Abstract
Riparian vegetation plays an important role in providing energy to small watercourses and maintaining ecological processes through organic matter input and together with hydrological and geomorphological watercourse characteristics influence on fish assemblages. The goal of this paper was partitioning and quantifying the influence of riparian zone (type of riverbank substrate, bank slope, type of riparian vegetation cover and percentage of riparian vegetation cover on the main channel), physical habitat (stream channel width and depth, type of substrate and aquatic habitat in channel, water velocity and organic matter), water quality (turbidity, temperature, conductivity, pH, dissolved oxygen and chlorophyll concentration) and spatial variables (linear distances between sampled points) on fish assemblages (richness and abundance per species) in headwater streams of the Upper Paraná River basin, Central Brazil. For this purpose, it was performed a variation partitioning analysis between riparian, physical habitat, water and spatial variables sets and a Redundancy Analysis to quantify the influence of variables on the fish assemblages. Only the physical habitat and water quality variables influenced the fish assemblages (richness and abundance per species).
Similar content being viewed by others
Introduction
Freshwater fish assemblages are structured by variables related to both water quality and riparian vegetation1,2,3,4,5,6. In this sense, warmer waters exhibit higher fish abundance and biomass while highly oxygenated waters may lead to greater species diversity7,8,9. Riparian vegetation is a transitional semiterrestrial system10 that provides energy in watercourses through the input of organic matter2. Leaves deposited on the watercourse bed contribute indirectly to fish food because they act as a substrate for numerous microorganisms11 and insects12,13. In addition, riparian trees and roots restrict channel widening, cause channel deepening and add coarse woody debris favoring fish concealment and channel complexity.
The influences of water and riparian vegetation on fish assemblages are not independent2,10,14,15; that is, riparian vegetation may directly or indirectly influence water variables16. For example, water temperature is directly influenced by riparian vegetation, which regulates the watercourse insolation level17,18 and influences primary production19. Conversely, channel depth and substrate heterogeneity are indirectly influenced by riparian vegetation because the riparian zone regulates the entry of sediment that can be deposited into the watercourse10,20,21.
Another factor that should not be neglected is the spatial factor (e.g., the river network), which includes geographical barriers that hamper or prevent species migration between locations. Abundance and richness are diversity metrics that are spatially structured22,23,24,25,26,27,28. Spatial factors are a consequence of the geological and local climatic influence on the streams in a river network29,30,31 and the position of the watercourse along a longitudinal gradient (upstream-downstream32) for the 1st-3rd 33 and 4th-7th order34 streams. A spatial model coupled with a river network accurately explains fish richness patterns35,36. Additionally, the river network acts as a corridor37, facilitating fish species dispersion38 or acting as a filter39.
Furthermore, the individual influence of water, riparian or spatial processes on the structure of fish assemblages is not necessarily consistent40. Instead, the influence of these processes results more often from their interaction41. Therefore, physical habitat variables influence fish assemblages either alone27 or in combination with water quality variables42.
The aim of this paper was to partition and quantify the influence of riparian, physical habitat, water quality and spatial variables on fish assemblages in headwater streams located in the Upper Paraná River basin, Central Brazil.
Results
A total of 4879 specimens belonging to 59 species and 19 families were collected (Table 1).
Influence of the environmental conditions on fish assemblages
The variation partitioning analysis indicated that fish abundance variation is explained by water quality (18.7% of variation), physical habitat (8.4%), spatial (6.2%) and riparian zone variables (5.1%; Fig. 1). The interactions among the spatial variables, water quality and physical habitat explain 16.7% of the variation, those among the physical habitat, riparian zone and water quality explain 8.9%, those between the physical habitat and water quality explain 8.2%, and the other interactions represent ≤3.4% (Fig. 2). The Procrustes analyses indicated a significant correlation between the fish abundance and the physical habitat (M2 = 0.295; p < 0.001) and water quality variables (M2 = 0.565; p < 0.001) and no significance for the riparian zone (M2 = 0.200; p = 0.526) or spatial variables (M2 = 0.150; p = 0.744). All the non-metric multidimensional scaling NDMS analyses performed had a good fit (stress < 0.02).
Analyses of redundancy (RDA) output correlating stream fish assemblage to environmental water variables. aspfus = Aspidoras fuscoguttatus; astalt = Astyanax altiparanae; astfas = Astyanax fasciatus; astsca = Astyanax scabripinnis; brystr = Bryconamericus stramineus; poeret = Poecilia reticulate; piaarg = Piabina argentea; steins = Steindachnerina insculpta; CO = conductivity; CL = chlorophyll concentration; MO = organic matter; CW = channel width; TU = turbidity; DO = dissolved oxygen; CD = channel depth; WT = water temperature. P1 – P22 = stream sites. Only species with >90.0% of contribution to the structure of RDA are represented.
Fish assemblages-environmental conditions relationships
According to the broken stick criteria, there were two significant axes for the PCAs performed separately on the water quality (77% of the variation) and physical habitat (81% of the variation), eight axes for the PCA performed on the riparian zone (87% of the variation) and three significant MENs (Moran’s Index = 0.01 for each one) for the spatial variables.
The multiple linear regression showed no significant relation between fish abundance and the variables of the four groups considered (r2 = 0.566; F (12, 27) = 2.536; p = 0.128). In contrast, a significant relation was observed between fish richness (r² = 0.784; F (12, 27) = 5.865; p = 0.001) and the PCA-1 of the physical habitat variables (p = 0.005; Table 2). All of the other compartments did not display significant relationship (Table 2).
The relationship between assemblage and physical habitat variables is detailed by the RDA (total variance explained by the two axes = 53.4%; F (10, 17) = 3.543; p = 0.003). The first axis (35.17%) was positively correlated with conductivity, dissolved oxygen and chlorophyll concentration and negatively correlated with water temperature, whereas the second axis (18.23%) was positively correlated with organic matter, channel depth, pH and channel width and negatively correlated with turbidity (Fig. 2). The characins Piabina argentea and Astyanax altiparanae and the scrapetooths Parodon nasus were related to high values of water conductivity, dissolved oxygen and chlorophyll concentration, whereas the characins Astyanax fasciatus and Astyanax scabripinnis and the poeciliid Poecilia reticulata were associated with elevated water temperature values. The scrapetooths Apareiodon ibitiensis, the headstander Leporinus microphthalmus, and the toothless Steindachnerina insculpta were associated with elevated organic matter and pH values and a large and deep channel stream. The characin Bryconamericus stramineus, the callichthyid armored catfish Aspidoras fuscoguttatus and the South American darter Characidium zebra were correlated with high values of turbidity (Fig. 2).
Discussion
The riparian zone does not display any significant influences on fish abundance or richness in the headwater streams sampled. Similar results using a different methodology were obtained for fish diversity43 in 1st to 3rd order headwaters streams in the Amazon region. This result suggests a low influence of riparian vegetation removal, assessed indirectly in this paper by the variables of the riparian zone group (type and percentage of the vegetation cover), on fish assemblages. However, studies focused on this subject have stressed the influence of the riparian zone on fish assemblages in the Amazon (channel fragmentation, deforestation44; mechanized agriculture43), São Francisco (deforestation42,45) and Paraná River basin (deforestation45), the last two of which contain the same vegetation cover of the area sampled in this paper (i.e., Cerrado).
The spatial component also showed no significant influence on fish assemblages. The abundance and richness of plants and animals, including stream organisms, are spatially structured45,46 because of the influence of geology, the local climate30 and the watercourse position along a longitudinal gradient32, especially for 1st to 3rd order streams33. However, if the 1st and 2nd order streams sampled in this study were in the same geologic (a combination of Precambrian metamorphic rocks, continental sedimentary rocks and tholeiitic basalts47) and climatic (tropical climate with a dry season) domain, a similarity of fish abundance and richness could be expected. It suggest that the influence of environmental conditions and resources appear to be more influent than the spatial process, even that the sample sites are located in different basins.
In this study, fish richness was influenced by physical habitat (stream channel width and depth, and organic matter) and water quality (conductivity, water temperature, pH, chlorophyll, dissolved oxygen, and turbidity) variables. These variables are known to structure not only fish assemblages4,48,49 but also their specific attributes, such as richness50,51,52,53. The results agree with those reported for Amazonian43 and Cerrado fish assemblages of 1st to 3rd order headwater streams42, although some previous studies did not separate the influence of physical habitat and water quality variables from those of the riparian zone, as was done in this paper. Additionally, these physical habitat and water quality variables are better predictors of fish assemblage variability than riparian or catchment variables43 or land use and the geophysical landscape42 in Amazon and Cerrado headwater streams, respectively.
The influence of water conductivity on fish assemblages, as observed in this study, was also reported for tropical54 and temperate watercourses51. Conductivity is a surrogate or correlate of water productivity, which influences freshwater fish body condition45, because it measures the electrical conductivity resulting from the concentration of dissociated ions55. Fish species can prefer aquatic habitats with specific requirements, such as elevated values of water conductivity, dissolved oxygen and chlorophyll concentration (as seen in the scrapetooths Parodon nasus and the characins Astyanax altiparanae and Piabina argentea in the watercourses sampled). In the case of P. nasus, the relationship observed is explained because this species is found in riffles56 where there are elevated levels of dissolved oxygen. Furthermore, P. nasus, a periphyton scraper that prefers rocky substrates where algae and bryophytes are abundant, is associated with waters with high conductivity because of eutrophication57. On the other hand, the characin A. altiparanae is considered tolerant to aquatic environmental changes and disturbances such as pollution58, which elevates water conductivity, and displays adaptations (i.e., a projection of the lower lip increase oxygen capture from water surface) to survive in low concentrations of dissolved oxygen56. Finally, the characin P. argentea is a midwater swimmer described as an opportunistic generalist species abundant in disturbed watercourses (modified from lotic to lentic conditions)59 that is also positively correlated to dissolved oxygen concentrations in streams of the Upper Paraná River basin60.
The poecilid P. reticulata, an exotic species in Brazilian watercourses, and the characin A. fasciatus are tolerant to habitat alterations57,61. Additionally, A. fasciatus and A. scabripinis (to a lesser extent62) are sensitive to water temperature because of the influence on their reproduction cycles63, whereas P. reticulata displays female-choice sexual selection64, fry production65, schooling behavior66, and aquatic surface respiration (ASR) to meet oxygen demand in hypoxic water67 regulated by the water temperature. These relationships explain the affinity of these species for the water temperatures found in the streams sampled. However, this affinity, especially for P. reticulata and A. scabripinis, can change during the low- and high-water seasons, when both species are associated with low water temperature68.
The accumulation of organic matter, such as trunks and bundles of leaves, may be responsible for species coexistence in different habitats. This coexistence can occur because of the increase in habitat heterogeneity resulting from organic matter input69,70 from the surrounding riparian zone or the transport of leaves and other matter from upstream to downstream71,72,73,74,75, which are then deposited in stream areas with low water velocity76. This seems to be the case in this study for the scrapetooths Apareiodon ibitiensis, a detritivorous species that scrape the algal film adhered on the surfaces of rocks and logs77, the toothless characin Steindachnerina insculpta, a bottom feeding fish55, and the headstander Leporinus microphthalmus, which, like other anostomids, feeds on sponges, detritus, insects, seeds, leaves, and filamentous algae, in the substrate78,79.
Additionally, the preference of these species for relatively large and deep streams can be related to their body length (A. ibitiensis = 11.3 cm, S. insculpta = 16.1 cm, L. microphthalmus = 11,8 cm54), as reported for A. ibitiensis80. However, the results found can be influenced by local or regional modifications. For example, the fragmentation of a channel or watercourse and local/regional deforestation influence the organic matter inputs (leaves, trunks and stems in this case), habitat complexity and riverbed stability. This, in turn, influences fish richness, as pointed out for Amazonian headwater streams44.
Among the species sampled, the callichthyid armored catfishes Aspidoras fuscoguttatus, the characin Bryconamericus stramineus and the South American darter Characidium zebra are associated with high water turbidity. The callichthyid A. fuscoguttatus is a bottom dwelling species that swims near the watercourse substrate gathering food (“grubber excavating while moving”81). This behavior can explain its ability to exploit the watercourse substrates, which are covered by fine sediments56 that are transported by water, and its capacity to survive in streams that have remarkable seasonal oscillation in turbidity, with lower values during the dry period and higher values in the rainy period82. On the other hand, the characin B. stramineus is a predominantly insectivorous83 active swimmer84 that is abundant in shallow streams of the Upper Paraná basin with elevated turbidity83,85 and water velocity85. The relationship of C. zebra with water turbidity is unexpected considering that it is an indicator species of pristine environments, with a sit-and-wait behavior for capturing prey86 and rheophilic preferences that can be affected by high levels of suspended sediments in the water column and the resulting siltation of the substrate54.
Among the four groups of environmental variables considered, only those related to the physical habitat and water quality significantly influenced the richness of the fish assemblages. This influence is explained by the interaction of the fish assemblages with nine variables (conductivity, water temperature, pH, chlorophyll, organic matter, dissolved oxygen, turbidity, channel width and channel depth). These results indicate that local instream characteristics of headwater streams have more influence on fish assemblages than factors associated with the riparian zone in Cerrado river basin draining areas. The comparison between these findings and those from the Amazon River basin suggests that this influence exists regardless of the river basin and its vegetation cover (Cerrado and Amazon in this case).
Materials and Methods
Study area
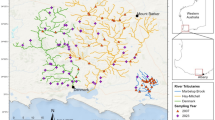
Twenty-seven sites (one sample site per stream) of the 1st and 2nd order tributaries of the Meia Ponte River (seven streams; 2.7 to 10.2 km apart from each other), Piracanjuba River (14; 4.8 to 17.8 km) and Santa Maria River (six; 4.8 to 6.0 km) were sampled, all of which are located in the Southeast Region, Goiás state, Upper Paraná River basin, Central Brazil (Fig. 3, Table 3). Sampling was conducted between April and September 2009, which corresponded with the dry season of the regional climate (Aw per the Köppen-Geiger classification). The Paraná River basin drainage is located on sedimentary deposits corresponding with the Paleozoic and Cenozoic and covered by basalt from the Jurassic-Cretaceous age47. The sampling stations are located on a combination of three types of rocks: i) Precambrian metamorphic rocks; (ii) continental sedimentary rocks; and (iii) tholeiitic basalts, which are abundant in the Paraná basin47. The vegetation cover of the Meia Ponte and Piracanjuba River basin was deciduous forest, and that of the Santa Maria basin was a semideciduous forest, all of which belong to the Cerrado (the Brazilian savanna biome).
In each stream, one 100-m site was selected according to its accessibility, marked and georeferenced (Garmin GPSMAP64. Each site was divided into 11 transects, one every ten meters, where the data collection for both fish assemblages and variables was performed.
All sites were away from urban areas and were found in a landscape matrix formed mainly by pasture. The exception was site P17, which was surrounded by a sugarcane crop. The sites sampled had riparian vegetation covering the stream channel and at least one opening, which was intended for watering livestock or replaced by grass for feeding cattle (site P5), in the riparian cover along the site. The channel depth of the stream sites ranged from a minimum of 0.10 (P2 and P20) to a maximum of 0.53 m (P12), whereas the channel width ranged from 0.60 (P7) to 7.78 m (P14; Table 3). The predominant substrate in the sites sampled was sand, except in P4, P13, P19 (gravel) and P11 (rocky outcrops; Table 3). The predominant aquatic habitat type was lotic except in stretch P9. Upstream site P17 was located in a reservoir.
Sampling protocols
Sixteen environmental variables were measured in each site. Six variables were associated with physical habitat, six with water quality and four with the riparian zone (Table 4).
Riverbank substrate, riverbank slope, aquatic habitat, type of riparian vegetation cover and percentage of riparian vegetation cover were visually characterized at each transect (along both riverbanks) along with luminosity (photometer; Polaris), stream channel width (measuring tape), stream channel depth (graduated rope) and water velocity (flowmeter; General Oceanic 2030). At the initial, middle and final transects of each site, organic matter samples of the stream channel bed and water were collected to determine algae biomass and to measure the physical and chemical variables.
Organic matter was collected using a Surber sampler (30 × 30 cm). In the laboratory, the samples were dried at 100 °C for 24 hours and weighed (SC2020 – Ohaus; 0.001 g)87.
Alpha chlorophyll concentration was used as a reliable and common proxy for the total phytoplankton biomass88, which may vary according to the degree of shading caused by riparian forests in headwater streams19. In the field, 25 L of water was filtered directly from the stream using a plankton net (mesh 1 μm) and a water pump (P835; Stihl). The product of the filtering process was placed in a 600 ml opaque bottle containing 1 ml of saturated magnesium carbonate. In the laboratory, the samples were filtered (cellulose ester membrane; porosity 0.45 μm) and quantified by spectrophotometry (spectrophotometer; Varian-Cary-50 CONC)89. The a, b and c chlorophyll concentrations were calculated following the Jeffrey and Humphrey equation90.
Water turbidity (turbidimeter; LaMotte 2020), temperature and conductivity (thermometer/conductivity meter WTW 3015i) and dissolved oxygen (DO-Lutron 5510) were measured at ~20 cm depth. The water turbidity, temperature, conductivity, dissolved oxygen and water velocity were measured at ~20 cm depth, whereas luminosity and air temperature were measured at ~20 cm above the water surface.
Fish were collected by shore electrofishing (electrofisher DC, 100–600 V plugged into a 220 V electric generator) modified from91; that is, the site’s length was 100 m and traversed only one time instead of being 50–80 m in length and traversed three times. Both modifications were performed based on the results of92, taking into account the logistics of the electrofishing gear used and displacement difficulties that occur along Cerrado streams because of physical conditions (e.g., trunks and steep stream bank). Four people collected samples for one hour in each site. The collected fish were placed in plastic bags, euthanized with a saturated clove oil solution and fixed in formalin (10%). All the bags were identified with tags containing the stream and site code. Fish was collected in the dry season when captures are more efficient because of lower water levels93. Fish sampling, transport and preservation of the sampled specimens were carried out in accordance with the relevant guidelines and regulations of the Sistema de Autorização e Informação em Biodiversidade, Instituto Chico Mendes de Conservação da Biodiversidade, Ministerio do Meio Ambiente (license # 20226 granted to the second author).
Data analysis
The dataset was organized into five matrices. The first matrix was composed of species abundance (the total number of individuals per species). The second consisted of physical habitat variables (frequency values by category or average values): stream channel width and depth, stream channel substrate, aquatic habitat, water velocity and organic matter. The third consisted of water quality variables (average values): turbidity, water temperature, conductivity, pH, dissolved oxygen and chlorophyll concentration. The fourth consisted of variables related to the riparian zone (frequency values by category): riverbank substrate, riverbank slope, type of riparian vegetation cover and percentage of riparian vegetation cover in the channel. The fifth data matrix grouped the main spatial eigenvectors (MENs)94, which constitute a representation of the spatial process resulting from the analyses performed on the spatial data matrix (geographic coordinates) considering a linear distance (Euclidean distance) between sampling points. The MENs represent spatial autocorrelations (Moran’s index) and can be used as a surrogate for the dispersion ability of species94,95. Significant MENs were considered those with Moran’s index values < 0.05. All the procedures to obtain the MENs were performed in SAM macroecology software96.
To determine the influence of the variable groups (physical habitat, water quality, riparian zone and spatial) (environmental variables) on the fish (biotic structure), a variation partitioning analysis was performed. After that, each data matrix was transformed to a similarity matrix using a specific index (Bray-Curtis for fish species abundance and Euclidean distance for all the other data matrices) and nonparametric multidimensional scaling (NMDS) was performed97. Using the resulting NMDS, a correlation (Procrustes analysis98) was performed separately between the fish assemblages and the physical habitat, water quality, riparian zone and spatial groups (9999 permutations99).
To determine the relationship between the fish assemblages and the variable groups (physical habitat, water quality and riparian zone), two multiple linear regressions were performed: the first one was for fish species richness, and the second one was for fish species abundance. A principal component analyses (PCA) was performed separately on each variable’s group (physical habitat, water quality, riparian zone). The significant axes were retained based on the broken stick criteria and used to perform the multiple linear regressions. The PCA axes were used in place of the original variables to avoid multicollinearity.
Finally, redundancy analyses (RDA), which consider the percentage of explained variation (R²) followed by a bootstrap procedure100, were performed to test the interaction between fish and the physical habitat, water quality and riparian zone groups. These analyses were performed only for the data matrices with significant relationships with the fish matrices (abundance and/or richness). All the statistical analyses were performed in R software using the vegan package98.
References
Gorman, O. T. & Karr, J. R. Habitat Structure and Stream Fish Communities. Ecology 59, 507–515 (1978).
Vannote, R. L., Minshall, G. W., Cummins, K. W., Sedell, J. R. & Cushing, C. E. The River Continuum Concept. Can. J. Fish. Aquat. Sci. 37, 130–137 (1980).
Penczak, T. Effects of removal and regeneration of bankside vegetation on fish population dynamics in the Warta River, Poland. Hydrobiologia 303, 207–210, https://doi.org/10.1007/BF00034057 (1995).
Tejerina-Garro, F. L. et al. Effects of natural and anthropogenic environmental changes on riverine fish assemblages: A framework for ecological assessment of rivers. Brazilian Arch. Biol. Technol. 48, 91–108, https://doi.org/10.1590/S1516-89132005000100013 (2005).
Rivaes, R. et al. Reducing river regulation effects on riparian vegetation using flushing flow regimes. Ecol. Eng. 81, 428–438 (2015).
Teresa, F. B. & Romero, R. D. M. Influence of the riparian zone phytophysiognomies on the longitudinal distribution of fishes: evidence from a Brazilian savanna stream. Neotrop. Ichthyol. 8, 163–170, https://doi.org/10.1590/S1679-62252010000100019 (2010).
Mendonça, F. P., Magnusson, W. E. & Zuanon, J. Relationships between Habitat Characteristics and Fish Assemblages in Small Streams of Central Amazonia. Copeia 2005, 751–764 (2005).
Murray, S. & Innes, J. L. Effects of environment on fish species distributions in the Mackenzie River drainage basin of northeastern British Columbia, Canada. Ecol. Freshw. Fish 18, 183–196, https://doi.org/10.1111/j.1600-0633.2008.00336.x (2009).
Warren, D. R., Mineau, M. M., Ward, E. J. & Kraft, C. E. Relating fish biomass to habitat and chemistry in headwater streams of the northeastern United States. Environ. Biol. Fishes 88, 51–62, https://doi.org/10.1007/s10641-010-9617-x (2010).
Naiman, R. J., Décamps, H. & McClain, M. E. Riparia: Ecology, Conservation, and Management of Streamside Communities. (Elsevier Academic Press, 2005).
Uieda, V. S., Uieda, W. & Paulista, U. E. Species composition and spatial distribution of a stream fish assemblage in the east coast of Brazil: comparison of two field study methodologies. Brazilian J. Biol. 61, 377–388, https://doi.org/10.1590/S1519-69842001000300006 (2001).
Melo, A. S. & Froehlich, C. G. Colonization by macroinvertebrates of experimentally disturbed stones in three tropical streams differing in size. Int. Rev. Hydrobiol. 89, 317–325 (2004).
Santos, G. M., Mérona, B. de, Juras, A. A. & Jégu, M. Peixes do Baixo Tocantins: 20 Anos depois da Usina Hidrelétrica de Tucuruí. (Eletronorte, 2004).
Webster, J. R. & Meyer, J. L. Organic Matter Budgets for Streams: A Synthesis. J. North Am. Benthol. Soc. 16, 141–161, https://doi.org/10.2307/1468247 (1997).
Montgomery, D. R. Process domains and the river continuum. J. Am. Water Resour. Assoc. 35, 397–410 (1999).
Lowe-McConnell, R. H. Estudos ecológicos de comunidades de peixes tropicais. (Edusp - Editora da Universidade de São Paulo, 1999).
Castro, R. M. C. & Casatti, L. The fish fauna from a small forest stream of the upper Parana River basin, southeastern Brazil. Ichtyol.Explor. Freshw. 7, 337–352, https://doi.org/10.1179/174329307X173706 (1997).
Pusey, B. J. & Arthington, A. H. Importance of the riparian zone to conservation and management of freshwater fish: a review. Mar. Freshw. Res. 54, 1–16, https://doi.org/10.1071/MF02041 (2003).
Knight, A. W. & Bottorff, R. L. The importance of riparian vegetation to stream ecosystems. in California Riparian Systems: Ecology, Conservation, and Production Management (eds Warner, R. E. & Hendrix, K. M.) 160–167 (University of California Press, 1984).
Grubaugh, J. W., Wallace, J. B. & Houston, E. S. Longitudinal changes of macroinvertebrate communities along an Appalachian stream continuum. Can. J. Fish. Aquat. Sci. 53, 896–909 (1996).
Braccia, A. & Voshell, R. Benthic Macroinvertebrate Fauna in Small Streams Used by Cattle in the Blue Ridge Mountains, Virginia. Northeast. Nat. 13, 269–286 (2006).
Diniz-Filho, J. A. F., Bini, L. M. & Hawkins, B. A. Spatial autocorrelation and red herrings in geographical ecology. Glob. Ecol. Biogeogr. 12, 53–64 (2003).
Dormann, F. C. et al. Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography (Cop.). 30, 609–628 (2007).
Kühn, I. Incorporating spatial autocorrelation may invert observed patterns. Divers. Distrib. 13, 66–69, https://doi.org/10.1111/j.1472-4642.2006.00293.x (2007).
Bini, L. M. et al. Coefficient shifts in geographical ecology: an empirical evaluation of spatial and non-spatial regression. Ecography (Cop.). 32, 193–204 (2009).
Lv, S. J. & Zhao, M. The dynamic complexity of a three species food chain model. Chaos Solitons & Fractals 37, 1469–1480 (2008).
Legendre, P. & Legendre, L. Canonical analysis. In Developments in Environmental Modelling 20, https://doi.org/10.1016/B978-0-444-53868-0.50011-3 (Elsevier Science B. V., 2012).
Legendre, P. & Legendre, L. Ordination in reduced space. in Developments in Environmental Modelling 20, https://doi.org/10.1016/B978-0-444-53868-0.50009-5 (Elsevier Science B. V. 2012).
Thomas, G. H. et al. Regional variation in the historical components of global avian species richness. Glob. Ecol. Biogeogr. 17, 340–351 (2008).
Das, M. K. et al. Influence of ecological factors on the patterns of fish species richness in tropical Indian rivers. Acta Ichthyol. Piscat. 42, 47–58, https://doi.org/10.3750/AIP2011.42.1.06 (2012).
Davies, P. M., Bunn, S. E. & Hamilton, S. K. Primary Production in Tropical Streams and Rivers. In Tropical Stream Ecology (ed. Dudgeon, D.) 23–42, https://doi.org/10.1016/B978-012088449-0.50004-2 (Elsevier Inc., 2008).
Osborne, L. L. & Wiley, M. J. Influence of Tributary Spatial Position on the Structure of Warmwater Fish Communities. Can. J. Fish. Aquat. Sci. 49, 671–681, https://doi.org/10.1139/f92-076 (1992).
Yan, Y. Z., Xiang, X., Chu, L., Zhan, Y. & Fu, C. Influences of local habitat and stream spatial position on fish assemblages in a dammed watershed, the Qingyi Stream, China. Ecol. Freshw. Fish 20, 199–208, https://doi.org/10.1111/j.1600-0633.2010.00478.x (2011).
Dias, M. S. et al. Global imprint of historical connectivity on freshwater fish biodiversity. Ecol. Lett. 17, 1130–1140, https://doi.org/10.1111/ele.12319 (2014).
Dong, X., Muneepeerakul, R., Olden, J. D. & Lytle, D. A. The effect of spatial configuration of habitat capacity on β diversity. Ecosphere 6, 220 (2015).
Rosindell, J., Hubbell, S. P., He, F., Harmon, L. J. & Etienne, R. S. The case for ecological neutral theory. Trends in Ecology and Evolution 27, 203–208 (2012).
Rodriguez-Iturbe, I., Muneepeerakul, R., Bertuzzo, E., Levin, S. A. & Rinaldo, A. River networks as ecological corridors: A complex systems perspective for integrating hydrologic, geomorphologic, and ecologic dynamics. Water Resour. Res. 45, 1–22, https://doi.org/10.1029/2008WR007124 (2009).
Hanski, I., Foley, P. & Hassell, M. Random walks in a metapopulation: How much density dependence is necessary for long-term persistence? J. Anim. Ecol. 65, 274–282 (1996).
Olden, J. D., Poff, N. L. & Bestgen, K. R. Life-history strategies predict fish invasions and extirpations in the Colorado River Basin. Ecol. Monogr. 76, 25–40 (2006).
Cottenie, K. & De Meester, L. Metacommunity structure: Synergy of biotic interactions as selective agents and dispersal as fuel. Ecology 85, 114–119 (2004).
Thompson, R. & Townsend, C. A truce with neutral theory: Local deterministic factors, species traits and dispersal limitation together determine patterns of diversity in stream invertebrates. J. Anim. Ecol. 75, 476–484 (2006).
Macedo, D. R. et al. The relative influence of catchment and site variables on fish and macroinvertebrate richness in cerrado biome streams. Landsc. Ecol. 29, 1001–1016, https://doi.org/10.1007/s10980-014-0036-9 (2014).
Leal, C. G. et al. Is environmental legislation conserving tropical stream faunas? A large-scale assessment of local, riparian and catchment-scale influences on Amazonian fish. J. Appl. Ecol. 55, 1312–1326, https://doi.org/10.1111/1365-2664.13028 (2018).
Leitão, R. P. et al. Disentangling the pathways of land use impacts on the functional structure of fish assemblages in Amazon streams. Ecography (Cop.). 41, 219–232, https://doi.org/10.1111/ecog.02845 (2018).
de Carvalho, D. R. et al. A fish-based multimetric index for Brazilian savanna streams. Ecol. Indic. 77, 386–396, https://doi.org/10.1016/j.ecolind.2017.02.032 (2017).
Bini, L. M. et al. Coefficient shifts in geographical ecology: an empirical evaluation of spatial and non-spatial regression. Ecography (Cop.) 32, 193–204 (2009).
Brea, M. & Zucol, A. F. Paraná-Paraguay Basin: A review of the geology and the plant fossil record. In Historical biogeography of Neotropical freshwater fishes. (eds. JS, A. & RE, R.) 69–87 (University of California Press, 2011).
Jackson, C. R., Churchill, P. F. & Roden, E. E. Successional changes in bacterial assemblage structure during epilithic biofilm development. Ecology 82, 555–566 (2001).
Oberdorff, T., Hugueny, B. & Vigneron, T. Is assemblage variability related to environmental variability? An answer for riverine fish. OIKOS 93, 419–428, https://doi.org/10.1034/j.1600-0706.2001.930307.x (2001).
Väliverronen, E. Biodiversity and the power of metaphor in environmental discourse. Sci. Stud. (St. Bonaventure). 11, 19–34 (1998).
Hoeinghaus, D. J., Winemiller, K. O. & Birnbaum, J. S. Local and regional determinants of stream fish assemblage structure: inferences based on taxonomic vs. functional groups. J. Biogeogr. 34, 324–338 (2007).
Fernandes, I. M., Henriques-Silva, R., Penha, J., Zuanon, J. & Peres-Neto, P. R. Spatiotemporal dynamics in a seasonal metacommunity structure is predictable: The case of floodplain-fish communities. Ecography (Cop.). 37, 464–475, https://doi.org/10.1111/j.1600-0587.2013.00527.x (2014).
Monroe, J. B., Baxter, C. V., Olden, J. D. & Angermeier, P. L. Freshwaters in the Public Eye: Understanding the Role of Images and Media in Aquatic Conservation. Fisheries 34, 581–585 (2009).
Casatti, L., Langeani, F. & Ferreira, C. Effects of Physical Habitat Degradation on the Stream Fish Assemblage Structure in a Pasture Region. Environ. Manage. 38, 974–982, https://doi.org/10.1007/s00267-005-0212-4 (2006).
Pinto, B., Araujo, F. & Hughes, R. Effects of Landscape and Riparian Condition on a Fish Index of Biotic Integrity in a Large Southeastern Brazil River. Hydrobiologia 556, 69–83, https://doi.org/10.1007/s10750-005-9009-y (2006).
Rocha, F. C., Casatti, L., Carvalho, F. R. & Silva, A. M. Fish assemblages in stream stretches occupied by cattail (Typhaceae, Angiospermae) stands in Southeast Brazil. Neotropical Ichthyology 7, 241–250, https://doi.org/10.1590/S1679-62252009000200016 (2009).
Alexandre, C. V., Esteves, K. E. & de Moura e Mello, M. A. M. M. Analysis of fish communities along a rural-urban gradient in a neotropical stream (Piracicaba River Basin, São Paulo, Brazil). Hydrobiologia 641, 97–114, https://doi.org/10.1007/s10750-009-0060-y (2010).
Silva, A. G. & Martinez, C. B. R. Morphological changes in the kidney of a fish living in an urban stream. Environ. Toxicol. Pharmacol. 23, 185–192, https://doi.org/10.1016/j.etap.2006.08.009 (2007).
Ferreira, A., Hahn, N. S. & Delariva., R. L. Ecologia alimentar de Piabina argentea (Teleostei, Tetragonopterinae) nas fases de pré e pós-represamento do rio Corumbá, GO. Acta Limnologica Brasiliensia 14, 43–52 (2002).
Takahashi, E. L. H., Rosa, F. R. T., Langeani, F. & Nakaghi, L. S. O. Spatial and seasonal patterns in fish assemblage in Córrego Rico, upper Paraná River basin. Neotrop. Ichthyol. 11, 143–152, https://doi.org/10.1590/S1679-62252013000100017 (2013).
Schulz, U. H. & Martins-Junior, H. Astyanax fasciatus as bioindicator of water pollution of Rio dos Sinos, RS, Brazil. Braz. J. Biol. 61, 615–622, https://doi.org/10.1590/S1519-69842001000400010 (2001).
Melo, F. C. S. Ade, Maldonado, I. RdosS. C., Benjamin, LdosA. & Matta, S. L. Pda Biologia reprodutiva de fêmeas de Lambari-prata (Astyanax scabripinnis) (Characidae, Tetragonopterinae) em tanques de piscicultura. Rev Ceres 52, 811–829 (2005).
Carvalho, F. M. V., De Marco, P. & Ferreira, L. G. The Cerrado into-pieces: Habitat fragmentation as a function of landscape use in the savannas of central Brazil. Biol. Conserv. 142, 1392–1403 (2009).
Bischoff, R. J., Gould, J. L. & Rubenstein, D. I. Tail size and female choice in the guppy (Poecilia reticulata). Behav. Ecol. Sociobiol. 17, 253–255, https://doi.org/10.1007/BF00300143 (1985).
Dzikowski, R., Hulata, G., Karplus, I. & Harpaz, S. Effect of temperature and dietary L-carnitine supplementation on reproductive performance of female guppy (Poecilia reticulata). Aquaculture 199, 323–332, https://doi.org/10.1016/S0044-8486(01)00561-0 (2001).
Weetman, D., Atkinson, D. & Chubb, J. C. Effects of temperature on anti-predator behaviour in the guppy, Poecilia reticulata. Anim. Behav. 55, 1361–1372, https://doi.org/10.1006/anbe.1997.0666 (1998).
Kramer, D. L. & Mehegan, J. P. Aquatic surface respiration, an adaptive response to hypoxia in the guppy, Poecilia reticulata (Pisces, Poeciliidae). Environ. Biol. Fishes 6, 299–313, https://doi.org/10.1007/BF00005759 (1981).
Fialho, A. P., Oliveira, L. G., Tejerina-Garro, F. L. & De Mérona, B. Fish-habitat relationship in a tropical river under anthropogenic influences. Hydrobiologia 598, 315–324, https://doi.org/10.1007/s10750-007-9165-3 (2008).
MacKenzie, R. A. Impacts of riparian forest removal on Palauan streams. Biotropica 40, 666–675, https://doi.org/10.1111/j.1744-7429.2008.00433.x (2008).
Etheridge, E. C., Harrod, C., Bean, C. W. & Adams, C. E. Has habitat heterogeneity promoted phenotypic and ecological sub-structuring among a Coregonus lavaretus population in a large Scottish lake? J. Fish Biol. 77, 2391–2404, https://doi.org/10.1111/j.1095-8649.2010.02827.x (2010).
Lowrance, R. et al. Riparian Forests as Nutrient Filters in Agricultural Watersheds. Bioscience 34, 374–377, https://doi.org/10.2307/1309729 (1984).
Detenbeck, N. E., Devore, P. W., Niemi, G. J. & Lima, A. Recovery of temperate-stream fish communities from disturbance - a review of case studies and synthesis of theory. Environ. Manage. 16, 33–53 (1992).
Armour, C. L., Duff, D. A. & Elmore, W. The effects of livestock grazing on riparian and stream ecosystems. Am. Fish. Soc. 16, 7–12, 10.1577/1548-8446(1994)019<0009:TEOLGO>2.0.CO;2 (1991).
Allan, J. D. & Flecker, A. S. Biodiversity Conservation in Running Waters. Bioscience 43, 32–43, https://doi.org/10.2307/1312104 (1993).
Matlack, G. R. Vegetation dynamics of forest edge-trends in space and sucession time. J. Ecol. 82, 113–123 (1994).
Cummins, K. W. et al. Organic matter budgets for stream ecosystems: Problems in their evaluation. in Stream ecology: Application and Testing of General Ecological Theory. (eds. Barnes, J. R. & Minshall, G. W.) 299–353 (Plenum Press, 1983).
Sazima, M. & Sazima, I. Oil gathering bees visit flowers of eglandular morphs of the oil-producing Malpighiaceae. Bot. Acta 102, 106–111 (1989).
Garavello, J. C. & Britski, H. A. Family Anostomidae (Headstanders). in Check List of the Freshwater Fishes of South and Central America. (eds. Reis, R. E., Kullander, S. O. & Ferraris Jr., C. J.) 71–84 (Editora da Pontifícia Universidade Católica do Rio Grande do Sul, 2003).
Webster, J. R. & Benfield, E. F. Vascular plant breakdown in freshwater ecosystems. Annu. Rev. Ecol. Syst. 17, 567–594, https://doi.org/10.1146/annurev.es.17.110186.003031 (1986).
Valério, S. B., Súarez, Y. R., Felipe, T. R. A., Tondato, K. K. & Ximenes, L. Q. L. Organization patterns of headwater-stream fish communities in the Upper Paraguay–Paraná basins. Hydrobiologia 583, 241–250, https://doi.org/10.1007/s10750-006-0533-1 (2007).
Sazima, I. Similarities in feeding behaviour between some marine and freshwater fishes in two tropical communities. J. Fish Biol. 29, 53–65, https://doi.org/10.1111/j.1095-8649.1986.tb04926.x (1986).
de Araujo, R. B. & Garutti, V. Ecology of a stream from upper Paraná River basin inhabited by Aspidoras fuscoguttatus Nijssen & Isbrücker, 1976 (Siluriformes, Callichthyidae). Braz. J. Biol. 63, 363–372 (2003).
Brandão-Gonçalves, L., Lima-Júnior, S. E. & Suarez, Y. R. Hábitos alimentares de Bryconamericus stramineus Eigenmann, 1908 (Characidae), em diferentes riachos da sub-bacia do Rio Guiraí, Mato Grosso do Sul, Brasil. Biota Neotrop, https://doi.org/10.1590/S1676-06032009000100016 (2009).
Vasconcelos, L. P., Súarez, Y. R. & Lima-Junior, S. E. Population aspects of Bryconamericus stramineus in streams of the upper Paraná River basin, Brazil. Biota Neotrop. 11, 55–62, https://doi.org/10.1590/S1676-06032011000200006 (2011).
Felipe, T. R. A. & Súarez, Y. R. Caracterização e influência dos fatores ambientais nas assembléias de peixes de riachos em duas microbacias urbanas, Alto Rio Paraná. Biota Neotrop. 10, 143–151, https://doi.org/10.1590/S1676-06032010000200018 (2010).
Casatti, L. et al. From forests to cattail: how does the riparian zone influence stream fish? Neotrop. Ichthyol. 10, 205–214, https://doi.org/10.1590/S1679-62252012000100020 (2012).
Melo, T. L. de. Avaliação espacial das variáveis ambientais e da estrutura trófica da ictiofauna de tributários da bacia Tocantins-Araguaia, Brasil Central. Centro de Ciências Biológicas e da Saúde Ph D, (Universidade Federal de São Carlos, 2011).
Gregor, J. & Maršálek, B. Freshwater phytoplankton quantification by chlorophyll a: a comparative study of in vitro, in vivo and in situ methods. Water Res. 38, 517–522, https://doi.org/10.1016/j.watres.2003.10.033 (2004).
Marker, A. F. H., Nush, E. A., Rai, H. & Riemann, B. The measurement of photosynthetic pigments in freshwaters and standardization of methods: conclusions and recommendations. Arch. für Hydrobiol. Ergebnisse der Limnol. 14, 91–106 (1980).
Jeffrey, S. W. T. & Humphrey, G. F. New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem Physiol Pflanz BPP 167, 191–194, https://doi.org/10.1016/s0015-3796(17)30778-3 (1975).
Manzzoni, R. & Fenerich-Verani, E. P. N. C. A pesca elétrica como técnica de amostragem de populações e comunidades de peixes em rios costeiros do sudeste do Brasil. Rev. Bras. Biol. 6, 205–216 (2000).
Reynolds, L., Herlihy, A. T., Kaufmann, P. R., Gregory, S. V. & Hughes, R. M. Electrofishing Effort Requirements for Assessing Species Richness and Biotic Integrity in Western Oregon Streams. North Am. J. Fish. Manag. 23, 450–461 (2003).
Pease, A. A., González-Díaz, A. A., Rodiles-Hernández, R. & Winemiller, K. O. Functional diversity and trait–environment relationships of stream fish assemblages in a large tropical catchment. Freshw. Biol. 57, 1060–1075, https://doi.org/10.1111/j.1365-2427.2012.02768.x (2012).
Griffith, D. A. & Peres-Neto, P. R. Spatial Modeling in Ecology: The Flexibility of Eigenfunction Spatial Analyses. Ecology 87, 2603–2613, 10.1890/0012-9658(2006)87[2603:SMIETF]2.0.CO;2 (2006).
Dormann, C. F. et al. Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography (Cop.). 30, 609–628, https://doi.org/10.1111/j.2007.0906-7590.05171.x (2007).
Rangel, T. F., Diniz-Filho, J. A. F. & Bini, L. M. SAM: A comprehensive application for Spatial Analysis in Macroecology. Ecography (Cop.). 33, 46–50, https://doi.org/10.1111/j.1600-0587.2009.06299.x (2010).
Clarke, K. R. & Ainsworth, M. A method of linking multivariate community structure to environmental variables. Mar. Ecol. Prog. Ser. 92, 205–219, https://doi.org/10.3354/meps092205 (1993).
Oksanen, J. et al. Vegan: Community Ecology Package. https://cran.r-project.org, https://github.com/vegandevs/vegan (2019).
Legendre, P. & Legendre, L. Numerical Ecology. Development in Environmental Modelling, 20, https://doi.org/10.1017/CBO9781107415324.004 (Elsevier Science B. V., 1998).
Peres-Neto, P. R., Legendre, P., Dray, S. & Borcard, D. Variation partitioning of species data matrices: Estimation and comparison of fractions. Ecology 87, 2614–2625, 10.1890/0012-9658(2006)87[2614:VPOSDM]2.0.CO;2 (2006).
Acknowledgements
We thank the Centro de Biologia Aquática – Pontifice Universidade Catolica de Goias - PUC Goiás team, especially Mr. Waldeir Francisco de Menezes for collaborating with field data collection; the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) by the scholarship granted to TBV and to the Fundação de Amparo à Pesquisa do Estado de Goiás (FAPEG) for funds granted to FLTG that supported data collection.
Author information
Authors and Affiliations
Contributions
T.B.V. performed data analysis and wrote the original draft. F.L.T.G. supervised and reviewed the draft’s elaboration.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vieira, T.B., Tejerina-Garro, F.L. Relationships Between Environmental Conditions And Fish Assemblages In Tropical Savanna Headwater Streams. Sci Rep 10, 2174 (2020). https://doi.org/10.1038/s41598-020-59207-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-59207-9
- Springer Nature Limited