Abstract
To describe clinical outcomes of patients aged 75 years and above after partial nephrectomy (PN), and to assess independent factors of postoperative complications. We retrospectively reviewed information from our multi-institutional database. Every patient over 75 years old who underwent a PN between 2003 and 2016 was included. Peri-operative and follow up data were collected. Multivariate logistic regression was performed to determine independent predictive factors of postoperative complications. We reviewed 191 procedures including 69 (40%) open-surgery, and 122 (60%) laparoscopic procedures, of which 105 were robot-assisted. Median follow-up was 25 months. The mean age was 78 [75–88]. The American Society of Anesthesiologist’s score was 1, 2, 3 and 4 in 10.5%, 60%, 29% and 0.5% of patients respectively. The mean tumor size was 4.6 cm. Indication of PN was elective in 122 (65%) patients and imperative in 52 patients (28%). The median length of surgery was 150(± 60) minutes, and the median estimated blood loss 200 ml. The mean glomerular filtration rate was 71.5 ml/minute preoperatively, and 62 ml/min three months after surgery. The severe complications (Clavien III-V) rate was 6.2%. On multivariate analysis, the robotic-assisted procedure was an independent protective factor of medical postoperative complications (Odds Ration (OR) = 0.31 [0.12–0.80], p = 0.01). It was adjusted for age and RENAL score, robotic-assisted surgery (OR = 0.22 [0.06–0.79], p = 0.02), and tumor size (OR = 1.13 [1.02–1.26], p = 0.01), but the patients age did not forecast surgical complications. Partial nephrectomy can be performed safely in elderly patients with an acceptable morbidity, and should be considered as a viable treatment option. Robotic assistance is an independent protective factor of postoperative complications.
Similar content being viewed by others
Introduction
Renal cell carcinoma (RCC) represents 3% of all cancer, with an increasing incidence in western countries1. The median age at diagnosis is 64 years old and over 20% of new cases are discovered after the age of 752. The estimated incidence increases particularly in the older population. Indeed, the age-standardized rates per 10,000 is 0.5 below the age of 40 years old and 35.0 over 75 years old3. Partial nephrectomy (PN) is the gold standard for cT1 renal masses management. In comparison with radical nephrectomy, it offers equivalent oncological outcomes and improvement of renal function. It reduces the risk of chronic kidney disease (CKD). Hence, it is recommended whenever technically feasible4. Active surveillance can be considered as an alternative for small renal masses (< 2 cm), particularly for frail patients5. While older patients are more likely to harbor more aggressive RCC with higher cancer-specific mortality5, PN is under-represented in this population, even among patients presenting alteration of the renal function. While PN appears under-utilized among older patients population, the general population follows the opposite trend with an increasing application6. This age-specific management can be explained by the competing risks of RCC and surgery, in a population with presumably more comorbidities. There is currently little data in the literature about the specific risks of PN in elderly patients, in terms of morbidity.
This study aims to depict PN outcomes, in terms of complications, and renal function in an over 75 yr(s) old population. It also seeks to identify independent factors of complications, in order to improve the selection of elderly patients eligible for partial surgery. The main outcome of interest was the safety of the procedure, analyzed through the surgical and medical complications occurring up to 3 months after the procedure.
Material and methods
Patients
This is a retrospective analysis of a multi-institutional, prospectively-maintained dataset of patients treated with PN for renal tumors between 2003 and 2016 at 15 French centers that are involved in the French network of research on kidney cancer UroCCR (NCT03293563). Patients > 75 yr(s) old undergoing PN for a localized renal tumor were included. The technical approach for the PN was at the surgeon’s discretion. The study was approved by the Institutional Review Board CPP Sud-Ouest et Outre mer III (DC 2012/108). All methods were performed in accordance to the relevant guidelines and regulations. All patients received oral and written information about the objectives and methodology of the UroCCR project, and informed consent was obtained.
Data measurements
Based on UroCCR database, we evaluated the patients peri-operative data and the complications up to 3 months after surgery. General demographics information were analyzed along with the main comorbidities.
Predictive operative risk was evaluated with the American Society of Anesthesiologists (ASA) and Eastern Cooperative Oncology Group (ECOG) scores.
The specific tumor characteristics analyzed were the TNM stage and the RENAL and PADUA nephrometry scores.
The technical characteristics of the surgery analyzed were surgical approach, indication for PN (elective or imperative), operative and warm ischemia times, estimated blood loss (EBL) and transfusion.
Regarding the safety, we analyszed the length of stay (LoS), decrease in renal function (estimated glomerular filtration rate (eGFR) calculated with the Modification of Diet in Renal Disease formula (MDRD)and based on the creatinine level measured in µmol/L), intra and post-operative complications reported with the modified Clavien-Dindo classification7.
The pathological findings with the RCC histological subtype, pTNM stage, Fuhrmann grade, surgical margins, peri-renal or sinusal fat invasion, renal vein thrombus, microvascular invasion, sarcomatoid component were also analyzed.
The trifecta achievement was assessed according to the Khalifeh criteria: combination of negative surgical margin, no perioperative complications, and ischemia time < 25 min8.
Statistical analysis
Odds ratio (OR) and 95% confidence intervals (CI) were obtained using logistic regression models.
Univariate models were fitted for all clinical and demographic variables. Multivariate logistic regression was performed to determine independent predictive factors of postoperative complications (medical and surgical). A backward stepwise manual method was used for the reduced final multivariate model to predict postoperative complications, adjusted for variables significant at the 0.20 level in univariate analysis and for age, regardless of the significance level in univariate analysis. All analyses were performed in SAS, 9.4.
The significance level alpha was 0.05.
Results
A total of 191 procedures were reviewed, including 69 (40%) open-surgery and 122 (60%) laparoscopic procedures, of which 105 were robot-assisted. The mean age was 78 [75–88], 60% of patients (n = 117) were men. Median follow-up was 25 months [10.0–31.2]. The ASA score was 1, 2, 3 and 4 in 10.5%, 60%, 29% and 0.5% of patients respectively. One hundred and forty-four patients (75.4%) were asymptomatic at diagnosis. The mean tumor size was 4.6 cm. The indication of PN was elective in 122 (65%) patients and imperative in 52 patients (28%). Median eGFR was 72.7 mL/min/m2 and 51 (26.7%) patients had a pre-operative eGFR < 60 mL/min/m2. The mean length of surgery was 161 (± 60) minutes. The median estimated blood loss was 200 ml and the postoperative transfusion rate was 14.7%. Trifecta was achieved in 45.0% of the procedures. Clinical and surgical data are summarized in Table 1, histological data in Table 2.
The intra-operative complication rates were 9%, of which most were hemorrhages requiring transfusions. The medical and surgical postoperative complication rates were respectively 19% and 10.5%. Details of the complications are reported in Table 3. According to the modified Clavien-Dindo classification, the severe complications (III-V) rate was 6.2%. On multivariate logistic regression, robotic-assisted procedure was an independent protective factor of both medical (adjusted for age, RENAL score, bilateral location and tumor size) and surgical (adjusted for age and creatinine level in µmol/L) postoperative complications (OR = 0.22 [0.06–0.79], p = 0.02 and OR = 0.31 [0.12–0.80], p = 0.01, respectively). The higher pre-operative creatinine level was associated with increased odds of surgical complication (OR = 1.01 [1.00–1.02], p = 0.04), while the bilateral tumor location (OR = 4.91 [1.04–23.15], p = 0.04) and tumor size (OR = 1.14 [1.02–1.27], p = 0.01) were associated with increased odds of medical complications (Table 4).
Among this > 75 yr(s) old population, age was not significantly associated with postoperative complications. The median length of stay at hospital was 6 days. Sixteen patients died, with only 3 deaths related to RCC. Four patients died in the early post-operative period, 2 presented a pulmonary embolism, 2 had a respiratory distress, or complicating decompensation of a chronic obstructive pulmonary disease for the former and asthma for the latter.
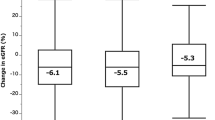
Regarding the renal function, the mean pre, per and post-operative (3 months) eGFR were 71.5 ml/min, 64.8 ml/min and 61.8 ml/min. The mean eGFR decrease was 7.1 ml/min (standard deviation 15.8) at 3 months.
Discussion
In this large retrospective series, we report the feasibility and safety of partial nephrectomy in elderly patients. Nephron-sparing surgery appears to be a valuable option, with a low morbidity rate and good quality outcomes, thus allowing both preservation of renal function and control of the cancer. The results of this study were obtained in spite of the overall challenging presentations of this cohort, with high rates of complex tumors and imperative indications. This procedure should be considered regardless of the patients age—if the individual RCC specific risk is high and competing comorbidities allow a curative strategy. The robot-assisted laparoscopic approach was associated with a low morbidity rate, with similar rates of surgical margins and warm ischemia time.
While partial nephrectomy is the standard of care, and recommended as the first treatment option for T1 tumors with a strong level of evidence4, it seems to remain underutilized in elderly patients9. Using the Surveillance, Epidemiology, and End Results database, Tan et al. demonstrated that the use of PN vs RN was significantly higher among younger patients. In comparison to the 65–69 yr(s) old age group, the PN odds ratio among the 70–74, 75–79, 80–84 and ≥ 85 yr(s) old patients were 0.82, 0.80, 0.55 and 0.30 respectively10. Leppert et al. reported the evolution of PN and RN relative use (2002–2014) from the Veterans Health administration database. While they found an overall shift from 17 to 38% in favor of PN during this period, older patients, and patients with worse baseline kidney function showed the least increase11. The large development of this procedure in clinical practices was more limited among older patients, who were likely to remain treated with RN6,9. The older patients were nevertheless more likely to present altered renal function at the time of diagnosis. In our study, 26.7% of the patients presented at least a moderate CKD before surgery, and PN was imperative in 27.8% of cases. Therefore, the rationale for PN seems more relevant among elderly patients, where preservation of kidney function is paramount to prevent end-stage renal disease and potential cardiovascular disorders4.
The paradoxical underutilization of PN might stem from the surgical risk of a potentially frail population, with more pre-existing comorbidities. Higher rates of peri-operative complications have been reported with PN, in comparison to RN12,13. However, in our study, the complication rate was acceptable with a 6.2% of Clavien ≥ III complication rate. This rate is comparable to previous series8,14. These results were found despite a high rate of complex tumors with a median tumor size of 4.7 cm, and 72.3% of patients harboring intermediate or highly complex tumors based of the RENAL nephrometry score. It is noteworthy to precise that in this > 75 yr(s) old population, age was not a predictive factor of surgical complications in our multivariate analyses. We assessed the trifecta achievement, according to Khalifeh definition, to evaluate the global surgical quality. This composite marker requires the association of negative surgical margins for cancer control, warm ischemia time under 25 min (as a surrogate of renal function preservation), and the absence of peri-operative complications8. We found that 45.0% of the procedures met these 3 criteria together. In their original article, Khalifeh el al compared trifecta achievement between patients receiving laparoscopic (231 patients), and robot-assisted (269 patients) PN. Patients median ages were respectively 57.7 and 58.8 yr(s) old. The Trifecta was achieved in 31.6% and 58.7% of patients respectively, with an overall 5.2% Clavien ≥ III-IV complication rate. Thus, the complication rate and overall surgical quality appear similar in our elderly patients cohort to this younger population-based study. The feasibility and safety of PN in older patients was also recently reported by Bindayi et al., who analyzed 653 over-75 yr old patients who underwent PN14. Using Hung’s trifecta definition15, they found 40.4% rate of achievement and 6.0% of Clavien ≥ III complications. Altogether, these results confirm that age should not be considered an absolute contraindication for PN. A global frailty and comorbidity assessment should be performed in older patient, and balanced with the potential risk of tumor progression on individual basis. Even after 75 yr(s) old, when a patient is fit for a surgical intervention, PN is probably the best option to control the tumor, in order to prevent CKD and quality-of-life deterioration.
The surgical approach seems to play a role in PN outcomes. In our multivariate analyses, we found that a robotic-assisted approach was the only predictive factor that prevented both medical and surgical complications. The worst renal function was associated with more surgical complications, while the tumor size and bilateral tumors were linked to more medical complications. The association between altered renal function and surgical complication could be explained with the increased risk of bleeding and developing hematoma with chronic kidney disease16. Larger and bilateral tumors impose longer procedure. Prolonged operative time is associated with higher risk of complications and a recent meta-analysis demonstrated a 14% increase in the likelihood of complications for every 30 min of additional operating time17.
This improvement with the robotic approach was already described in the overall population series. Peyronnet et al. retrospectively reviewed 1800 cases of PN that were performed in 6 academic centers, and compared robotic to the open approach. They found that the robotic approach was associated with less complications, less blood loss, and shorter hospital stay with similar oncologic outcomes18. Similarly, Larcher et al. reported a lower rate of overall (21% vs 36%; p < 0.0001) and major (3% versus 9%, p = 0.03) complications for those treated with robot-assisted versus open procedures. This was based on a retrospective analysis of 472 patients undergoing PN for cT1-2N0M0 renal masses. The functional and oncologic outcomes were similar in both arms19. Another series compared the functional and oncologic outcomes of open, laparoscopic and robot-assisted PN from 1308 patients. After an over 5-year median follow-up, the local recurrence, distant metastasis, and cancer-related death rates were similar, while CKD upstaging was lower in the robot-assisted PN arm20. To our knowledge, the present study is the first to demonstrate the robotic approach advantage among elderly patients in respect to post-operative complications.
The main limitations of our study are the retrospective and non-comparative design. As previously discussed, proposing a RN, when a PN is technically feasible, is hardly warrantable. It would be interesting to compare PN with active surveillance and thermal ablation in elderly patients, as to our knowledge this has never yet been performed. The aim of this study was to define the feasibility and safety of PN in an older patient population. Therefore, this endpoint did not require a comparison. Despite this, our complication rates were comparable with contemporary studies in elderly patients and in the overall PN population. Active surveillance is a valuable option for comorbid patients harboring small renal masses, and significant competing-cause of mortality21,22. It consists of tumor size monitoring, through regular abdominal imaging, that should trigger treatment in case of growth4. While large series have demonstrated usual slow tumor growth rates for patients under active surveillance and rare progressions to metastatic stages23,24,25, tumor size appears to be a reliable surrogate of tumor aggressivity24. Thus, AS is particularly relevant in patients with tumor sizes < 2-3 cm26. The same applies for ablative strategies, which can achieve acceptable oncologic outcomes for small renal masses, with good renal function preservation and low complication rates27. However, tumor size is the main risk factor of tumor recurrence with a reported threshold of 3-4 cm28,29,30,31. In our study, the median tumor size was 4.7 cm. Thus, most of the patients were probably not eligible for AS, nor ablative treatments. Despite this, our results should be confirmed by further investigations. The large number of patients included in our study, and the concordance with previous reports, confirm that age should not be a limitation when a treatment is indicated for patients harboring a RCC. Nephron-sparing surgery should be considered as the standard of care, when technically feasible, with the same quality outcomes expected as in the general population. Indications should be balanced in light of the competing comorbidities and the tumor characteristics, in order to expect benefits in terms of overall survival and quality of life.
Conclusion
Nephron-Sparing surgery is feasible and safe, even for older patients. When they are fit for surgery, age should not be a constraint in considering this therapeutic strategy. We have demonstrated a low surgical morbidity rate in this population, comparable to the contemporary series of younger patients. It remains a valuable option, even for large tumors that are not amenable for active surveillance or ablative techniques. Faster and less morbid procedures should not be reasons to prefer RN to PN. The Robot-assisted approach seems to offer a lower rate of complications.
Abbreviations
- ASA:
-
American society of anesthesiologists
- CI:
-
Confidence interval
- CKD:
-
Chronic kidney disease
- EBL:
-
Estimated blood loss
- eGFR:
-
Estimated glomerular filtration rate
- ECOG:
-
Eastern cooperative oncology group
- LoS:
-
Length of stay
- MDRD:
-
Modification of diet in renal disease
- OR:
-
Odds ratio
- PN:
-
Partial nephrectomy
- RCC:
-
Renal cell carcinoma
- RN:
-
Radical Nephrectomy
- yr(s) old:
-
Years old
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J.Clin. 68(6), 394–424 (2018).
Cancer of the Kidney and Renal Pelvis - Cancer Stat Facts [Internet]. SEER. [cited 2020 Nov 20]. Available from: https://seer.cancer.gov/statfacts/html/kidrp.html
Capitanio, U. et al. Epidemiology of renal cell carcinoma. Eur.Urol. 75(1), 74–84 (2019).
Ljungberg, B. et al. European association of urology guidelines on renal cell carcinoma: the 2019 update. Eur.Urol. 75(5), 799–810 (2019).
Scoll, B. J. et al. Age, tumor size and relative survival of patients with localized renal cell carcinoma: a surveillance, epidemiology and end results analysis. J. Urol. 181(2), 506–511 (2009).
Kim, S. P. et al. National treatment trends among older patients with T1-localized renal cell carcinoma. Urol. Oncol. 35(3), 113.e15-113.e21 (2017).
Dindo, D., Demartines, N. & Clavien, P.-A. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 240(2), 205–213 (2004).
Khalifeh, A. et al. Comparative outcomes and assessment of trifecta in 500 robotic and laparoscopic partial nephrectomy cases: a single surgeon experience. J. Urol. 189(4), 1236–1242 (2013).
Woldrich, J. M. et al. Trends in the surgical management of localized renal masses: thermal ablation, partial and radical nephrectomy in the USA, 1998–2008. BJU Int. 111(8), 1261–1268 (2013).
Tan, H.-J. et al. Health status and use of partial nephrectomy in older adults with early-stage kidney cancer. Urol. Oncol. 35(4), 153.e7-153.e14 (2017).
Leppert, J. T. et al. Contemporary use of partial nephrectomy: are older patients with impaired kidney function being left behind?. Urology 100, 65–71 (2017).
Van Poppel, H. et al. A prospective randomized EORTC intergroup phase 3 study comparing the complications of elective nephron-sparing surgery and radical nephrectomy for low-stage renal cell carcinoma. Eur. Urol. 51(6), 1606–1615 (2007)
Mir, M. C. et al. Partial nephrectomy versus radical nephrectomy for clinical T1b and T2 renal tumors: a systematic review and meta-analysis of comparative studies. Eur. Urol. 71(4), 606–617 (2017).
Bindayi, A. et al. Trifecta Outcomes of Partial Nephrectomy in patients Over 75 Years Old: Analysis of the REnal SU. Eur Urol Focus. 6(5), 982–90 (2020).
Hung, A. J., Cai, J., Simmons, M. N. & Gill, I. S. “Trifecta” in partial nephrectomy. J. Urol. 189(1), 36–42 (2013)
Ocak, G. et al. Chronic kidney disease and bleeding risk in patients at high cardiovascular risk: a cohort study. J. Thromb. Haemost. JTH. 16(1), 65–73 (2018)
Cheng, H. et al. Prolonged operative duration is associated with complications: a systematic review and meta-analysis. J. Surg. Res. 229, 134–144 (2018)
Peyronnet, B. et al. Comparison of 1800 robotic and open partial nephrectomies for renal tumors. Ann. Surg. Oncol. 23(13), 4277–4283 (2016).
Larcher, A. et al. Is Robot-assisted surgery contraindicated in the case of partial nephrectomy for complex tumours or relevant comorbidities? a comparative analysis of morbidity, renal function, and oncologic outcomes. Eur. Urol. Oncol. 1(1), 61–68 (2018).
Chang, K. D. et al. Functional and oncological outcomes of open, laparoscopic and robot-assisted partial nephrectomy: a multicentre comparative matched-pair analyses with a median of 5 years’ follow-up. BJU Int. 122(4), 618–626 (2018).
Lane, B. R. et al. Active treatment of localized renal tumors may not impact overall survival in patients aged 75 years or older. Cancer 116(13), 3119–3126 (2010).
Hollingsworth, J. M., Miller, D. C., Daignault, S. & Hollenbeck, B. K. Five-year survival after surgical treatment for kidney cancer: a population-based competing risk analysis. Cancer 109(9), 1763–1768 (2007).
Jewett, M. A. S. et al. Active surveillance of small renal masses: progression patterns of early stage kidney cancer. Eur. Urol. 60(1), 39–44 (2011).
Smaldone, M. C. et al. Small renal masses progressing to metastases under active surveillance: a systematic review and pooled analysis. Cancer 118(4), 997–1006 (2012).
Finelli, A. et al. Small renal mass surveillance: histology-specific growth rates in a biopsy-characterized cohort. Eur. Urol. 78(3), 460–467 (2020).
Campbell, S. et al. Renal mass and localized renal cancer: AUA guideline. J. Urol. 198(3), 520–529 (2017).
Thompson, R. H. et al. Comparison of partial nephrectomy and percutaneous ablation for cT1 renal masses. Eur. Urol. 67(2), 252–259 (2015).
Balageas, P. et al. Ten-year experience of percutaneous image-guided radiofrequency ablation of malignant renal tumours in high-risk patients. Eur. Radiol. 23(7), 1925–1932 (2013).
Iannuccilli, J. D. et al. Effectiveness and safety of computed tomography-guided radiofrequency ablation of renal cancer: a 14-year single institution experience in 203 patients. Eur. Radiol. 26(6), 1656–1664 (2016).
Ferakis, N., Bouropoulos, C., Granitsas, T., Mylona, S. & Poulias, I. Long-term results after computed-tomography-guided percutaneous radiofrequency ablation for small renal tumors. J. Endourol. 24(12), 1909–1913 (2010).
Wah, T. M. et al. Radiofrequency ablation (RFA) of renal cell carcinoma (RCC): experience in 200 tumours. BJU Int. 113(3), 416–428 (2014).
Author information
Authors and Affiliations
Contributions
A.I. Manuscript writing/editing. S.D. manuscript writing/editing, project development. K. B. data collection, manuscript editing. P.B. data collection, manuscript editing. P. P. data collection, manuscript editing. J.B.B. data collection, manuscript editing. L.S. data collection, manuscript editing. A.D.L.T. data collection, manuscript editing. C.L. data collection, manuscript editing. H. L. data collection, manuscript editing. F.N. data collection, manuscript editing. J.B.D.C. data analysis. C.D. data collection, manuscript editing. H.B. data collection. M.R. data collection, manuscript editing. T.W. data collection, manuscript editing. J.A.L. data collection, manuscript editing. F.H. data collection, manuscript editing. J.J.P. data collection, manuscript editing. N.D. data collection, manuscript editING. A.M. data collection, manuscript editing. M.N.V. data collection, manuscript editing. J.C.B. data collection, manuscript editing, project development, data management.
Corresponding author
Ethics declarations
Competing interests
Alexandre INGELS Intuitive Surgical. Karim BENSALAH Intuitive Surgical. Alexandre DE LA TAILLE Intuitive Surgical. Jean-Christophe BERNHARD Intuitive Surgical. Other authors declare no competing conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ingels, A., Duc, S., Bensalah, K. et al. Postoperative outcomes of elderly patients undergoing partial nephrectomy. Sci Rep 11, 17201 (2021). https://doi.org/10.1038/s41598-021-96676-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-96676-y
- Springer Nature Limited
This article is cited by
-
Robot-assisted partial nephrectomy in younger versus older adults with renal cell carcinoma: a propensity score-matched analysis
World Journal of Urology (2024)
-
Assessing the safety of minimally invasive partial nephrectomy in octogenarians
Journal of Robotic Surgery (2023)
-
Late complications and 5 years outcomes of robotic partial nephrectomy in France: prospective assessment in the French Kidney Cancer Research Network (UroCCR 10)
World Journal of Urology (2023)
-
Minimally invasive nephron-sparing treatments for T1 renal cell cancer in patients over 75 years: a comparison of outcomes after robot-assisted partial nephrectomy and percutaneous ablation
European Radiology (2023)
-
Perioperative outcomes following robot-assisted partial nephrectomy in elderly patients
World Journal of Urology (2022)