Abstract
Age related macular degeneration (AMD) is a common retina-related disease leading to blindness. Little is known on the origin of the disease, but it is well documented that oxidative stress generated in the retinal pigment epithelium and choroid neovascularization are closely involved. The study of circulating miRNAs is opening new possibilities in terms of diagnosis and therapeutics. miRNAs can travel associated to lipoproteins or inside small Extracellular Vesicles (sEVs). A number of reports indicate a significant deregulation of circulating miRNAs in AMD and experimental approaches, but it is unclear whether sEVs present a significant miRNA cargo. The present work studies miRNA expression changes in sEVs released from ARPE-19 cells under oxidative conditions (i.e. hydrogen peroxide, H2O2). H2O2 increased sEVs release from ARPE-19 cells. Moreover, 218 miRNAs could be detected in control and H2O2 induced-sEVs. Interestingly, only two of them (hsa-miR-302a and hsa-miR-122) were significantly under-expressed in H2O2-induced sEVs. Results herein suggest that the down regulation of miRNAs 302a and 122 might be related with previous studies showing sEVs-induced neovascularization after oxidative challenge in ARPE-19 cells.
Similar content being viewed by others
Introduction
microRNA (miRNA) are small non-coding RNA sequences (21-25 nucleotides) able to regulate the expression of one or more mRNAs1,2. miRNAs regulate protein translation by targeting their complementary mRNAs and by repressing translation or degrading a target mRNA. Nowadays, miRNA research has been rocketed from 214 publications in 2014 to 11,610 in 2018 (source: Pubmed). Many clinical fields are now focused on the study of miRNA for diagnosis or treatment purposes3,4. Moreover, miRNAs play an important role in different cellular processes such as angiogenesis5,6, oxidative stress7, and immune responses8.
Most of the cells can release extracellular vesicles (EVs), which may interact with both neighbouring or distant target cells9,10. EVs typically include genetic material and proteins, making these vesicles key in cell to cell communication11. Among these EVs, are the small EVs (sEVs; <100 nm diameter)12, which were observed to carry miRNAs13. Cargo of sEVs depends on the cellular origin and the homeostatic state14.
Recent studies analysed miRNA expression in biological samples from patients with age related macular degeneration (AMD), in order to identify those miRNAs related to the pathophysiology and progression of the disease. Among these, miR-9, miR-23a, miR-27a, miR-34a, miR-146a, miR-155 have been proposed as potential candidates15,16,17,18,19. The “wet” form of AMD is characterized by neovascularization20. The retinal pigment epithelium (RPE) plays a pivotal role between the photoreceptor cell layer and the choroid, the vascular network surrounding the eye. This interaction is critical for retinal homeostasis21. Ren and collaborators analysed circulating miRNAs from AMD patients and proposed miR-27a-3p, miR-29b-3p, and miR-195-5p as candidate biomarkers for AMD diagnosis19.
One of the most challenging issues in ophthalmological sciences is to identify early markers for AMD in order to prevent its clinical evolution. To date AMD treatments are limited to diminish the evolution of the disease as anti-VEGF drugs (eg. Bevacizumab)22. The present work studies the oxidative-induced response in RPE cells, in order to find potential miRNAs related to neovascularization within sEVs.
Results
H2O2-induced oxidative stress in ARPE-19 cells
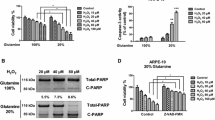
ARPE-19 cells exposed for 24 h to H2O2 significantly increased intracellular reactive oxygen species (ROS) levels when using a concentration of 600 μM (106 ± 1.69) and 800 μM (103.1 ± 0.9892), compared to control (100 ± 0). Nevertheless, 400 μM H2O2, or lower, did not increase ROS levels compared to control (Fig. 1A). In order to assess whether H2O2-induced ROS affected ARPE-19 cell viability, a XTT assay was performed. Cell viability was significantly decreased when using 800 μM H2O2 (95.45 ± 0.9999) but not with lower concentrations (Fig. 1B). Moreover, 800 μM H2O2 concentration also increased early apoptosis (3.038 ± 0.1689) with respect to control (1.713 ± 0.4796) (Fig. 1C,D).
Effect of oxidative stress in ARPE-19 cells. Superoxide anions were measured by DHE after 24 h of H2O2 treatment. (A) XTT cell viability as percentage to control. (B) Early apoptotic cells measured using AnnexinV-IP. (C) Flow cytometry histograms: alive cells (annexin V−, PI−), early apoptosis (annexin V+, PI−) and necrotic cells (annexin V+, IP+). (D) Values are expressed as mean ± SEM (n = 3). Statistically significant differences were set at *p < 0.05 and **p < 0.01.
H2O2 increased sEVs release in ARPE-19
Matching previous results on cell viability (see Fig. 1), 600 µM H2O2 was used to stress ARPE-19 cells without killing them (sub-lethal concentration). After 24 h of 600 µM H2O2 exposure, ARPE-19 cells significantly increased the number of EVs released to the medium (Fig. 2). More precisely, a 40% increase in the release of EVs was observed after 600 µM H2O2 exposure, compared to controls. Number and size of EVs were studied using a nanoparticle tracking system (NanoSight) (Fig. 2A). Besides, EVs were observed using transmission electron microscopy (TEM), which showed that, according to size and morphology, most of the EVs observed can be classified as sEVs (Fig. 2B,C).
miRNA Expression in ARPE-19 cells and sEVs
In the present study, 384 miRNAs were analysed, and two different miRNA clusters could be set after hierarchical clustering (Fig. 3). The same proceeding was performed with the extracellular medium to collect sEVs and miRNAs providing also two clusters (Fig. 4).
Heatmap of miRNA expression profile from array assay of ARPE-19 cells. After 24 h of 600 μM H2O2, the miRNA expression profile was analyzed, using an array for microRNA. Hierarchical unsupervised clustering was performed with the expressed miRNAs in ARPE-19 cells. Each column represents an individual cell sample. Orange columns are ARPE-19 cells treat with 600 μM H2O2 and green columns ARPE-19 cell controls. Control sample (n = 4) and 600 μM H2O2 sample (n = 4). The overexpressed miRNAs are shown in green and under-expressed miRNAs in red. Expression values are calculated with respect to the reference genes of the array assay.
Heatmap of miRNA expression profile from array assay of the sEVs released by ARPE-19 cells. After 24 h of 600 μM H2O2, the miRNA expression profile of sEVs was analyzed, using an array of microRNA. Hierarchical unsupervised clustering was performed with the expressed miRNAs in ARPE-19-released sEVs. Each column represents an individual cell sample. Orange columns are sEVs released by ARPE-19 cells treat with 600 μM H2O2 and green columns are sEVs released by ARPE-19 control cells. Control sample (n = 4) and 600 μM H2O2 sample (n = 4). Overexpressed miRNAs (green) and under-expressed miRNAs (red). The expression values are calculated with respect to the reference genes of the array.
miRNA Expression in ARPE-19 cells (Cell miRNA)
As shown in Fig. 3, there is a significant miRNA repression after H2O2 treatment when compared with control ARPE-19 cells. As a result, 306 out of 384 Cell miRNAs were detected by the array. From those, 59 Cell miRNAs were significantly expressed in ARPE-19 cells (p < 0.05) (Table 1). Moreover, seven out of 59 Cell miRNAs were significantly under-expressed in H2O2-treated cells (fold change, FC > 1) than in control conditions. Different detected miRNAs are shown in Fig. 5A,C. It is noteworthy to underline that hsa-miR-205 and hsa-miR-302c presented a dramatic decrease in treated cells when compared to others (FC > 2).
miRNAs significantly regulated in ARPE-19 cells. Cell miRNAs (A) and sEV miRNA (B) up-regulated in ARPE-19 cells with fold change (FC) > 1. Volcano plot of miRNA expression profile. The X axis represent the miRNA expression level and the Y axis represents p-value (T-student Test). The red lines indicate FC ± 1 P < 0.05 (C,D). Relative expression of the selected Cell miRNAs (E) and sEV miRNAs (F) with FC > 1 (control vs 600 μM H2O2). miRNA expression was quantified by qRT-PCR and calculate using the 2−ΔΔCt. The p-value was calculated by Two-way ANOVA. Results are expressed as mean ± SEM of n = 3–4. *p-value < 0.05 vs Control and ***p-value < 0.001.
miRNA Expression in ARPE-19 sEVs (sEV miRNA)
As above-mentioned, ARPE-19 cells released a significantly higher number of sEVs after 600 µM H2O2 exposure than control cells. In contrast, these H2O2 induced sEVs showed a significant lower miRNA expression compared to control. More concretely, 218 out of 384 sEV miRNAs were detected by the array. However, only 2 out of 218 sEV miRNAs were significantly lower in sEVs released from treated cells (p < 0.05) hsa-miR-302a (FC = 1.159) and hsa-miR-122 (FC = 1.576) than in control sEVs (Fig. 5B,D).
Microarray validation
Among the miRNAs differently expressed in ARPE-19 cells, seven out of 59 presented a significant fold-change. In order to validate this finding, qRT-PCR was performed by using independent RNA samples. We selected three out of seven significant miRNAs (those with the highest differences observed): hsa-miR-205-5p, hsa-miR-521, and hsa-miR-302c; plus a miRNA that was unchanged: hsa-miR-130b, as a control. The outcome confirmed the results obtained, hsa-miR-205-5p, hsa-miR-521 and hsa-miR-302c were down-regulated by 600 μM H2O2 compared to control (Fig. 5E).
The same procedure, qRT-PCR, was performed using the two sEV miRNAs which expression had changed: hsa-miR-302a and hsa-miR-122. As expected, miRNAs were under-expressed in sEVs released from ARPE-19 cells treated with 600 μM H2O2 (Fig. 5F).
Pathway analysis and prediction of miRNA targets regulated by oxidative stress
Subsequently, the role of the under-expressed miRNAs in treated ARPE-19 cells and in sEVs from treated cells (seven in cells and two in sEVs), was analysed. For that reason, two independent ¨in silico¨ analysis were performed in order to determine potential biological processes related to oxidative stress induction. The analysis of the KEGG pathway, regulated by Let-7a, miR-518d-3p, miR-521, miR-338-5p, miR-548b-5p, miR-205, and miR-302c, shows a large number of pathways involving these miRNAs. Among them, cell cycle, adherent junction, p53 signalling pathway, and HIF-1 signalling pathway are the most relevant (Fig. 6A). Both sEV miRNAs, miR-302a and miR-122, are involved in different pathways, such as TGF-beta signalling pathway, FoxO signalling pathway, and cell cycle (Fig. 7A).
Potential targets of miRNAs
In order to identify biological functions of the validated miRNAs and to select their putative targets, two programs were handled: DIANA TOOLS mirPath and Target Scan Human. The outcome on sEV miRNA and Cell miRNA related pathways are not completely equivalent. Most of the significantly involved KEGG are related to cancer or cell cycle pathways (Figs. 6B and 7B).
Discussion
The study of circulating miRNAs on AMD has been addressed to find biological markers which might help implementing an early diagnosis for the disease, or to find new therapeutic strategies18. Although neovascular processes and oxidative stress are well known characters involved on AMD, the origin of the disease is far from being completely understood. Several studies were focused in identifying neovascularization-related miRNAs or oxidative stress-related ones23,24,25,26. On this line, the use of RPE cell cultures, as ARPE-19 under oxidative challenges (eg. H2O2, rotenone or EtOH), results useful to study the RPE response to oxidative stimuli27,28,29.
As mentioned earlier, 600 µM was the highest H2O2 concentration used which increased ROS levels without generating early apoptosis (800 µM H2O2 exposure significantly increased early apoptosis). Other authors have used different H2O2 ranges (200-600 µM) and some differences can be found in terms of cell viability (XTT or MTT), apoptosis and ROS production7,27,30. Plausibly, the differences on time exposure (12–24 h) or cell confluence level could explain these discrepancies.
Fitting with previous data, pro-oxidant challenge resulted on significant high sEVs release from ARPE-19 cells. It is well documented that EtOH or high glucose conditions resulted in the same response increasing sEVs release from ARPE-19 cells28,31.
Extracellular or circulating miRNAs can be included on sEVs, associated to lipoproteins or proteins13. In this respect, we have just focused on miRNAs contained as sEVs cargo (sEV miRNA). In spite of the fact that in sEVs only 218 out of 384 miRNAs could be detected, only two sEV miRNAs were significantly high in control released sEVs than in sEVs from treated cells. The hsa-miR-302a and hsa-miR-122 have already been seen in sEVs confirming our results32,33,34,35.
When comparing the two sEV miRNAs, hsa-miR-302a and hsa-miR-122, to those miRNAs on ARPE-19 cells (Cell miRNA), no matches could be found. Surprisingly, the sEV miRNA hsa-miR-302a and the Cell miRNA hsa-miR-302c belong to the miR 302/367 La-related protein 7 (LARP7) intragenic cluster. This includes hsa-miR-367, hsa-miR-302d, hsa-miR-302a, hsa-miR-302c, and hsa-miR-302b. This cluster is involved in several processes coordinating proliferation, differentiation, pluripotency maintenance, and cell reprogramming36. Moreover, the cluster, regulates TGF pathway, PI3K–AKT and BMP cell signaling37,38,39. Additionally, hsa-miR-302a acts as a tumor suppressor40 and repressor of cell division, and more concretely, VEGFA is one of the direct targets for this miRNA41. In addition, hsa-miR-122 has been related to VEGFC42. Interestingly, low hsa-miR-302a/hsa-miR-122 expression levels are inversely related to VEGF levels in hepatocellular carcinoma, promoting vascular changes43,44. In addition, miR-122 seems to have a role against oxidative stress, since the use of pre-miR-122 protects from H2O2-induced oxidative stress45, targeting the mitochondrial ribosomal protein S1146.
Recent data from other groups indicate how diverse oxidative insults -EtOH or high glucose, and now H2O2 - lead to increased sEVs release from ARPE-19 cells28,31. Furthermore, those oxidative-induced sEVs were capable of promoting neovascular processes in endothelial cell cultures, whereas control-released sEVs inhibited this phenomenon28,31. In view of these findings, we hypothesize that sEVs hsa-miR-122 and hsa-miR-302a decreased levels could play a role on angiogenesis involving oxidative stress related pathways.
Several reports on AMD or experimental AMD, have reported significant changes on miRNAs (see Table 2). Among them, hsa-miR-23a is present in five reports7,16,17,42,47. This miRNA seems to be downregulated in both AMD patients and H2O2-treated ARPE-19 cells. In fact, H2O2 induced-apoptotic cell death is significantly observed in ARPE-19 cells after hsa-miR-23a inhibition7. Fitting with this, we found significant expression differences on hsa-miR-23a after H2O2 exposure (FC = 0.79). Table 2 summarizes those Cell miRNAs matching with previous reports on AMD or AMD experimental models. Besides, there are reports that locate the sEVS-miRNAs in eye tissue. MiR-302 expression was observed already in RPE cells by Li and collaborators, playing an important role in the RPE differentiation48. Other authors observed a miR-122 differential expression on canine retinas49, in aqueous humor50 and are related in fact with diabetic retinopathy51. In agreement with others, up/down regulated miRNAs are related to several and different cell signaling pathways. After seeing the results in both sEV miRNA and Cell miRNAs related pathways, cancer related pathways are commonly involved in many of the deregulated miRNAs. More research must be performed on these generic pathways to determine the concrete role of these cell signaling routes.
As a conclusion, H2O2 significantly increased sEVs release from ARPE-19 cells compared to control cells. Paradoxically, the miRNA sEVs cargo (hsa-miR-302a and hsa-miR-122) resulted in significantly lower in H2O2-induced sEVs compared to control. Since hsa-miR-302a and hsa-miR-122 regulates vasculogenic targets, these results support those on ARPE-19 cells indicating that oxidative-induced sEVs promote angiogenesis28,31.
Material and Methods
Cell culture
Arising retinal pigment epithelium (ARPE-19) human cell line was obtained from American Type Culture Collection (ATCC, Barcelona, Spain) at passage 19. ARPE-19 cells were cultured in Dulbecco’s modified Eagle’s DMEM/F12 (Invitrogen, Carlsbad, CA, USA), as previously described21. Cells were used until passage 30. Cells were cultured to 80–90% confluence at a starting density of 1 × 106 cells/cm2 in different plates depending on the technique. After 2 days, the cells were treated for 24 h with 600 µM H2O2 (Scharlau, Senmenat, Spain), using filtered media with 1% of Fetal Bovine Serum, exosome-depleted (FBS; Thermo Fisher Scientific, Gibco, USA). Cells and supernatant were collected and preserved for future experiments.
Determination of intracellular ROS
Intracellular ROS levels were measured using dihydroethidium, (DHE; Thermo Fisher Scientific, Waltham, MA, USA), which is a superoxide indicator. This molecule has a blue fluorescence, but, when oxidized to ethidium, it stains DNA in red. ARPE-19 cells were seeded at 6 × 103 cells/well in a 96 well plate. Cells were rinsed with PBS (phosphate-buffered saline) twice and incubated with 5 μM of DHE during 30 min at 37 °C and 5% CO2. ROS levels were measured by a fluorescence multiplate reader (Victor X5; Perkin Elmer) excited at 518 nm and read at 605 nm.
Apoptosis detection
Number of dead cells by apoptosis or necrosis was measured by flow cytometry using the FITC Annexin V Apoptosis detection Kit (Immunostep, Salamanca, Spain) that can discriminate live cells from those in early apoptosis, late apoptosis, or necrosis. A total of 10,000 cells per condition were analysed using the FACS Verse (Beccton Dikinson, New Jersey, USA). Four populations are detected: unmarked Annexin (−)/Propidium Iodide (−) are live cells; double marked Annexin (+)/Propidium Iodide (+) represent apoptotic cells; simple marked Annexin (+)/Propidium Iodide (−) are early apoptotic cells; and simple marked Annexin (−)/Propidium Iodide (+) are necrotic cells.
sEVs isolation and size distribution
sEVs isolation was performed by successive ultracentrifugation as previously reported21. The sEVs pellet was stored at 4 °C until further processing in PBS solution. For microarray assay, sEVs were isolated using ExoQuick-TC (Systems Biosciences, Mountain View, CA, USA) following the manufacturer’s instructions. sEVs identity was confirmed by the nanoparticle tracking system NanoSight NS300 following manufacturer’s protocols (Malvern Instruments, Malvern, UK).
Electron microscopy
sEVs pellets were resuspended in PBS and ultracentrifuged at 120.000 × g for 70 min at 4 °C. After that, approximately 10 µg of the samples were resuspended in PBS on parafilm. The sample was fixed by depositing a drop of 2% Paraformaldehyde on the parafilm and placing the grid (Mesh with Formvar) on top of the drop. Negative staining was performed with 2% uranyl acetate. Photomicrographs were obtained using the transmission electron microscope FEI Tecnai G2 Spirit (FEI Europe, Eindhoven, Netherlands) using a digital camera Morada (Olympus Soft Image Solutions GmbH, Münster, Germany). EVs were identified under the microscope solely based on size and morphology.
RNA isolation and miRNA expression analysis
To perform microarray analysis, ARPE-19 cells from 4 separate cultures were exposed to control and H2O2 600 µM treatment for 24 h. Total RNA was extracted using SeraMir Kit (System Biosciences, Mountain View, CA, USA) according to manufacturer’s instructions. Therefore, four microarrays were performed for each condition: control, ARPE-19 cells exposed to H2O2 600 µM, sEVs released by control cells and sEVs released by ARPE-19 cells exposed to H2O2 600 µM.
Total RNA quantity and quality (260/280 absorbance ratio) were assessed using NanoDrop 2000 (Thermo Fisher Scientific, Waltham, MA, USA). Total RNA was reversely transcribed (cDNA synthesis) using PeqSTAR 96 Universal Gradient (PeqLAb, Erlangen, Germany) under the following conditions: 60 °C/5 min, RT/2 min, 42 °C/30 min, 95 °C/10 min and 15 °C/hold. Real-time quantitative PCR (qPCR) was performed using 384 well SeraMir Profiler using RT-PCR QuantStudioTM 3 y 4 system (Thermo Fisher Scientific, Waltham, MA, USA) with the appropriate temperature cycles 50 °C/2 min, 95 °C/10 min, 40 ciclos; 95 °C/15 s, 6 °C/1 min. The miRNA expression values were calculated using three endogenous controls: RNU43, RNU1Q and RNU6. The expression was calculated according to the 2−∆∆Ct method.
Array analysis
We analyzed miRNA expression differences between ARPE-19 control cells and ARPE-19 treated with H2O2. Moreover, differences in miRNA expression between EVs released by ARPE-19 cells treated and EVs released by ARPE-19 control cells were also studied. Differences were analyzed using a t-test study from genefilter package from R Bioconductor. P-values were adjusted by the Benjamini-Hochberg method. MiRNAs that presented an adjusted p-value < 0.05 were considered statistically significant. Significantly modified miRNAs from different samples were represented in a hierarchical clustering heatmap representation. Heatmaps were performed using heatmap.3 package from R Bioconductor.
Analysis of miRNA target genes
In silico analysis of the pathways in which the miRNAs regulated by H2O2 were involved using DIANA TOOLS mirPath v.3 algorithm (http://snf-515788.vm.okeanos.grnet.gr/). Moreover, we carry out an analyse of the putative miRNAs target using TargetScanHuman (http://www.targetscan.org/vert_72/).
Quantitative real-time PCR validation
Quantitative real time PCR (qRT-PCR) was used to validate the miRNA expression profile of the selected miRNAs in an independent sample set. The RNA was isolated from ARPE-19 cells by miRNeasy Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. 100–300 ng of RNA were retro transcribed using TaqMan MicroRNA Reverse Transcription Kit (Thermo Fisher Scientific, Waltham, MA, USA) using specific TaqMan RT primers and the thermocycler PeqSTAR 96 Universal Gradient (PeqLAB, Erlangen, Germany), the cycles used were 16 °C/30 min, 42 °C/30 min, 85 °C/5 min and 4 °C/infinity. Quantitative real time PCR was performed using TaqMan™ microRNA Assays (Thermo Fisher Scientific, Waltham, MA, USA) with TaqMan Gene Expression master Mix (Thermo Fisher Scientific, Waltham, MA, USA) and RT-PCR Roche 234 LighterCycler 480 with the appropriate temperature cycles (50 °C/2 min, 95 °C/10 min, 40 cycles: 95 °C/15 s and 60 °C/1 min). Normalisation was performed with RNU6B snoRNA and RNU43 snoRNA. Relative expression was calculated as 2−ΔΔCt.
Statistical analysis
The results of each experiment are presented as mean ± SEM. Statistical significance was determined using t-test and 2-way-ANOVA.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Kumar, S., Vijayan, M., Bhatti, J. S. & Reddy, P. H. MicroRNAs as Peripheral Biomarkers in Aging and Age-Related Diseases. Prog. Mol. Biol. Transl. Sci. 146, 47–94 (2017).
Bhaskaran, M. & Mohan, M. MicroRNAs: history, biogenesis, and their evolving role in animal development and disease. Vet. Pathol. 51, 759–774 (2014).
Elbay, A., Ercan, Ç., Akbaş, F., Bulut, H. & Ozdemir, H. Three new circulating microRNAs may be associated with wet age-related macular degeneration. Scand. J. Clin. Lab. Invest. 79, 1–7 (2019).
Ménard, C. et al. MicroRNA signatures in vitreous humour and plasma of patients with exudative AMD. Oncotarget 7, 19171–19184 (2016).
Tian, B. et al. miR-17-3p Exacerbates Oxidative Damage in Human Retinal Pigment Epithelial Cells. PLoS One 11, e0160887 (2016).
Yoon, C. et al. Delivery of miR-155 to retinal pigment epithelial cells mediated by Burkitt’s lymphoma exosomes. Tumor Biol. 37, 313–321 (2016).
Lin, H. et al. Effect of miR-23 on Oxidant-Induced Injury in Human Retinal Pigment Epithelial. Cells. Investig. Opthalmology Vis. Sci. 52, 6308 (2011).
He, J.-F., Du, Y., Jiang, B.-L. & He, J.-F. Increased microRNA-155 and decreased microRNA-146a may promote ocular inflammation and proliferation in Graves’ ophthalmopathy. Med. Sci. Monit. 20, 639–643 (2014).
Turturici, G., Tinnirello, R., Sconzo, G. & Geraci, F. Extracellular membrane vesicles as a mechanism of cell-to-cell communication: advantages and disadvantages. Am. J. Physiol. Cell Physiol. 306, C621–C633 (2014).
Yang, X., Weng, Z., Mendrick, D. L. & Shi, Q. Circulating extracellular vesicles as a potential source of new biomarkers of drug-induced liver injury. Toxicol. Lett. 225, 401–406 (2014).
Février, B. & Raposo, G. Exosomes: Endosomal-derived vesicles shipping extracellular messages. Curr. Opin. Cell Biol. 16, 415–421 (2004).
Théry, C. et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. vesicles 7, 1535750 (2018).
Creemers, E. E., Tijsen, A. J. & Pinto, Y. M. Circulating MicroRNAs. Circ. Res. 110, 483–495 (2012).
Biasutto, L., Chiechi, A., Couch, R., Liotta, L. A. & Espina, V. Retinal pigment epithelium (RPE) exosomes contain signaling phosphoproteins affected by oxidative stress. Exp. Cell Res. 319, 2113–2123 (2013).
Wang, S., Koster, K. M., He, Y. & Zhou, Q. miRNAs as potential therapeutic targets for age-related macular degeneration. Future Med. Chem. 4, 277–287 (2012).
Romano, G. L. et al. Retinal and Circulating miRNAs in Age-Related Macular Degeneration: An In vivo Animal and Human Study. Front. Pharmacol. 8, 168 (2017).
Szemraj, M. et al. Serum MicroRNAs as Potential Biomarkers of AMD. Med. Sci. Monit. 21, 2734–2742 (2015).
Berber, P., Grassmann, F., Kiel, C. & Weber, B. H. F. An Eye on Age-Related Macular Degeneration: The Role of MicroRNAs in Disease Pathology. Mol. Diagnosis Ther. 21, 31–43 (2017).
Ren, C. et al. Circulating miRNAs as potential biomarkers of age-related macular degeneration. Cell. Physiol. Biochem. 41, 1413–1423 (2017).
Kim, J. M. et al. Responses of Types 1 and 2 Neovascularization in Age-Related Macular Degeneration to Anti-Vascular Endothelial Growth Factor Treatment: Optical Coherence Tomography Angiography Analysis. Semin. Ophthalmol. 34, 168–176 (2019).
Strauss, O. The Retinal Pigment Epithelium in Visual Function. Physiol. Rev. 85, 845–881 (2005).
Hernández-Zimbrón, L. F. et al. Age-Related Macular Degeneration: New Paradigms for Treatment and Management of AMD. Oxid. Med. Cell. Longev. 2018, 8374647 (2018).
Shen, J. et al. MicroRNAs Regulate Ocular Neovascularization. Mol. Ther. 16, 1208–1216 (2008).
Grassmann, F. et al. A circulating MicroRNA profile is associated with late- stage neovascular age-related macular degeneration. PLoS One 9, 1–8 (2014).
Magenta, A., Greco, S., Gaetano, C. & Martelli, F. Oxidative Stress and MicroRNAs in Vascular Diseases. Int. J. Mol. Sci. 14, 17319–17346 (2013).
Baker, J. R. et al. Oxidative stress dependent microRNA-34a activation via PI3Kα reduces the expression of sirtuin-1 and sirtuin-6 in epithelial cells. Sci. Rep. 6, 35871 (2016).
Dinç, E., Ayaz, L. & Kurt, A. H. Effects of Bevacizumab, Ranibizumab, and Aflibercept on MicroRNA Expression in a Retinal Pigment Epithelium Cell Culture Model of Oxidative Stress. J. Ocul. Pharmacol. Ther. 34, 1–8 (2018).
Atienzar-Aroca, S. et al. Oxidative stress in retinal pigment epithelium cells increases exosome secretion and promotes angiogenesis in endothelial cells. J. Cell. Mol. Med. 20, 1457–1466 (2016).
Ayaz, L. & Dinç, E. Evaluation of microRNA responses in ARPE-19 cells against the oxidative stress. Cutan. Ocul. Toxicol. 37, 121–126 (2018).
Howell, J. C. et al. Global microRNA expression profiling: curcumin (diferuloylmethane) alters oxidative stress-responsive microRNAs in human ARPE-19 cells. Mol. Vis. 19, 544–560 (2013).
Maisto, R. et al. ARPE-19-derived VEGF-containing exosomes promote neovascularization in HUVEC: the role of the melanocortin receptor 5 has been built and requires approval. Cell Cycle 18, 413–424 (2019).
Bukong, T. N., Momen-Heravi, F., Kodys, K., Bala, S. & Szabo, G. Exosomes from hepatitis C infected patients transmit HCV infection and contain replication competent viral RNA in complex with Ago2-miR122-HSP90. PLoS Pathog. 10, e1004424 (2014).
Mittelbrunn, M. et al. Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat. Commun. 2, 282 (2011).
van Balkom, B. W. M., Eisele, A. S., Pegtel, D. M., Bervoets, S. & Verhaar, M. C. Quantitative and qualitative analysis of small RNAs in human endothelial cells and exosomes provides insights into localized RNA processing, degradation and sorting. J. Extracell. vesicles 4, 26760 (2015).
Jiao, X. et al. Serum and exosomal miR-122 and miR-199a as a biomarker to predict therapeutic efficacy of hepatitis C patients. J. Med. Virol. 89, 1597–1605 (2017).
Gao, Z., Zhu, X. & Dou, Y. The miR-302/367 cluster: a comprehensive update on its evolution and functions. Open Biol. 5, 150138 (2015).
Kuo, C.-H., Deng, J. H., Deng, Q. & Ying, S.-Y. A novel role of miR-302/367 in reprogramming. Biochem. Biophys. Res. Commun. 417, 11–16 (2012).
Barroso-del Jesus, A., Lucena-Aguilar, G. & Menendez, P. The miR-302-367 cluster as a potential stemness regulator in ESCs. Cell Cycle 8, 394–398 (2009).
Lipchina, I., Studer, L. & Betel, D. The expanding role of miR-302–367 in pluripotency and reprogramming. Cell Cycle 11, 1517–1523 (2012).
Chen, W., Zhuang, X., Qi, R. & Qiao, T. MiR-302a-5p suppresses cell proliferation and invasion in non-small cell lung carcinoma by targeting ITGA6. Am. J. Transl. Res. 11, 4348–4357 (2019).
Qin, C. et al. MicroRNA-302a inhibits cell proliferation and invasion, and induces cell apoptosis in hepatocellular carcinoma by directly targeting VEGFA. Mol. Med. Rep. 16, 6360–6367 (2017).
Li, C. et al. Chronic inflammation contributes to the development of hepatocellular carcinoma by decreasing miR-122 levels. Oncotarget 7, 17021–17034 (2016).
Qin, C. et al. MicroRNA-302a inhibits cell proliferation and invasion, and induces cell apoptosis in hepatocellular carcinoma by directly targeting VEGFA. Mol. Med. Rep. 16, 6360–6367 (2017).
Wang, Y. et al. MiR-122 targets VEGFC in bladder cancer to inhibit tumor growth and angiogenesis. Am. J. Transl. Res. 8, 3056–3066 (2016).
Bai, J. et al. DNA Methylation of miR-122 Aggravates Oxidative Stress in Colitis Targeting SELENBP1 Partially by p65NF- κ B Signaling. Oxid. Med. Cell. Longev. 2019, 1–14 (2019).
Song, L. et al. MicroRNA-122 is involved in oxidative stress in isoniazid-induced liver injury in mice. Genet. Mol. Res. 14, 13258–13265 (2015).
Zhou, Q. et al. Regulation of angiogenesis and choroidal neovascularization by members of microRNA-23~27~24 clusters. Proc. Natl. Acad. Sci. USA 108, 8287–8292 (2011).
Li, W.-B. et al. Development of Retinal Pigment Epithelium from Human Parthenogenetic Embryonic Stem Cells and MicroRNA Signature. Investig. Opthalmology Vis. Sci. 53, 5334–5343 (2012).
Genini, S., Guziewicz, K. E., Beltran, W. A. & Aguirre, G. D. Altered miRNA expression in canine retinas during normal development and in models of retinal degeneration. BMC Genomics 15, 172 (2014).
Drewry, M. D. et al. Differentially expressed microRNAs in the aqueous humor of patients with exfoliation glaucoma or primary open-angle glaucoma. Hum. Mol. Genet. 27, 1263–1275 (2018).
Pastukh, N., Meerson, A., Kalish, D., Jabaly, H. & Blum, A. Serum miR-122 levels correlate with diabetic retinopathy. Clin. Exp. Med. 19, 255–260 (2019).
Ertekin, S. et al. Evaluation of circulating miRNAs in wet age-related macular degeneration. Mol. Vis. 20, 1057–1066 (2014).
Wu, J. et al. Protective effects of methane-rich saline on diabetic retinopathy via anti-inflammation in a streptozotocin-induced diabetic rat model. Biochem. Biophys. Res. Commun. 466, 155–161 (2015).
Wang, H.-C. et al. Profiling the microRNA Expression in Human iPS and iPS-derived Retinal Pigment Epithelium. Cancer Inform. 13, 25–35 (2014).
Karali, M. et al. High-resolution analysis of the human retina miRNome reveals isomiR variations and novel microRNAs. Nucleic Acids Res. 44, 1525–1540 (2016).
Loscher, C. J. et al. Altered retinal microRNA expression profile in a mouse model of retinitis pigmentosa. Genome Biol. 8, R248 (2007).
Xiang, L. et al. miR-183/96 plays a pivotal regulatory role in mouse photoreceptor maturation and maintenance. Proc. Natl. Acad. Sci. 114, 6376–6381 (2017).
Li, Ddan et al. Inhibition of the oxidative stress-induced miR-23a protects the human retinal pigment epithelium (RPE) cells from apoptosis through the upregulation of glutaminase and glutamine uptake. Mol. Biol. Rep. 43, 1079–1087 (2016).
Zhang, J. et al. miR-25 Mediates Retinal Degeneration Via Inhibiting ITGAV and PEDF in Rat. Curr. Mol. Med. 17, 359–374 (2017).
SanGiovanni, J. P., SanGiovanni, P. M., Sapieha, P. & De Guire, V. miRNAs, single nucleotide polymorphisms (SNPs) and age-related macular degeneration (AMD). Clin. Chem. Lab. Med. 55, 763–775 (2017).
Desjarlais, M. et al. MicroRNA expression profile in retina and choroid in oxygen-induced retinopathy model. PLoS One 14, e0218282 (2019).
Walz, J. M. et al. Impact of angiogenic activation and inhibition on miRNA profiles of human retinal endothelial cells. Exp. Eye Res. 181, 98–104 (2019).
Zhang, Y. et al. Regulation of Matrix Metalloproteinase-2 Secretion from Scleral Fibroblasts and Retinal Pigment Epithelial Cells by miR-29a. Biomed. Res. Int. 2017, 1–7 (2017).
Zhang, Y. et al. miR-29a regulates the proliferation and differentiation of retinal progenitors by targeting Rbm8a. Oncotarget 8, 31993–32008 (2017).
Mazzeo, A. et al. Functional analysis of miR-21-3p, miR-30b-5p and miR-150-5p shuttled by extracellular vesicles from diabetic subjects reveals their association with diabetic retinopathy. Exp. Eye Res. 184, 56–63 (2019).
Haque, R. et al. MicroRNA-30b-Mediated Regulation of Catalase Expression in Human ARPE-19 Cells. PLoS One 7, e42542 (2012).
Acknowledgements
The present project was supported by internal funds from Universidad Católica de Valencia San Vicente Mártir (2018-128-001), Centro de Investigación Traslacional San Alberto Magno de la UCV (2019-128-001), and Conselleria de Educación, Investigación, Cultura y Deporte; Generalitat Valenciana (PROMETEO/2016/094). Oltra M and Vidal-Gil L, PhD training program fellowship UCV (EDUCV-PRE-2016-005 and EDUCV-PRE-2015-006 Personal Investigador en Formación UCV, respectively).
Author information
Authors and Affiliations
Contributions
M.O. Cell cultures and sEVs isolation, flow citometry, miRNA extraction and quantification, qPCR, validation. L.V.G. Cell cultures and sEVs isolation, electronic microscopy. R.M. Cell cultures and sEVs isolation. S.S.O. Statistical analysis. F.J.R. MS revision and interpretation of results. J.S.P. Corresponding author and discussion. J.M.B. supervision and MS writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oltra, M., Vidal-Gil, L., Maisto, R. et al. miR302a and 122 are deregulated in small extracellular vesicles from ARPE-19 cells cultured with H2O2. Sci Rep 9, 17954 (2019). https://doi.org/10.1038/s41598-019-54373-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-54373-x
- Springer Nature Limited
This article is cited by
-
Contribution of extracellular vesicles for the pathogenesis of retinal diseases: shedding light on blood-retinal barrier dysfunction
Journal of Biomedical Science (2024)