Abstract
A novel composite MOF@cotton fibre (HCF) was prepared and characterized by FTIR, SEM, XPS and TGA. The effect of various parameters on the adsorption efficiency, such as the solution pH, contact time, initial U(VI) concentration and temperature, was studied. The maximal sorption capacity (Qm) is 241.28 mg g−1 at pH 3.0 for U(VI) according to the Langmuir isotherm adsorption model, and the kinetic and thermodynamic data reveal a relatively fast entropy-driven process (ΔH0 = 13.47 kJ mol−1 and ΔS0 = 75.47 J K−1 mol−1). The removal efficiency of U(VI) by HCF is comparable with that of pure cotton fibre and as-prepared MOF (noted as HST). However, the HST composite with cotton fibre significantly improved the treatment process of U(VI) from aqueous solutions in view of higher removal efficiency, lower cost and faster solid–liquid separation. Recycling experiments showed that HCF can be used up to five times with less than 10% efficiency loss.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Uranium (U) is a key radioactive element for nuclear fuel in the nuclear reactors. The remarkable development of nuclear activities related to U such as mining, refinement and recovery of U is of critical importance for the continued development of nuclear energy1,2. To decrease the release of U into the environment and contamination risks in case of incidents or nuclear events, an adsorption approach based on various high-efficiency sorbents has been proposed and attracted considerable attentions3,4,5,6,7. Adsorption has been examined as an alternative method to remove U from aqueous solutions due to its simple operation, low cost, energy saving, high efficiency, etc Cellulose and their derivatives have achieved extensive attention due to their advantages such as the most abundant resource, biocompatibility, inexpensiveness and eco-friendliness8,9. However, pure cellulose generally exhibits poor capacity in practical applications and must be chemically modified10,11,12. The composite of cellulose with other substances is an efficient approach to enhance their performance and extend their applications13,14,15,16.
As a class of important porous materials, metal–organic frameworks (MOFs) have outstanding characteristics including extra-high surface areas, diverse functionalities, linkers tailorability, acid and base robustness and tunable aperture17,18. Thus, they have great potentials in diverse domains such as gas storage and separation19, sensing20, adsorption21,22,23, biomedicine24, magnetism25, luminescence26, environmental remediation27,28,29, and catalysis30,31. MOFs@cellulose composites have been proven to provide better benefits for their versatile applications than pure cellulose or MOFs32. However, most of the recent achievements have highlighted the antibacterial properties33,34,35,36,37, gas adsorption38 and pollutant removal39,40,41,42 of MOF@cellulose composites. To our best knowledge, the integration of HKUST-1 with cellulose to synergistically enhance their intrinsic prominent properties for the removal of U(VI) has never been reported, except for a few reports on the removal of U(VI) using cellulose derivatives43,44,45,46,47,48,49,50,51,52. In addition, the production cost of cellulose derivatives must be significantly reduced to further improve their real applications. Our research has the following contributions: (i) a novel, facile and low-cost method to prepare MOF@cotton fibre (HCF) composite, which uses a much cheaper benzoic acid (BA; the cost of the analytical-grade product is 16.4 ¥/250 g) as the ligand to replace part of the traditional ligand trimesic acid (H3BTC to synthesize HKUST-1; the cost of the analytical-grade product is 566 ¥/500 g); (ii) fibriform HCF, which is very easily separated from the liquid phase; (iii) favourable adsorption efficiency for U(VI), where the maximal adsorption capacity is 241.28 mg g−1.
In the present study, a novel MOF@cotton fibre (HCF) composite was facilely synthesized by a simple hydrothermal method. To characterize the obtained materials, X-ray photoelectron spectroscopy (XPS), FT-IR spectroscopy (FTIR), scanning electron microscopy (SEM) and thermogravimetric analysis (TGA) were used. The effects of a few critical parameters (pH value, contact time, initial U(VI) concentration and temperature) and the adsorption kinetics, adsorption isotherms and reusability of HCF were investigated in detail.
Results and discussion
Characterization
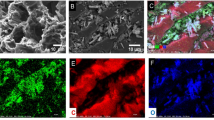
The preparation flow diagram and FTIR and SEM image characterization of the samples are shown in Fig. 1. To reduce the preparation cost of the adsorbent, BA with lower cost was used as the ligand to replace part of the traditional ligand H3BTC. The blue powder product HST was obtained by a simple solvothermal method. While we added CF into the HST precursor (CuII, BA and H3BTC) solution for the solvothermal synthesis, the blue fibrous product HCF was fabricated (see Fig. 1A). The thermal stability of HCF was shown in Fig. 1A.
The functional group structures of the samples were characterized by FTIR spectra as shown in Fig. 1B. The characteristic peaks at 1370 cm–1, 1448 cm–1 and 1647 cm–1 were attributed to C–O, C=O and aromatic C=C of H3BTC, respectively53. The characteristic peaks at 1368 cm–1 and 1443 cm–1 appeared in the FTIR of HCF. Similar characteristic peaks at ~ 3330 cm–1, ~ 2900 cm–1, 1055 cm–1 and 1030 cm–1 appeared in the IR of both HCF and CF. These results show that HST combined with CF successfully to produce HCF.
The surface morphologies of HST and HCF observed in SEM images are shown in Fig. 1C. The as-synthesized HST particles with octahedral and spherical shape (highlighted as yellow dashed circles) were distinctly observed in the surfaces of CF. XPS was used to study the composition and valence of HCF and CF. Figure 2 shows the XPS survey spectra of HCF and CF. In Fig. 2 and the insert, two main peaks at 930.2 and 952.0 eV, which are ascribed to Cu 2p3/2 and Cu 2p1/2 signals, correspond to the characteristic peak of Cu2+ in the XPS survey spectrum of HCF, which indicates the presence of HST in the composite HCF54.
Effect of the pH and ion strength on the U removal
The chemistry species of uranium in the solutions significantly varies at different pH, which makes the solution pH greatly affect the adsorption efficiency. The effect of the solution pH of 2–8 and ion strength with different concentrations of NaClO4 (0.001, 0.01, 0.1 and 1.0 M) on the uranium adsorption is shown in Fig. 3. In Fig. 3 (insert), the removal rate of U(VI) by HCF and CF was maximal at pH = 3. It is well known that the most predominant uranium species is UO22+ at low pH; therefore, the removal rate was low due to the competition between abundant H+ and UO22+ ions55. With increasing solution pH when U is mainly present as negatively charged species (UO2)3(OH)7- and (UO2)(OH)3-, the removal efficiency increased due to the electrostatic attraction between the negatively charged U(VI) species and the positively charged sorbent56. When the pH further increased, the hydrolysation of UO22+ and formation of U(VI)-carbonate species (UO2)2(CO3)(OH)3− inhibited the U(VI) adsorption, which decreased the adsorption capacity57. Therefore, an optimal pH of 3.0 was used in subsequent experiments. In addition, the ion strength clearly had little effect on the adsorption process, which suggests that the inner-sphere surface complexation mechanism plays a main role in the adsorption process58.
Effect of the contact time and kinetic studies
The adsorption efficiency of the adsorbents can be evaluated by the adsorption equilibrium time and kinetic process. Figure 4 presents the effect of the contact time of 5–120 min on the U(VI) sorption by HCF and CF in regard to the kinetics at an initial U(VI) concentration of 10 mg L−1 at pH 3.0 at room temperature. With the extension of time, the adsorption efficiency significantly increased until it reached an equilibrium within 30 min. The removal rate of HCF for U(VI) was nearly 100% within 30 min, while the removal efficiency of CF was poor (only approximately 70%). Pseudo-first-order and pseudo-second-order models were used to study the adsorption kinetics of U(VI) on HCF. Figure 4 (insert) clearly shows that the pseudo-second-order model had a superior correlation coefficient (R2) compared to the pseudo-first-order model, which indicates that the U(VI) adsorption processes of HCF well fit the pseudo-second-order model.
Adsorption isotherms and thermodynamic studies
Langmuir and Freundlich adsorption isotherm models are expressed in Eqs. (1) and (2), respectively59. According to Eqs. (1) and (2), the maximal adsorption capacity (Qm) was fitted when the initial U(VI) concentration was 5–150 mg L−1 (Fig. 5). The calculated Langmuir and Freundlich isotherm parameters from the fitting processes are listed in Table 1. Table 1 shows that Langmuir isotherm fitted the experimental data well with a higher correlation coefficient (R2), and the maximum adsorption capacity was 241.28 mg g−1 for the adsorption of HCF. Moreover, Fig. 5 shows that HCF is a promising sorbent for the removal of uranium from aqueous solutions in terms of the preparation cost, adsorption efficiency and simple solid–liquid separation for uranium from aqueous solutions.
where Qe (mg g−1) is the equilibrium adsorption capacity; Ce (mg L−1) is the uranium concentration at equilibrium; Qm (mg g−1) is the maximum adsorption capacity; KL (L mg−1) and KF (mg1−n Ln g−1) are Langmuir constant and Freundlich constant, respectively; n is Freundlich adsorption exponent.
Dubinin–Radushkevich (D–R) isotherm is usually employed to explain the adsorption mechanism with respect to Gaussian energy distribution onto a heterogeneous surface and determine the adsorption nature as physical or chemical based on the mean free energy (E)60. The D–R isotherm model is expressed by Eq. (3). Model parameters ε, β and E can be determined and calculated by Eqs. (4) and (5).
where β (mol2 (J2)−1) is D–R isotherm constant, and ε (J mol−1) is Polanyi potential. The calculated D–R isotherm parameters are listed in Table 1. From the obtained E value, the physical or chemical sorption mechanism can be revealed. According to the literature61, if E is 8–16 kJ mol−1, the sorption process chemically occurs, whereas E < 8 kJ mol−1 follows the physical sorption. For HCF, the low E value of 0.56 kJ mol−1 in this study suggests that the U adsorption was a physical adsorption process due to electrostatic or Van der Waal’s attractions.
The linear form of the Toth equation was as following62. A linear relationship can be obtained by plotting (ce/qe)T against (ce)T at different T values, and then the values of qT, bT and R2 can be calculated.
where qT (mg g−1) and bT (L mg−1) are the parameters of the Toth equation, T is exponent of the Toth equation.
Figure 6 shows the regression results for the adsorption of uranium on HCF. The best regression line is identified when T is above 0.8 and the R2 value of the line is closest to 1. The calculated values of qT, bT and R2 are also listed in Table 2. Table 2 indicates that the values of R2 increase with the increase of the values of T. As a result, the Toth equation (T > 0.8) reveals better-fitting results than the Langmuir, Freundlich and D–R equations. According to the reference63, T is 0.6–1.0 for the adsorption of uranium, meaning that the adsorption occurs mostly on homogeneous surfaces in this study.
A comparison of the maximum adsorption capacity (Qm) of various adsorbents is shown in Table 3. According to Table 3, the as-synthesized HCF clearly presents higher Qm than other cellulose-based adsorbents. This increased adsorption capacity of HCF for U(VI) may be attributed to its fibrous structure and the addition of HST with higher specific surface area and excellent adsorption efficiency64 in the surface of CF. Qm of the as-prepared HCF shows that HCF is a promising adsorbent for the treatment of uranium-bearing wastewater.
To evaluate the adsorption thermodynamic parameters, the effect of temperature on the uranium removal was investigated using 20 mL solutions containing 0.008 g of HCF and 10 mg L−1 U(VI), which was shaken for 24 h at pH 3.0. After the adsorption equilibrium, adsorption results of U(VI) by HCF were obtained, and the plots of lnKd versus 1/T onto HCF is shown in Fig. 7. Thermodynamic parameters (i.e., enthalpy (ΔH0), entropy (ΔS0) and standard free energy ΔG0) from 303 to 333 K in the adsorption process were calculated from the slope and intercept of the linear line of lnKd versus 1/T according to Vander Hoff Eqs. (7) ~ (9)65. The evaluated ΔH0 value is 13.47 kJ mol−1, which reflects that the adsorption reaction was endothermic. The obtained positive ΔS0 and negative ΔG0 values (Fig. 7 insert) suggest that the adsorption process was spontaneous.
where Kd (mL g−1) is the distribution coefficient of U(VI); cad (mg L−1) is the concentration of metal ions on the adsorbent at equilibrium; ce (mg L−1) is the equilibrium concentration of metal ions in solution; R (8.314 J mol−1 K−1) is the universal gas constant; T (K) is the absolute temperature.
Regeneration and reuse of HCF
The adsorbent reuse is an important index to further evaluate their adsorption performance and reduce the treatment cost. After adsorption and filtration, the adsorbent loaded U(VI) was collected and treated with excess HNO3 (0.1 mol L−1) for 24 h in a shaker. Then, the adsorbent was separated from the liquid by filtration and washed several times by DW to be used for the next cycle adsorption experiment. The reuse experiments of HCF were performed for five cycles. According to Fig. 8, the U(VI) removal percentage with HCF was 99.11% after the first cycling experiment. After five recycling experiments, the U(VI) removal percentage remained at 88.24%, which suggests that HCF can be recovered and reused several times and has favourable recycling capability.
Materials and methods
Materials
The cotton fibre (CF) in this study was medical purified cotton from Xvzhou hygienic materials plant (Jiangsu, China). Copper(II) nitrate trihydrate (Cu(NO3)2·3H2O) was provided by Chengdu Kelong Chemical Co., Ltd. Benzoic acid (BA), and trimesic acid (H3BTC) was purchased from Aladdin Chemical Reagent Co., Ltd. All reagents were of analytical grade and used without further purification. UO2(NO3)2·6H2O was provided by Xi’an Dingtian Chemical Reagent Co., Ltd. The deionized water (DW) was used throughout the experiments.
Synthesis
HCF was synthesized through a simple solvothermal method. In a typical synthesis of HCF, 1.0870 g of Cu(NO3)2·3H2O was dissolved in 15 mL DW. The mixture of 0.3948 of H3BTC and 0.0769 g of BA was dissolved in 15 mL absolute ethanol. Then, both solutions were mixed and stirred for 30 min. The resulting mixture and 0.5 g of CF were transferred into a Teflon autoclave and heated in an oven at 110 ℃ for 24 h. The resultant blue fibrous products (HCF) were filtered in vacuum and completely washed by ethanol and DW. Finally, the products were dried at 100 ℃ in vacuum.
In the above preparation process of HCF, by the hydrothermal synthesis method firstly H3BTC react with BA in a Teflon autoclave to obtain the MOF product (noted as HST), and then HST composites with the raw material CF to produce HCF.
Characterization
The structure characteristics of HCF, CF and HST were analysed by Fourier transform infrared (FTIR) spectroscopy (Bruker VERTEX 70, Germany). The morphology characteristics of the samples were obtained on a scanning electron microscope (SEM) (Helios 600i, Japan). X-ray photoelectron spectroscopy (XPS) of HCF and CF were studied using an ESCALAB 250 X-ray photoelectron spectroscope (Thermo fisher, USA). Thermal stability of the products was studied by thermogravimetric analysis (TGA) spectroscopy (Netzsch STA449F5, Germany) from 30 to 900 ℃ at a heating rate of 10 K/min under an argon flow.
Adsorption studies
The U(VI) stock solutions were prepared by dissolving UO2(NO3)2·6H2O in DW; then, small amounts of concentrated HNO3 was added to avoid the hydrolysis of UO22+. The working U(VI) solutions were prepared by appropriately diluting the stock solutions immediately before their use. The adsorption capacities of U(VI) onto HCF and CF were investigated as a function of the solution pH, contact time, initial U concentration and temperature by batch adsorption experiments. The solution pH was adjusted by adding NaOH and HCl and measured using a glass electrode (Leici PHS-3C, China). HCF or CF was added to 20 mL U(VI) solution and shaken in a shaker (Kangshi, China). After filtration, the U(VI) concentration in the solution was determined by a micro-quantity uranium analyser (MUA model, China). All experiments were performed in triplicate, and the data are presented as the mean values. The removal efficiency (R (%)) and adsorption amount of U(VI) on HCF or CF (q (mg g−1)) of U(VI) in solution were calculated using Eqs. (10) and (11)59, respectively.
where c0 (mg L−1) is the initial U(VI) concentration, ct (mg L−1) is the U(VI) concentration at time t in the solution, V (L) is the solution volume, and m (g) is the adsorbent mass.
In the desorption experiments, the obtained U(VI)-loaded HCF was washed with DW and rinsed in 3 M HNO3 for 24 h; then, it was thoroughly washed in DW until U(VI) ions were not detected in the rinsing solution. The dried and regenerated adsorbent was reused for further adsorption experiments, and this recycling procedure was repeated five times.
Conclusion
The composite HCF with lower cost was fabricated via a facile solvothermal approach to adsorb U(VI) from aqueous solutions. The preparation cost of the adsorbent was significantly reduced by using BA with lower cost as the ligand to replace part of the traditional ligand H3BTC. HCF shows favourable adsorption capacity for U(VI) with maximum adsorption capacities of 241.28 mg g−1 at pH 3.0 compared to CF. HCF shows favourable regenerability, and the U(VI) removal percentage was 88.24% after five cycles. This work offers a new and cost-effective adsorbent HCF, which can be effectively used as a promising sorbent to remove U(VI) from the real multi-component U(VI)-containing nuclear waste influents. Furthermore, due to the advantageous fibrous form, HCF can be easily separated from aqueous solutions, which enhances post-treatment efficiency for further practical applications.
References
Xiong, Y. Y. et al. MOF catalysis of FeII-to-FeIII reaction for an ultrafast and one-step generation of the Fe2O3@MOF composite and uranium(VI) reduction by iron(II) under ambient conditions. Chem. Commun. 52, 9538–9541 (2016).
Luo, W. et al. Engineering robust metal-phenolic network membranes for uranium extraction from seawater. Energy Environ. Sci. 12, 607–614 (2019).
Liu, W. J. et al. Efficiency and mechanism of adsorption of low-concentration uranium from water by a new chitosan/aluminum sludge composite aerogel. Dalton Trans. 49(10), 3209–3221 (2020).
Abney, C. W., Mayes, R. T., Saito, T. & Dai, S. Materials for the recovery of uranium from seawater. Chem. Rev. 117, 13935–14013 (2017).
Sun, Q. et al. Bio-inspired nano-traps for uranium extraction from seawater and recovery from nuclear waste. Nat. Commun. 9, 1644 (2018).
Wang, F. et al. Study of adsorption performance and adsorption mechanism for U(VI) ion on modified polyacrylonitrile fibers. J. Radioanal. Nucl. Chem. 323, 365–377 (2020).
Huang, S. Y. et al. Dual functional nanocomposites of magnetic MnFe2O4 and fluorescent carbon dots for efficient U(VI) removal. Chem. Eng. J. 368, 941–950 (2019).
Xie, Y. et al. Emerging natural and tailored materials for uranium-contaminated water treatment and environmental remediation. Prog. Mater. Sci. 103, 180–234 (2019).
Garza-Cervantes, J. A. et al. Antimicrobial activity of a silver-microfibrillated cellulose biocomposite against susceptible and resistant bacteria. Sci. Rep. 10, 7281 (2020).
Hausmann, M. K. et al. Complex-shaped cellulose composites made by wet densification of 3D printed scaffolds. Adv. Funct. Mater. 30, 1904127 (2020).
Guo, L. F., Li, D. F., Lennholm, H., Zhai, H. M. & Ek, M. Structural and functional modification of cellulose nanofibrils using graft copolymerization with glycidyl methacrylate by Fe2+–thiourea dioxide–H2O2 redox system. Cellulose 26, 4853–4864 (2019).
Liu, X. L. et al. Improving salt tolerance and thermal stability of cellulose nanofibrils by grafting modification. Carbohyd. Polym. 211, 257–265 (2019).
Li, M. F. et al. PVA-co-PE nanofiber/epoxy composites with cellulose nanocrystals. Compos. Sci. Technol. 188, 107990 (2020).
Chen, W., Yuan, Y. & Chen, Y. L. Visualized bond scission in mechanochemiluminescent polymethyl acrylate/cellulose nanocrystals composites. ACS Macro Lett. 9(4), 438–442 (2020).
Onur, A., Ng, A., Garnier, G. & Batchelor, W. The use of cellulose nanofibres to reduce the wet strength polymer quantity for development of cleaner filters. J. Clean. Prod. 215, 226–231 (2019).
Chen, J. et al. Direct reduction of graphene oxide/nanofibrillated cellulose composite film and its electrical conductivity research. Sci. Rep. 10, 3124 (2020).
Cui, W. G., Hu, T. L. & Bu, X. H. Metal-organic framework materials for the separation and purification of light hydrocarbons. Adv. Mater. 32, 1806445 (2020).
Lo, S. H. et al. Rapid desolvation-triggered domino lattice rearrangement in a metal-organic framework. Nat. Chem. 90, 90–97 (2020).
Knebel, A. et al. Defibrillation of soft porous metal-organic frameworks with electric fields. Science 358, 347–351 (2017).
Wu, S. Y., Min, H., Shi, W. & Cheng, P. Multicenter metal-organic framework-based ratiometric fluorescent sensors. Adv. Mater. 32, 18058 (2020).
Zheng, J. et al. Molecular insight into fluorocarbon adsorption in pore expanded metal−organic framework analogs. J. Am. Chem. Soc. 142, 3002–3012 (2020).
Bhadra, B. N., Yoo, D. K. & Jhung, S. H. Carbon-derived from metal–organic framework MOF-74: A remarkable adsorbent to remove a wide range of contaminants of emerging concern from water. Appl. Surf. Sci. 504, 144348 (2020).
Dhaka, S. et al. Metal−organic frameworks (MOFs) for the removal of emerging contaminants from aquatic environments. Coord. Chem. Rev. 380, 330–352 (2019).
Pang, Y. C. et al. Metal−organic framework nanoparticles for ameliorating breast cancer-associated osteolysis. Nano Lett. 20, 829–840 (2020).
Gonzalez, M. I. et al. Confinement of atomically defined metal halide sheets in a metal–organic framework. Nature 577, 64–68 (2020).
Wang, Z. et al. Breathing-ignited long persistent luminescence in a resilient metal−organic Framework. Chem. Mater. 32, 841–848 (2020).
Yu, F. et al. Enhancing the separation efficiency of a C2H2/C2H4 mixture by a chromium metal–organic framework fabricated via post-synthetic metalation. J. Mater. Chem. A 8, 2083–2089 (2020).
Xin, Z. F. et al. Metallocene implanted metalloporphyrin organic framework for highly selective CO2 electroreduction. Nano Energy 67, 104233 (2020).
Yang, W. P. et al. Applications of metal–organic-framework-derived carbon materials. Adv. Mater. 31, 1804740 (2019).
Jiang, Z. W. et al. Controllable synthesis of porphyrin-based 2D lanthanide metal–organic frameworks with thickness- and metal-node-dependent photocatalytic performance. Angew. Chem.-Int. Edit. 59, 3300–3306 (2020).
Wang, Z. X. et al. Metal–organic frameworks and their derivatives with graphene composites: Preparation and applications in electrocatalysis and photocatalysis. J. Mater. Chem. A 8, 2934–2961 (2020).
Zhou, S. Y., Strømme, M. & Xu, C. Highly transparent, flexible, and mechanically strong nanopapers of cellulose nanofibers@metal–organic Frameworks. Chem. Eur. J. 3, 3515–3520 (2019).
Ma, S. S. et al. Multifunctional cellulose-based air filters with high loadings of metal–organic frameworks prepared by in situ growth method for gas adsorption and antibacterial applications. Cellulose 25, 5999–6010 (2018).
Rickhoff, T. A., Werth, L. K., Kissel, D. S., Keleher, J. J. & Sullivan, E. A biomimetic cellulose-based composite material that incorporates the antimicrobial metal-organic framework HKUST-1. J. Appl. Polym. Sci. https://doi.org/10.1002/APP.46978 (2019).
Wang, C., Qian, X. R. & An, X. H. In situ green preparation and antibacterial activity of copper-based metal–organic frameworks/cellulose fibers (HKUST-1/CF) composite. Cellulose 22, 3789–3797 (2015).
Rodríguez, H. S. et al. Antibacterial activity against escherichia coli of Cu-BTC (MOF-199) metal-organic framework immobilized onto cellulosic fibers. J. Appl. Polym. Sci. https://doi.org/10.1002/APP.40815 (2014).
Duan, C. et al. Synthesis of novel cellulose- based antibacterial composites of Ag nanoparticles@ metal–organic frameworks@ carboxymethylated fibers. Carbohyd. Polym. 193, 82–88 (2018).
Yang, Q., Zhang, M. Y., Song, S. X. & Yang, B. Surface modification of PCC filled cellulose paper by MOF-5 (Zn3(BDC)2) metal–organic frameworks for use as soft gas adsorption composite materials. Cellulose 24, 3051–3060 (2017).
Lu, H. L. et al. Cellulose-supported magnetic Fe3O4–MOF composites for enhanced dye removal application. Cellulose 26, 4909–4920 (2019).
Cui, X. F. et al. In-situ fabrication of cellulose foam HKUST-1 and surface modification with polysaccharides for enhanced selective adsorption of toluene and acidic dipeptides. Chem. Eng. J. 369, 898–907 (2019).
Shen, C. K. et al. Catalytic MOF-loaded cellulose sponge for rapid degradation of chemical warfare agents simulant. Carbohyd. Polym. 213, 184–191 (2019).
Lange, L. E. & Obendorf, S. K. Functionalization of cotton fiber by partial etherification and self-assembly of polyoxometalate encapsulated in Cu3(BTC)2 metal−organic framework. ACS Appl. Mater. Inter. 7, 3974–3980 (2015).
Orabi, A. H. et al. Potentiality of uranium adsorption from wet phosphoric acid using amine-impregnated cellulose. J. Radiat. Res. Appl. Sci. 9, 193–206 (2016).
Orabi, A. H. Synthesis of a cellulose derivative for enhanced sorption and selectivity of uranium from phosphate rocks prior to its fluorometric determination. Int. J. Environ. Anal. Chem. 99(8), 741–766 (2019).
Wu, L. P., Lin, X. Y., Du, X. C. & Luo, X. G. Biosorption of uranium(VI) from aqueous solution using microsphere adsorbents of carboxymethyl cellulose loaded with aluminum(III). J. Radioanal. Nucl. Chem. 310, 611–622 (2016).
Rule, P., Balasubramanian, K. & Gonte, R. R. Uranium(VI) remediation from aqueous environment using impregnated cellulose beads. J. Environ. Radioactiv. 136, 22–29 (2014).
Yousif, A. M., El-Afandy, A. H., Wahab, G. M. A., Mubark, A. E. & Ibrahim, I. A. Selective separation of uranium(VI) from aqueous solutions using amine functionalized cellulose. J. Radioanal. Nucl. Chem. 303, 1821–1833 (2015).
Başarır, S. Ş & Bayramgil, N. P. The uranium recovery from aqueous solutions using amidoxime modified cellulose derivatives. IV. Recovery of uranium by amidoximated hydroxypropyl methylcellulose. Cellulose 20, 827–839 (2013).
Anirudhan, T. S. & Sreekumari, S. S. Synthesis and characterization of a functionalized graft copolymer of densified cellulose for the extraction of uranium(VI) from aqueous solutions. Colloid. Surface. A 361, 180–186 (2010).
Anirudhan, T. S., Nima, J. & Divya, P. L. Adsorption and separation behavior of uranium(VI) by 4-vinylpyridine-grafted-vinyltriethoxysilane-cellulose ion imprinted polymer. J. Environ. Chem. Eng. 3, 1267–1276 (2015).
Villalobos-Rodríguez, R., Montero-Cabrera, M. E., Esparza-Ponce, H. E., Herrera-Peraza, E. F. & Ballinas-Casarrubias, M. L. Uranium removal from water using cellulose triacetate membranes added with activated carbon. Appl. Radiat. Isotopes 70, 872–881 (2012).
Zolfonoun, E. & Yousefi, S. R. On-line extraction and determination of uranium in aqueous samples using multi-walled carbon nanotubes-coated cellulose acetate membrane. Int. J. Environ. Anal. Chem. 96(3), 203–211 (2016).
Lin, K. Y. A. & Hsieh, Y. T. Copper-based metal organic framework (MOF), HKUST-1, as an efficient adsorbent to remove p-nitrophenol from water. J. Taiwan Inst. Chem. E. 50, 223–228 (2015).
Gu, C. M. et al. Ultrasensitive non-enzymatic pesticide electrochemical sensor based on HKUST-1-derived copper oxide @ mesoporous carbon composite. Sens. Actuators B Chem. 305, 127478 (2020).
Wang, W. X., Chen, Z., Zhou, H. J., Zhang, Y. F. & Wang, X. K. Two-dimensional lamellar magnesium silicate with large spacing as an excellent adsorbent for uranium immobilization. Environ. Sci. Nano 5, 2406–2414 (2018).
Li, M. X., Liu, H. B., Chen, T. H., Dong, C. & Sun, Y. B. Synthesis of magnetic biochar composites for enhanced uranium (VI) adsorption. Sci. Total Environ. 651, 1020–1028 (2019).
Meng, J. et al. Adsorption capacity of kelp-like electrospun nanofibers immobilized with bayberry tannin for uranium(VI) extraction from seawater. RSC Adv. 9, 8091–8103 (2019).
Zhu, J. H. et al. Metal–organic frameworks (MIL-68) decorated graphene oxide for highly efficient enrichment of uranium. J. Taiwan Inst. Chem. E. 99, 45–52 (2019).
Yu, S. J. et al. Efficient removal of uranium(VI) by layered double hydroxides supported nanoscale zero-valent iron: A combined experimental and spectroscopic studies. Chem. Eng. J. 365, 51–59 (2019).
Muthukumaran, C., Sivakumar, V. M. & Thirumarimurugan, M. Adsorption isotherms and kinetic studies of crystal violet dye removal from aqueous solution using surfactant modified magnetic nanoadsorbent. J. Taiwan Inst. Chem. E. 63, 354–362 (2016).
Liu, R., Zhang, W., Chen, Y. T. & Wang, Y. S. Uranium (VI) adsorption by copper and copper/iron bimetallic central MOFs. Colloid. Surface. A. https://doi.org/10.1016/j.colsurfa.2019.124334 (2020).
Wu, K. T., Wu, P. H., Wu, F. C., Jreng, R. L. & Juang, R. S. A novel approach to characterizing liquid-phase adsorption on highly porous activated carbons using the Toth equation. Chem. Eng. J. 221, 373–381 (2013).
Toth, J. Uniform interpretation of gas/solid adsorption. Adv. Colloid Interface Sci. 55, 1–239 (1995).
Yang, A. L., Li, P. & Zhong, J. R. Facile preparation of low-cost HKUST-1 with lattice vacancies and high-efficiency adsorption for uranium. RSC Adv. 9, 10320–10325 (2019).
Abdi, S., Nasiri, M., Mesbahi, A. & Khani, M. H. Investigation of uranium (VI) adsorption by polypyrrole. J. Hazard. Mater. 332(1), 132–139 (2017).
Yang, A. L., Wu, J. H. & Huang, C. P. Graphene oxide-cellulose composite for the adsorption of uranium(VI) from dilute aqueous solutions. J. Hazard. Toxic Radioact. Waste 22(2), 04017029 (2018).
Zong, P. F. et al. Carboxymethyl cellulose supported magnetic graphene oxide composites by plasma induced technique and their highly efficient removal of uranium ions. Cellulose 26, 4039–4060 (2019).
Cai, Y. W. et al. Rational synthesis of novel phosphorylated chitosan-carboxymethyl cellulose composite for highly effective decontamination of U(VI). ACS Sustain. Chem. Eng. 7, 5393–5403 (2019).
El-Bohy, M. N. et al. Grafting of arginine and glutamic acid onto cellulose for enhanced uranyl sorption. Cellulose 24, 1427–1443 (2017).
Wu, L. P., Lin, X. Y., Zhou, X. B. & Luo, X. G. Removal of uranium and fluorine from wastewater by double-functional microsphere adsorbent of SA/CMC loaded with calcium and aluminum. Appl. Surf. Sci. 384, 466–479 (2016).
Shao, D. D. et al. Immobilization of uranium by biomaterial stabilized FeS nanoparticles: Effects of stabilizer and enrichment mechanism. J. Hazard. Mater. 302, 1–9 (2016).
Anirudhan, T. S., Deepa, J. R. & Binusreejayan. Synthesis and characterization of multi-carboxyl-functionalized nanocellulose/nanobentonite composite for the adsorption of uranium(VI) from aqueous solutions: Kinetic and equilibrium profiles. Chem. Eng. J. 273, 390–400 (2015)
Popescu, I. C. et al. Removal of uranium (VI) from aqueous systems by nanoscale zero-valent iron particles suspended in carboxy-methyl cellulose. J. Nucl. Mater. 443, 250–255 (2013).
Acknowledgements
This work is financially supported by National Natural Science Foundation of China (21407132) and Environmental Protection Foundation of China Academy of Engineering Physics (No. YAHZY-2018-008).
Author information
Authors and Affiliations
Contributions
A.Y. contributed to the experiments design and carried out the adsorbents preparation and adsorption experiments. Z.W. and Y.Z. performed the adsorption tests. A.Y. prepared the figures and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, A., Wang, Z. & Zhu, Y. Facile preparation and adsorption performance of low-cost MOF@cotton fibre composite for uranium removal. Sci Rep 10, 19271 (2020). https://doi.org/10.1038/s41598-020-76173-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-76173-4
- Springer Nature Limited
This article is cited by
-
Preparation of phosphinimide and its adsorption of uranium in aqueous solution
Research on Chemical Intermediates (2024)
-
Novel Zn metal–organic framework with the thiazole sites for fast and efficient removal of heavy metal ions from water
Scientific Reports (2023)
-
One-step rapid synthesis of HKUST-1 and the application for europium(III) adsorbing in solution
Journal of Radioanalytical and Nuclear Chemistry (2022)
-
Phosphate group functionalized magnetic metal–organic framework nanocomposite for highly efficient removal of U(VI) from aqueous solution
Scientific Reports (2021)
-
Removal of uranium(VI) from simulated wastewater by a novel porous membrane based on crosslinked chitosan, UiO-66-NH2 and polyvinyl alcohol
Journal of Radioanalytical and Nuclear Chemistry (2021)