic interventions would fall into two categories, observational studies and RCTs.
Methods
Literature search strategy
This study was carried out in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis statement (PRISMA)15. This research has been registered via PROSPERO (CRD42022345385). Two investigators searched from four databases (PubMed, Embase, WOS, and Cochrane Library) published From January 2011 to June 2022. Our search strategy combined Medical Subject Headings (MeSH) and free words.
Selection criteria
The selection criteria were based on the PICOS (population, intervention, comparison, outcomes, and study design) approach. The following criteria served as the basis for our study screening. Inclusion criteria: (1) The studies were observational studies and RCTs; (2) Data from adults (age ≥ 18); (3) Studies provided the quantitative estimates and their 95% confidence interval (95% CI). Exclusion criteria: (1) Studies were literature reviews, protocols, case reports, comments, editorial articles, cell experiments, or animal experiments; (2) Patients of AIS with non-anterior circulation in large vessel occlusion (LVO).
Participants and interventions
We included AIS patients with LVO in the anterior circulation. Each participant received the MT alone or IVT + MT therapy. Most of included studies primarily used the medication alteplase. It should be highlighted that we did not exclude some other IVT medications from our analysis even though they were not recommended by the guidelines.
Outcomes
In this study, FI for three months or hospital discharge, defined as a modified Rankin Scale (mRS) score (range,0 to 2), was selected as the primary efficacy outcome. The primary safety indicator was symptomatic intracerebral hemorrhage (sICH) at 24 or 36 h according to Heidelberg Bleeding Classification (HBC) 16, or European Cooperative Acute Stroke Study 3 classification (ECASS III)17, or ECASS II, or Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST) criteria18.
Thrombolysis in Cerebral Infarction (TICI score of 2B, 2C, or 3), modified TICI (mTICI) score (2B or 3), or eTICI score (2B, 2C, or 3)19 was defined as SR with final cerebral angiography, and mRS score (range, 0 to 1) were adopted as secondary efficacy outcomes. Mortality at three months or discharge and any intracerebral hemorrhage (aICH) were analyzed as secondary safety outcomes.
Quality assessment
Given that we had both RCTs, and observational studies included, we employed the Cochrane Risk of Bias tool (RoB) to assess RCTs, which included blinding, baseline comparison, allocation concealment, and randomization analysis. The modified Newcastle–Ottawa Scales (NOS) were used to assess the authenticity and quality of observational studies20. The NOS consisted of three sections: patient selection, study group comparability, and outcome assessment. The methodological quality of studies was assessed using a star system. The NOS can award up to nine points, with NOS ≥ 7 indicating high-quality study. Beyond this, the study was considered “low quality”.
Sensitivity and publication bias analyses
We performed sensitivity analyses to test the stability of our results by excluding each study one by one. Moreover, contour-enhanced funnel plots, Peter's test and Egger's test were conducted only when at least 10 studies were available to detect publication bias.
Data extraction
Two investigators reviewed each title, abstract, and full-text articles individually to select eligible studies. Any controversies were addressed in discussions with the third author. A Microsoft Excel file had the extracted data that was present. Study title, authors, publication date, study setting, study design, study period, participants, FI, SR, sICH, and mortality definitions, other important outcomes, and adjustment methods were among the extracted study characteristics. Crude data and effects estimate with their 95% CI of crude and adjusted were also included. For more details or unpublished data from conference abstracts, the corresponding authors would be contacted.
Data synthesis and statistical analysis
Considering the heterogenicity of the methodology, data source, and so on existed in the included studies. We evaluated the inter-study heterogeneity using I2 tests and the P-value. I2 values < 25%, 25–50%, 50–75%, 75–100% indicated no, moderate, large, and high levels of heterogeneity, respectively. P-value < 0.1 was considerately statistically significant. For RCTs, the Mantel–Haenszel fixed-effects model was used if I2 < 50%. Otherwise, the random-effects model was applied. For observational studies and subgroups analysis, wo chose the random-effects model to control the potential bias. After thoroughly reviewing each included study, we analyzed crude data and adjusted data separately to increase the credibility. These methods were applicable to both crude data analysis and adjusted analysis. For studies that used covariates, we included data that was adjusted for covariates by the original authors in the adjusted analysis.
Also, we performed subgroup analysis by study design (prospective study and retrospective study), and study area (Asia, European, and America). All the analyses were conducted in the RevMan software version 5.3 and computer program R software version 4.1.1. Unless otherwise noted, all P-values were two-tailed and less than 0.05 was considered statistically significant.
Ethical approval
This article belonged to the category of systematic review and meta-analysis, and we have confirmed that no ethical approval is required.
Results
Literature retrieval and study characteristics
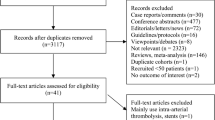
The study process as shown in Fig. 1. There were 4,930 items in total (1,830 from PubMed, 1,428 from WOS, 501 from Embase, and 1,171 from Cochrane Library). 2,863 items were included in the abstract screening after eliminating duplicates. Then 2,774 unrelated studies were excluded. Among the 2774 studies, 1184 were basic experimental studies involving animals and cells, 1023 were reviews, and 567 were studies that did not match the research topic. A total of 88 full-text articles were assessed for eligibility. We excluded 33 studies, 22 of which used therapies other than MT or IVT, seven studies were reviews, and four pieces involved RCTs protocol. Finally, 55 studies6,7,8,9,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70 met our protocol and were qualitatively synthesized and meta-analyzed.
Table 1 displays the characteristics of eligible studies, including the authors and years of publication, study design type, study period, study participants, age, gender, NIHSS score, location of occlusion, FI definition, SR definition, sICH definition, mortality definition, other outcomes, adjustment method, and adjustment of confounding factors. The study evaluated data from 17 nations, including 10 from Europe, four from Asia, two from The North American, and the one from Australia. Nine RCTs and 46 observational studies—29 retrospective (RS), 16 prospective (PS), and one cross-sectional (CS) were included in the analysis. almost all studies used an mRS score ≤ 2 to define FI. Methods to define SR included TICI 2b/3, mTICI 2b/3, and eTICI 2b/3. Additionally, several methods were adopted to assess sICH (ECASS II/III, HBC, and SITS-MOST). A portion of included studies adopted multivariate analysis, multivariate binary logistic regression, and propensity score method (PSM) to adjust the data.
Quality assessment for included studies
According to RoB, most trials were of high quality and possessed a low overall risk of bias. Supplemental Fig. 11 showed the specific details. Due to randomization and blinding items, a trial had a high risk of bias27. Additionally, Supplemental Table 4 showed how detailed information from OS were evaluated using the NOS scale. Except for one study62, which scored only 6 because controls for comparability between the two groups were omitted from the study. All other studies were rated as “high quality”.
Crude analysis
Primary outcomes
The results would be reported separately by RCTs and observational studies. Regarding efficacy outcomes, data from the nine RCTs indicated that MT + IVT group had superior FI than the MT alone group (OR: 1.27, 95% CI: 1.11–1.46, Fig. 2a), with large heterogeneity (I2 = 53%, P = 0.03). About safety outcomes, the results of sICH showed no significant difference between the two groups (OR: 1.13, 95% CI: 0.86–1.49, Fig. 2b), indicating no heterogeneity (I2 = 0, P = 0.82). Overall, 40 observational studies reported the results for FI, suggesting better results were seen in the MT + IVT group (OR: 1.34, 95% CI: 1.16–1.33, Fig. 2c), with large heterogeneity (I2 = 70%, P < 0.01). Data on sICH was extracted from 36 observational studies and found a 16% higher risk of HT (OR: 1.16, 95% CI: 1.11–1.21, Fig. 2d) in the MT + IVT group, with no heterogeneity (I2 = 0, P = 0.80).
Secondary outcomes
On the secondary efficacy outcomes, in nine RCTs, the MT + IVT group outperformed the MT alone group for excellent outcomes (mRS score: 0–1) (OR: 1.21, 95% CI: 1.03–1.43, Fig. 3a) with moderate heterogeneity (I2 = 43%, P = 0.09). Additionally, the MT + IVT group saw 23% more SR than the MT alone group (OR: 1.23, 95% CI: 1.05–1.45, Fig. 3b) in eight RCTs, no heterogeneity accompanied (I2 = 0, P = 0.96). Regarding safety outcomes of aICH from seven RCTs, the MT + IVT group had a 25% higher risk of HT than the MT alone group (OR: 1.25, 95% CI: 1.00–57, Fig. 3c), with low heterogeneity (I2 = 22%, P = 0.26). Mortality at 3-months or hospital discharge from eight RCTs in the MT + IVT group showed a lower mortality compared to the MT alone group (OR: 0.72, 95% CI: 0.54–0.97, Fig. 3d), with large heterogeneity (I2 = 54%, P = 0.03).
For efficacy outcomes, a total of 16 OS reported the excellent outcomes (mRS score: 0–1). Better results were seen in the MT + IVT group (OR: 1.30, 95% CI: 1.09–1.54, Fig. 4a) with large heterogeneity (I2 = 61%, P < 0.01). 38 OS showed SR outcomes, with the MT + IVT group increased the rate of SR (OR: 1.23, 95% CI: 1.05–1.44, Supplemental Fig. 4b), with large heterogeneity (I2 = 60%, P < 0.01). For safety outcomes, the MT + IVT group had higher aICH by 19% than the MT alone group (OR: 1.24, 95% CI: 1.05–1.46, Supplemental Fig. 4c) in 23 observational studies with moderate heterogeneity (I2 = 44%, P = 0.01). Additionally, in 34 investigations, mortality was 30% lower in the MT + IVT group compared to the MT alone group (OR: 0.70, 95% CI: 0.64–0.77, Supplemental Fig. 4d), with moderate heterogeneity (I2 = 42%, P = 0.01).
Subgroup analysis
Given the large heterogeneity of some outcomes, subgroup analysis by study design (RS vs PS) and area (Asia vs Europe vs America) was conducted. Regarding subgroup outcomes by study region in the RCTs, there was significant difference between Europe and Asia group in terms of FI (P = 0.05), Specifically, the MT + IVT group had better outcomes in Europe (OR: 1.46, 95% CI: 1.07–1.98), whereas there were no significant differences in Asia subgroup between the MT + IVT and the MT alone therapy (OR: 0.95, 95% CI: 0.75–1.21). Moreover, stratifying studies according to mortality showed significant differences (P < 0.01,). In Europe, the MT + IVT group reduced mortality risk by 45% (OR: 0.55, 95% CI: 0.45–0.68), while in Asia there was no significant difference (OR: 1.07, 95% CI: 0.78–1.48). There were no significant differences regarding SR (P = 0.73), excellent outcomes (P = 0.14), sICH (P = 0.25), and aICH (P = 0.10). The above details were depicted in Supplemental Table 1. On the basis of the results of study area subgroup in OS, no statistically significant variations regarding FI (P = 0.28), excellent outcomes (P = 0.31), SR (P = 0.93), sICH (P = 0.63), aICH (P = 0.19), and mortality (P = 0.38), of which were detailed in Supplemental Table 2.
The results of the subgroup analysis for observational studies were described in more detail below. As shown in Supplemental Table 3, there was no difference in the outcomes of FI (P = 0.13), excellent outcomes (P = 0.14), SR (P = 0.37), sICH (P = 0.20), aICH (P = 0.70), and mortality (P = 0.92).
Adjusted analysis
Primary outcomes
Results by assessing the adjusted ORs among RCTs between the MT + IVT group and the MT alone group were non-significant for both FI (OR: 1.17, 95% CI: 0.99–1.38, Fig. 3a) and sICH (OR: 1.07, 95% CI: 0.79–1.46, Fig. 3b), suggested no heterogeneity (I2 = 0, P = 0.54), and (I2 = 0, P = 0.40), respectively. However, significant better outcomes were seen in the MT + IVT group on FI in observational studies (OR: 1.36, 95% CI: 1.21–1.52, Fig. 3c), with moderate heterogeneity (I2 = 48%, P = 0.02). We did not see the significant differences on sICH (OR: 0.92, 95% CI: 0.76–1.12, Fig. 3d) between groups with low heterogeneity (I2 = 13%, P = 0.32).
Secondary outcomes
Results from RCTs indicated that the MT + IVT group significantly decreased the risk of mortality by 35% (OR: 0.65, 95% CI: 0.49–0.88, Supplemental Fig. 3d), with large heterogeneity (I2 = 52%, P = 0.07). All other results were non-significant differences between the two groups regarding excellent outcomes (OR: 1.11, 95% CI: 0.90–1.38, Supplemental Fig. 3a), SR (OR: 0.92, 95% CI: 0.75–1.13, Fig. 3b), and aICH (OR: 0.93, 95% CI: 0.75–1.15, Fig. 3c). The heterogeneities of above analyses were none (I2 = 0, P = 0.89), low (I2 = 24%, P = 0.24), and moderate (I2 = 63%, P = 0.04).
About observational studies, better results were seen in the MT + IVT group about the outcomes of excellent outcomes (OR: 1.49, 95% CI: 1.26–1.75, Supplemental Fig. 4a) with low heterogeneity (I2 = 4%, P = 0.40). We also observed the MT + IVT group reduced the risks of mortality by 27% (OR: 0.73, 95% CI: 0.56–0.94, Supplemental Fig. 4d) with large heterogeneity (I2 = 67%, P = 0.40) between two groups. And no significant differences were seen in the outcomes of SR (OR: 1.21, 95% CI: 0.85–1.74, Supplemental Fig. 4b) with large heterogeneity (I2 = 74%, P < 0.01), and aICH (OR: 1.06, 95%CI: 0.83–1.35, Supplemental Fig. 4c) by large heterogeneity (I2 = 28%, P = 0.22).
Subgroup analysis
Due to the limited number of included RCTs, advanced subgroup analysis was performed solely in observational studies. Among the subgroup of study area, there were no distinguishable differences in the outcomes of FI (P = 0.25), excellent outcomes (P = 0.20), sICH (P = 0.31), and mortality (P = 0.53), except for SR (P = 0.04). Specifically, there was non-significance in Asia between two groups (OR: 0.59, 95% CI: 0.29–1.21). However, in contrast to the MT alone therapy, the MT + IVT therapy raised the rate of SR by 51% in Europe (OR: 1.51, 95% CI: 1.23–1.86). All details were depicted in Supplemental Table 1.
No discernible differences were observable in outcomes of FI (P = 0.93), excellent outcomes (P = 0.22), SR (P = 0.57), sICH (P = 0.82) and aICH (P = 0.96) within the subgroup of study design between the two groups, except for mortality (P = 0.03). In prospective studies, MT + IVT therapy reduced the risk of mortality by 47% (OR: 0.53, 95% CI: 0.43–0.78). Retrospective analyses, however, did not reveal significant differences in the findings (OR: 0.95, 95% CI: 0.68–1.34). All details were displayed in Supplemental Table 3.
Sensitivity analysis
The sensitivity analysis of RCTs in crude data showed the effects of sICH (Supplemental Fig. 5b), SR (Supplemental Fig. 5d), and mortality (Supplemental Fig. 5f) were not substantially modified by exclusion of a certain study. The effect size of FI varied (OR: 1.15, 95% CI: 0.97–1.35, Supplemental Fig. 5a) when one study was excluded22. When the trial was eliminated9, the total effect sizes showed no discernible improvement (OR: 1.18, 95% CI: 0.99–1.40) in the excellent outcome of MT + IVT therapy. When this study was excluded22, a similar outcome (OR: 1.07, 95% CI: 0.89–1.28) was observed. And the MT + IVT group did not increase the risk of aICH (Supplemental Fig. 5d) while removing the study26 and the trial25, the effect sizes were (OR: 1.16, 95% CI: 0.95–1.41) and (OR: 1.18, 95% CI: 0.98–1.44,), respectively. Similar outcomes were seen in the outcome of excellent outcomes (Supplemental Fig. 5c). As followed by the sensitivity analysis of RCTs in adjusted data, the direction of effect size did not change in the outcomes of our interest (Supplemental Fig. 6b–f) except for the FI. The MT + IVT therapy significantly increased FI (OR: 1.23, 95% CI: 1.03–1.48, Supplemental Fig. 6a) after eliminating the study23.
As with the above analyses with observational studies, no significant differences were found in the outcomes of observational studies about crude data (Supplemental Fig. 7a, c–f), with the exception of the sICH (Supplemental Fig. 7b). When excluding the study71, the effect of direction changed (OR: 1.11, 95% CI: 0.96–1.29). Referring to observational studies of adjusted data, there were no discernible variations in the outcomes (Supplemental Fig. 8a–f).
Publication bias
For observational studies of crude data, the inspection of contour-enhanced funnel plots showed evidence of asymmetry of outcomes of FI (Fig. 4a), aICH (Supplemental Fig. 9c), and mortality (Supplemental Fig. 9d). No asymmetry was seen in the outcomes of excellent outcomes (Supplemental Fig. 9a), SR (Supplemental Fig. 9b), and sICH (Fig. 4b). However, there was no evidence in the corresponding Peter's statistical tests for funnel plot asymmetry in terms of the outcomes of FI (P = 0.06), excellent outcomes (P = 0.56), SR (P = 0.83), sICH (P = 0.89), aICH (P = 0.14), and mortality (P = 0.21).
The inspection of contour-enhanced funnel plots for observational studies with adjusted data revealed indications of asymmetries in outcomes of FI and sICH (Fig. 5a–b). There was no asymmetry in the mortality results (Supplemental Fig. 10). Additionally, except for sICH (P = 0.01), there was no indication of funnel plot asymmetry in the appropriate Egger's statistical tests for the outcomes of FI (P = 0.46) or mortality (P = 0.67). We did not run the funnel plot, Peter's, or Egger's statistical tests due to the numerous limitations of including RCTs and other observational studies.
Discussion
In this systematic review and meta-analysis, a total of approximately 20,000 patients were included in the final analysis. Overall, MT + IVT treatment significantly improved FI, excellent outcomes, and mortality risk in the observational studies, both in raw and adjusted data. Furthermore, it is crucial to note that although in crude analysis we observed an increased risk of sICH and aICH with MT + IVT treatment, no significant difference was found in the adjusted analysis. In the RCTs, we found that MT + IVT treatment reduced the risk of mortality but did not increase the risk of sICH in either the crude or adjusted analyses. Similar effect size directions emerged in the raw and adjusted data in the FI, excellent outcome, and SR domains, implying that there was no significant difference between the two therapies. In addition, although MT + IVT treatment significantly increased the risk of aICH in the raw data, it was not present in the adjusted data. Clearly, the adjusted analysis was more plausible due to the controlled covariates. The use of IVT prior to MT was previously thought to enhance the likelihood of HT58,72. However, our results provided further evidence that MT + IVT treatment did not significantly increase the risk of HT. Particularly, adjusted data from observational studies and RCTs, were used to draw conclusions.
The quality of life of impaired patients after stroke was significantly reduced, which caused mental and physical trauma to them and their families as well as a huge economic burden to the public health system. As such, improving the FI of stroke patients was a major rehabilitation object. In this meta-analysis, we found that the MT + IVT group significantly improved the FI in observational studies. although the outcome of FI was at the margin of significance in RCTs. This may be caused by the small number of included RCTs.
Considering the current inconsistency of large RCTs across different study areas, including Asia23,25,26,28 and the Europe9,21,22,24,27, as well as a study pointing to regionally relevant differences in the safety of IVT treatment in patients with AIS73. We performed a subgroup analysis by study area in the meta-analysis. The results indicated that the differences between countries appeared in the outcomes of FI and mortality in RCTs. Additionally, similar results were seen in observational studies (adjusted data) about SR. Overall, European outcomes were better than Asian. Specifically, European studies using MT + IVT therapy showed better FI, higher rates of SR, and lower mortality rates. The findings may suggest that in addition to taking racial factors into account when using MT + IVT therapy, larger clinical research will also be necessary in the future.
Supplemental Table 5 provided a detailed comparison of the prior meta-analysis and the current study. We conducted the most thorough research in this paper, utilizing the largest number of pertinent studies and populations. In addition, crude and adjusted analyses were conducted to further enhance the validity of our findings. Of particular note, although we conducted subgroup analyses by study design and area only, these analyses were based on extracting available data directly from the included studies with the aim of minimizing randomization and sampling error. The primary efficacy results derived from the analysis of observational studies in our study were consistent with previous studies74. Regarding the outcomes of the FI and sICH between two regimens by evaluating the raw data, non-significances were both seen when comparing the findings of synthesizing RCTs with the meta-analysis carried out by Vidale and colleagues14. Notably, our analysis of the adjusted data revealed that the MT + IVT therapy considerably outperformed the MT alone therapy in terms of excellent outcomes, SR, and mortality.
Several strengths of this study should be noted, and the following were some of the benefits of this study. First, the breadth of the chosen research—observational studies and RCTs with sizable sample sizes—allowed us to perform joint and subgroup analyses and improve statistical analysis. Second, we conducted crude and adjusted data analyses, which increased the credibility of the findings by accounting for confounding factors. Third, except for the outcomes of FI, we also assessed the excellent outcomes (mRS score: 0–1).
However, some limitations must be remarked upon. First, we routinely followed current clinical guidelines so that we only included AIS patients with occlusion of anterior circulation. But there was a need to know whether the MT therapy would be effective for posterior circulation occlusion. However, few studies were seen in this field after searching for literature. Second, because most of the included studies did not provide adjusted data, we performed adjusted analysis by synthesizing only a portion of the included studies, suggesting that the adjusted data were insufficient. Moreover, the number of covariates varied across studies. Third, we only conducted subgroup analyses of study design and area in order to minimize the bias. This may make it challenging for us to investigate additional potential confounders.
Conclusion
In summary, our findings showed that the MT + IVT therapy did, in fact, raise the rate of SR and lower the risk of mortality. Furthermore, we demonstrated that the MT + IVT therapy did not increase the risk of HT compared with the MT alone therapy. Based on the findings of observational studies, we thought that the MT + IVT therapy was more beneficial in achieving the object of FI. Although the results of FI in RCTs showed the same trend, they formally failed to achieve statistical significance. This would obviously call for further RCTs and analysis, both of which are necessary for future work.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
GBD 2016 Stroke Collaborators. Global, regional, and national burden of stroke, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 18, 439–458 (2019).
GBD 2019 Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 20, 795–820 (2021).
Powers, W. J. et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: A guideline for healthcare professionals from the american heart association/American stroke association. Stroke 50, e344–e418 (2019).
Goyal, M. et al. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet 387, 1723–1731 (2016).
Goyal, N. et al. Comparative safety and efficacy of combined IVT and MT with direct MT in large vessel occlusion. Neurology 90, e1274–e1282 (2018).
Di Maria, F. et al. Intravenous thrombolysis prior to mechanical thrombectomy in acute ischemic stroke: silver bullet or useless bystander?. J. Stroke 20, 385–393 (2018).
Casetta, I. et al. Combined intravenous and endovascular treatment versus primary mechanical thrombectomy. The Italian registry of endovascular treatment in acute stroke. Int. J. Stroke 14, 898–907 (2019).
Ferrigno, M. et al. Intravenous recombinant tissue-type plasminogen activator: Influence on outcome in anterior circulation ischemic stroke treated by mechanical thrombectomy. Stroke 49, 1377–1385 (2018).
Gariel, F. et al. Mechanical thrombectomy outcomes with or without intravenous thrombolysis. Stroke 49, 2383–2390 (2018).
Shi, K. et al. tPA mobilizes immune cells that exacerbate hemorrhagic transformation in stroke. Circ. Res. 128, 62–75 (2021).
Nogueira, R. G. et al. Predictors and clinical relevance of hemorrhagic transformation after endovascular therapy for anterior circulation large vessel occlusion strokes: A multicenter retrospective analysis of 1122 patients. J. Neurointerv. Surg. 7, 16–21 (2015).
Katsanos, A. H. et al. Intravenous thrombolysis prior to mechanical thrombectomy in large vessel occlusions. Ann. Neurol. 86, 395–406 (2019).
Podlasek, A., Dhillon, P. S., Butt, W., Grunwald, I. Q. & England, T. J. Direct mechanical thrombectomy without intravenous thrombolysis versus bridging therapy for acute ischemic stroke: A meta-analysis of randomized controlled trials. Int. J. Stroke 16, 621–631 (2021).
Vidale, S., Romoli, M. & Clemente Agostoni, E. Mechanical thrombectomy with or without thrombolysis: A meta-analysis of RCTs. Acta Neurol. Scand. 143, 554–557 (2021).
Moher, D. et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement (Chinese edition). J. Integr. Med. 7, 889–896 (2009).
von Kummer, R. et al. The heidelberg bleeding classification: Classification of bleeding events after ischemic stroke and reperfusion therapy. Stroke 46, 2981–2986 (2015).
Hacke, W. et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N. Engl. J. Med. 359, 1317–1329 (2008).
Wahlgren, N. et al. Thrombolysis with alteplase for acute ischaemic stroke in the safe implementation of thrombolysis in stroke-monitoring study (SITS-MOST): An observational study. Lancet 369, 275–282 (2007).
Goyal, M. et al. 2C or not 2C: Defining an improved revascularization grading scale and the need for standardization of angiography outcomes in stroke trials. J. Neurointerv. Surg. 6, 83–86 (2014).
Wells, G. et al. The newcastle-ottawa scale (NOS) for assessing the quality of nonrandomized studies in meta- analysis. 12.
Coutinho, J. M. et al. Combined intravenous thrombolysis and thrombectomy vs thrombectomy alone for acute ischemic stroke: A pooled analysis of the SWIFT and STAR studies. JAMA Neurol. 74, 268–274 (2017).
Chalos, V. et al. Endovascular treatment with or without prior intravenous alteplase for acute ischemic stroke. J. Am. Heart Assoc. 8, e011592 (2019).
Yang, P. et al. Endovascular thrombectomy with or without intravenous alteplase in acute stroke. N. Engl. J. Med. 382, 1981–1993 (2020).
LeCouffe, N. E. et al. A randomized trial of intravenous alteplase before endovascular treatment for stroke. N. Engl. J. Med. 385, 1833–1844 (2021).
Zi, W. et al. Effect of endovascular treatment alone vs intravenous alteplase plus endovascular treatment on functional independence in patients with acute ischemic stroke: The DEVT randomized clinical trial. JAMA 325, 234–243 (2021).
Suzuki, K. et al. Effect of mechanical thrombectomy without vs with intravenous thrombolysis on functional outcome among patients with acute ischemic stroke: The SKIP randomized clinical trial. JAMA 325, 244–253 (2021).
Huu An, N. et al. Thrombectomy alone versus bridging therapy in acute ischemic stroke: Preliminary results of an experimental trial. Clin. Ter. 173, 107–114 (2022).
Sakai, N. et al. Safety, pharmacokinetics and pharmacodynamics of DS-1040, in combination with thrombectomy, in Japanese Patients with acute ischemic stroke. Clin. Drug Investig. 42, 137–149 (2022).
Dávalos, A. et al. Retrospective multicenter study of solitaire FR for revascularization in the treatment of acute ischemic stroke. Stroke 43, 2699–2705 (2012).
Pfefferkorn, T. et al. Preceding intravenous thrombolysis facilitates endovascular mechanical recanalization in large intracranial artery occlusion. Int. J. Stroke 7, 14–18 (2012).
Kass-Hout, T. et al. Is bridging with intravenous thrombolysis of any benefit in endovascular therapy for acute ischemic stroke?. World Neurosurg. 82, e453-458 (2014).
Leker, R. R. et al. Direct thrombectomy versus bridging for patients with emergent large-vessel occlusions. Interv. Neurol. 7, 403–412 (2018).
Maier, I. L. et al. Bridging-therapy with intravenous recombinant tissue plasminogen activator improves functional outcome in patients with endovascular treatment in acute stroke. J. Neurol. Sci. 372, 300–304 (2017).
Guedin, P. et al. Prior IV thrombolysis facilitates mechanical thrombectomy in acute ischemic stroke. J. Stroke Cerebrovasc. Dis. 24, 952–957 (2015).
Broeg-Morvay, A. et al. Direct mechanical intervention versus combined intravenous and mechanical intervention in large artery anterior circulation stroke: A matched-pairs analysis. Stroke 47, 1037–1044 (2016).
Behme, D. et al. Intravenous thrombolysis facilitates successful recanalization with stent-retriever mechanical thrombectomy in middle cerebral artery occlusions. J. Stroke Cerebrovasc. Dis. 25, 954–959 (2016).
Abilleira, S. et al. Outcomes after direct thrombectomy or combined intravenous and endovascular treatment are not different. Stroke 48, 375–378 (2017).
Bellwald, S. et al. Direct mechanical intervention versus bridging therapy in stroke patients eligible for intravenous thrombolysis: A pooled analysis of 2 registries. Stroke 48, 3282–3288 (2017).
Weber, R. et al. Comparison of outcome and interventional complication rate in patients with acute stroke treated with mechanical thrombectomy with and without bridging thrombolysis. J. Neurointerv. Surg. 9, 229–233 (2017).
de Alonso Leciñana, M. et al. Mechanical thrombectomy in patients with medical contraindications for intravenous thrombolysis: A prospective observational study. J. Neurointerv. Surg. 9, 1041–1046 (2017).
Froehler, M. T. et al. Interhospital transfer before thrombectomy is associated with delayed treatment and worse outcome in the STRATIS registry (systematic evaluation of patients treated with neurothrombectomy devices for acute ischemic stroke). Circulation 136, 2311–2321 (2017).
Rai, A. T. et al. Intravenous thrombolysis before endovascular therapy for large vessel strokes can lead to significantly higher hospital costs without improving outcomes. J. Neurointerv. Surg. 10, 17–21 (2018).
Wang, H. et al. Direct endovascular treatment: An alternative for bridging therapy in anterior circulation large-vessel occlusion stroke. Eur. J. Neurol. 24, 935–943 (2017).
Wee, C.-K. et al. Outcomes of endovascular thrombectomy with and without thrombolysis for acute large artery ischaemic stroke at a tertiary stroke centre. Cerebrovasc. Dis. Extra. 7, 95–102 (2017).
Merlino, G. et al. Short and long-term outcomes after combined intravenous thrombolysis and mechanical thrombectomy versus direct mechanical thrombectomy: A prospective single-center study. J. Thromb. Thrombolysis 44, 203–209 (2017).
Park, H.-K. et al. Preceding intravenous thrombolysis in patients receiving endovascular therapy. Cerebrovasc. Dis. 44, 51–58 (2017).
Choi, J. H. et al. Comparison of outcomes after mechanical thrombectomy alone or combined with intravenous thrombolysis and mechanical thrombectomy for patients with acute ischemic stroke due to large vessel occlusion. World Neurosurg. 114, e165–e172 (2018).
Al-Khaled, M. et al. Comparing outcome and recanalization results in patients with anterior circulation stroke following endovascular treatment with and without a treatment with rt-PA: A single-center study. Brain Behav. 8, e00974 (2018).
Heinrichs, A. et al. Relevance of standard intravenous thrombolysis in endovascular stroke therapy of a tertiary stroke center. Acta Neurol. Belg. 118, 105–111 (2018).
Sallustio, F. et al. Effect of mechanical thrombectomy alone or in combination with intravenous thrombolysis for acute ischemic stroke. J. Neurol. 265, 2875–2880 (2018).
Bourcier, R. et al. Is bridging therapy still required in stroke due to carotid artery terminus occlusions?. J. Neurointerv. Surg. 10, 625–628 (2018).
Goyal, N. et al. Impact of pretreatment with intravenous thrombolysis on reperfusion status in acute strokes treated with mechanical thrombectomy. J. Neurointerv. Surg. 11, 1073–1079 (2019).
Leker, R. R., Pikis, S., Gomori, J. M. & Cohen, J. E. Is bridging necessary? A pilot study of bridging versus primary stentriever-based endovascular reperfusion in large anterior circulation strokes. J. Stroke Cerebrovasc. Dis. 24, 1163–1167 (2015).
Guimarães Rocha, M. et al. Primary thrombectomy versus combined mechanical thrombectomy and intravenous thrombolysis in large vessel occlusion acute ischemic stroke. J. Stroke Cerebrovasc. Dis. 28, 627–631 (2019).
Balodis, A. et al. Endovascular thrombectomy in anterior circulation stroke and clinical value of bridging with intravenous thrombolysis. Acta Radiol. 60, 308–314 (2019).
Gong, L. et al. Bridging therapy versus direct mechanical thrombectomy in patients with acute ischemic stroke due to middle cerebral artery occlusion: A clinical- histological analysis of retrieved thrombi. Cell Transplant. 28, 684–690 (2019).
Maingard, J. et al. Outcomes of endovascular thrombectomy with and without bridging thrombolysis for acute large vessel occlusion ischaemic stroke. Intern. Med. J. 49, 345–351 (2019).
Hassan, A. E. et al. Pre-thrombectomy intravenous thrombolytics are associated with increased hospital bills without improved outcomes compared with mechanical thrombectomy alone. J. Neurointerv. Surg. 11, 1187–1190 (2019).
Reiff, T., Barthel, O., Ringleb, P. A., Pfaff, J. & Mundiyanapurath, S. Safety of mechanical thrombectomy with combined intravenous thrombolysis in stroke treatment 4.5 to 9 hours from symptom onset. J. Stroke Cerebrovasc. Dis. 29, 105204 (2020).
Yi, H. J., Sung, J. H. & Lee, D. H. Bridging Intravenous thrombolysis before mechanical thrombectomy for large artery occlusion may be detrimental with thrombus fragmentation. Curr. Neurovasc. Res. 17, 18–26 (2020).
Hinsenveld, W. H. et al. Intravenous thrombolysis is not associated with increased time to endovascular treatment. Cerebrovasc. Dis. 49, 321–327 (2020).
Imbarrato, G., Bentley, J. & Gordhan, A. Clinical outcomes of endovascular thrombectomy in tissue plasminogen activator versus non-tissue plasminogen activator patients at primary stroke care centers. J. Neurosci. Rural Pract. 9, 240–244 (2018).
Jian, Y. et al. Direct versus bridging mechanical thrombectomy in elderly patients with acute large vessel occlusion: A multicenter cohort study. Clin. Interv. Aging 16, 1265–1274 (2021).
Tong, X. et al. Thrombectomy versus combined thrombolysis and thrombectomy in patients with acute stroke: A matched-control study. Stroke 52, 1589–1600 (2021).
Kandregula, S. et al. Direct thrombectomy versus bridging thrombolysis with mechanical thrombectomy in middle cerebral artery stroke: A real-world analysis through national inpatient sample data. Neurosurg. Focus 51, E4 (2021).
Zha, M. et al. Bridge mechanical thrombectomy may be a better choice for acute large vessel occlusions. J. Thromb. Thrombolysis 52, 291–300 (2021).
Machado, M. et al. Functional outcome after mechanical thrombectomy with or without previous thrombolysis. J. Stroke Cerebrovasc. Dis. 30, 105495 (2021).
Platko, S. et al. Intravenous thrombolysis prior to mechanical thrombectomy does not affect clinical or procedural outcomes in patients with large vessel occlusion acute ischemic stroke. J. Clin. Neurosci. 100, 120–123 (2022).
Dicpinigaitis, A. J. et al. Endovascular thrombectomy with and without preceding intravenous thrombolysis for treatment of large vessel anterior circulation stroke: A cross-sectional analysis of 50,000 patients. J. Neurol. Sci. 434, 120168 (2022).
Minnerup, J. et al. Outcome after thrombectomy and intravenous thrombolysis in patients with acute ischemic stroke: A prospective observational study. Stroke 47, 1584–1592 (2016).
Banks, J. L. & Marotta, C. A. Outcomes validity and reliability of the modified rankin scale: Implications for stroke clinical trials: A literature review and synthesis. Stroke 38, 1091–1096 (2007).
Tian, B. et al. Clinical and imaging indicators of hemorrhagic transformation in acute ischemic stroke after endovascular thrombectomy. Stroke 53, 1674–1681 (2022).
Mehta, R. H. et al. Race/Ethnic differences in the risk of hemorrhagic complications among patients with ischemic stroke receiving thrombolytic therapy. Stroke 45, 2263–2269 (2014).
Katsanos, A. H. et al. Intravenous thrombolysis prior to mechanical thrombectomy in large vessel occlusions. Ann. Neurol. 86, 395–406 (2019).
Acknowledgements
I would like to express my gratitude for the hard work and dedication of all the authors involved in this article.
Funding
This work was funded by the National Natural Science Foundation of China (Grant number: 82074303, 82174345).
Author information
Authors and Affiliations
Contributions
M.Z.: Conceptualization, Methodology, Software, Data curation, Original draft preparation; L.L.: Data curation, Writing- Reviewing and Editing; L.C., B.L., and C.F.: Supervision,Validation.
Corresponding authors
Ethics declarations
Competing interests
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zheng, M., Li, L., Chen, L. et al. Mechanical thrombectomy combined with intravenous thrombolysis for acute ischemic stroke: a systematic review and meta-analyses. Sci Rep 13, 8597 (2023). https://doi.org/10.1038/s41598-023-35532-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-35532-7
- Springer Nature Limited