Abstract
It is known that patients with rheumatoid arthritis (RA) have a higher risk of coronary heart disease and sudden cardiac death. Abnormalities in cardiac geometry appear to be involved in the setting of the cardiovascular risk, but it has never been specifically investigated in RA. We enrolled 44 patients with RA compared to 131 subjects without RA (normal, N): The RA aged between 18 and 70 years (mean 48.3 ± 2.1), 25 females, BMI 27.6 ± 0.9; N, of equal age (48.6 ± 1.2, n.s.), included 80 females (BMI 26.7 ± 0.2, ns). Cardiac Ultrasounds showed an increase of the diameter of the left ventricle but not in the septum with reduction of relative wall thickness (RWT) in the RA population compared to N. Relative wall thickness inversely correlates with biochemical parameters of inflammatory response (gamma globulin, p < 0.03; F = 5,660) and anti citrullinated peptides antibody (anti-CCP Ab) (p < 0.02; F = 7,1620) We conclude that unfavorable cardiac remodeling can increase cardiovascular risk in patients with RA.
Similar content being viewed by others
Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory disease with an autoimmune pathogenesis with a very high risk of invalidity (80% of cases) and reduced survival1,2. The incidence rate of RA differs between 20 and 50 new cases every 100,000 people per year in North American and European countries and prevalence of the disease is higher in women3,4,5,6. RA affects mainly the diarthrosis joints, especially the proximal interphalangeal, metacarpophalangea, wrists, metatarsophalangeal, knees, elbow, ankle, scapular-humeral, hip and cervical spine, temporo-mandibular joint. RA has a multifactorial genesis, where genetic, environmental and random factors contribute to the development of the disease7. Among these latter, smoking and irritants are prevalent in patients that are positive to rheumatoid factor (RF) and/or to the anti-citrullinated peptides (anti-CCP) antibody. Several epidemiological studies have observed the association between the systemic autoimmune disorders and cardiovascular disease with this last as the prevalent cause of death in patients with RA8. This association, albeit well described, remains largely unexplained. Indeed, cardiovascular diseases are the main cause of death and disability in western countries, prevalently due to atherosclerotic lesions of the vascular tree9. Cardiovascular complications can be predicted in the general population on the basis of the number risk factors as collectively considered, which all together concur to the definition of the “global cardiovascular risk”, and include parental history, lifestyle and unhealthy habits. The recently developed concept of the cardiovascular continuum10 has also introduced the research of subclinical organ damage for a better definition of the cardiovascular risk. In particular, changes in cardiac geometry including eccentric or concentric remodeling, associated with increase in left ventricle size can be assessed easily by cardiac ultrasounds (CUS), and allow to detect those modification that are more often associated with the risk of cardiovascular events11,12,13. Several findings have shown the presence of diastolic dysfunction without manifest cardiac disease14,15,16,17 in patients with RA as compared to control patients not affected by either RA or cardiovascular disease. Therefore, the employment of early markers of cardiac disease can possibly help to identify RA patients at higher risk to develop cardiac failure. In this study, we investigated the role of CUS-assessed eccentric remodeling in setting up the cardiovascular risk of patients with RA.
Materials and Methods
Population
For this study we enrolled 44 patients with diagnosis of RA according to the ACR/EULAR 2010 criteria18, from the Rheumatology Division of AOU San Giovanni di Dio and Ruggi d’Aragona. Patients with known cardiovascular conditions, including hypertension, cardiac ischemia, heart failure, diabetes, valves diseases, pulmonary and aortic complications were excluded. As control group, we selected 131 subjects, not suffering of RA or cardiovascular disease from our outpatient clinic of the San Giovanni di Dio e Ruggi d’Aragona Hospital. All parameters, including anamnesis, physical examination, biochemistry, EKG, cardiac and vascular ultrasound and ambulatory blood pressure monitoring were collected during ambulatory visits and stored in the server of the Hospital. All procedures were performed in accordance with the EULAR Guidelines. Written informed consent was obtained from patients. The protocol was approved by the Ethical Committee of the San Giovanni di Dio e Ruggi d’Aragona Hospital, Salerno.
Clinical and biochemical assessments
Disease activity according to the DAS28 and the degree of disability by HAQ were evaluated19. A blood sample was collected after 10 h fasting to measure RF, anti-CCP, antinuclear anti-body (ANA), C-reactive protein (PCR), Erythrocyte sedimentation rate (ESR). Moreover, all patients were subjected to mono-dimensional (M-mode), bi-dimensional (B-mode), Doppler and color Doppler cardiac ultrasound (CUS) and via CUS probe 5-1 MHz. We evaluated: interventricular septum (IVSTd) and posterior wall thickness in diastole (LVPWTd), ventricular end-diastolic and end-systolic diameters (LVIDd, LVIDs) aortic root diameter, the end-diastolic diameter of the atrium. Relative Wall Tickness (RWT) was calculated according to the formula: (IVSTd + LVPWTd)/ LVIDd.
Statistical analysis
A Power Analysis was performed to identify the sample size required to detect a difference in cardiac size of at least 7 mm with power of 85%. Anthropometric, biochemical and cardiac ultrasound parameters are expressed as mean ± standard error. Differences among patients and controls were analyzed by unpaired Student’s t test; relationships between variables were considered by linear correlation. All data were analyzed using Prism 6.
Results
Biochemical and CUS parameters
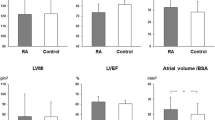
We considered 44 RA patients (group 1), aged between 18 and 70 years, including 25 females, and a control group (group 2) consisting of 131 healthy subjects (80 females) not affected by RA and CVD. The anthropometric parameters of the two groups were similar (Table 1). Similarly, the two groups did not show differences in the metabolic profile and renal function (Table 2). Comparing echocardiographic parameters between the two populations, we observed a statistically significant increase in both LVIDd and LVIDs in patients with RA, without differences in IVSTd and LVPWTd (Table 3). Eccentric dilatation is also confirmed by RWT, which is significantly decreased in patients with RA as compared to control, indicating a prevalent left ventricle eccentric remodeling in the RA group (Table 3).
Association of RWT and biochemical patterns in the population affected by AR
Patients with eccentric remodeling are considered at higher CVR. In order to identify possible determinants of eccentric remodeling in patients with RA, we evaluated through correlation analysis the impact of biochemical variables on the left ventricle geometry. We observed that in patients with RA, RWT directly correlates to age (Fig. 1A) and inversely correlates to the diastolic function as evidenced by E/A ratio (Fig. 1B) and E/E’ ratio (Fig. 1C). These findings are consistent with previous literature20 and validate the observation in our study group. In RA group, RWT value inversely correlates with the levels of total plasma proteins (Fig. 2A) and gamma globulin (Fig. 2B), but no significant correlation was found with the serum albumin level (Fig. 2C). Anti-CCPs are specific markers of RA and they can be found at the early stage of disease21,22 and serum levels > 60 U/ml indicate advanced RA condition. In our population, the highest level of anti-CCP was found in patients between fifth and sixth decade of age. Therefore, we selected patients with RA with anti-CCP Ab values between 60 and 2000 U/ml and aged between 40 and 60 years old. In this subgroup, we observed a significant inverse correlation between RWT and anti-CCP level (Fig. 3).
Discussion
Our study shows for the first time that RA patients present cardiac eccentric remodeling before clinical features of cardiovascular disease and in absence of the common cardiovascular risk factors. Our observations confirm and extend previous findings of reduced diastolic function in RA14 and are in line with recent publication evaluating alterations of cardiac geometry in RA patients. In particular, Fine et al.23 studied the myocardial deformation during ventricular contraction and relaxation by using speckle tracking echocardiography (STE). There, it was revealed that the longitudinal structural of the left and right ventricular wall at systolic peak is significantly worsened in patients with RA with preserved diastolic function. The result persisted after adjustment for age, gender, blood pressure, BMI and heart rate and also after comparing patients with controls. These data support the idea that measuring shortening abnormalities is a more sensitive parameter than diastolic dysfunction to detect presence of cardiomyopathy in subclinical phase, at least in RA population. Our data confirm that an early sign of cardiac damage is a feature of RA, and add the consideration that RWT being a parameter of easy consultation, internists can measure it with a standard CUS, and therefore assess it in all patients affected by RA, while STE being still an instrument available only to expert CUS laboratories is mainly available to cardiologists.
Our data show that aging directly correlates with RWT in RA patients as well as in controls. This is somewhat expected20, but this phenomenon might attenuate our finding of reduced RWT in the aged population (i.e. >65 years old). When limiting the analysis of RWT to patients <65 yrs old, though, we noticed that patients with the longest history of disease (>15 yrs) showed a tendency towards the lowest RWT (data not shown). This latter finding is consistent with previous literature14. Indeed, in the Lugo study, disease patients with diastolic dysfunction were older than those without this abnormality14.
Considering that cardiac eccentric remodeling in RA cannot be attributed to clinical cardiovascular risk factors, the inflammatory hypothesis responsible for structural changes holds the most likely. The observation that RWT inversely correlates with gamma globulin, and in particular with Anti-CCP levels fits properly in this pathophysiological context. CHF can evolve from a variety of pathogenic conditions including RA. However, it still remains difficult to discern the specific weight of RA respect to the common risk factors for CHF24. The problem of defining the incidence of CHF in patients with RA may reside in the diagnostic process; indeed, the use of only clinical criteria for the diagnosis can result inaccurate, with both over- or lack of diagnosis. This is the case of edema of the ankles that can be confused with swollen joints and/or lung congestion interpreted as pulmonary involvement of RA. For this reason, guidelines for RA state the need to include CUS into to the diagnostic flowchart for CHF, even when is only suspected25. So far only CUS markers of diastolic dysfunction26,27,28 have been associated to RA29,30, but here we demonstrated that RWT could be added to the list since it is strictly associated to level of inflammation. This last observation can have relevant implication in the therapeutic approach. Among patients without history of cardiac ischemic disease, those with higher serum levels of IL-6, CRP, TNF appeared to have a two or four times higher risk of developing CHF compared to patients with lower levels of cytokines31,32,33,34,35. Recently, the result of the CANTOS clinical trial testing the use of Canakinumab monoclonal anti-IL1b antibody in gout patients has shown a reduction in cardiovascular events, therefore providing further support to the link between excessive inflammatory response and increased CVR36. Cardiac remodeling too is driven by cytokines and serum levels TNFα have been demonstrated to regulate expression of metallo proteinases (MMPs) and of their inhibitors (TIMPs) producing increase in the ratio MMPs/TIMPs, degradation of interstitial collagen fibers and development of ventricular dilatation and CHF. Along the time, however, there is an increase of the production of TIMPs responsible for a reduction in the ratio MMPs/TIMPs, from which derives an increase in the production of collagen and subsequent fibrosis and ventricular dilatation37. This time-dependent effect of TNF in the induction of cardiac remodeling suggests presence of an early window where interstitial fibrosis can be prevented38. This explains why treatment with anti TNF is effective to reverse the process of ventricular dilatation only when administered in the early stage, and the apparent contradiction of the worsening of the clinical condition induced by biological therapy with anti-TNF in patients with advanced CHF39. These considerations support research of early markers of cardiac remodeling in RA, and here, we, once again, have identified that RWT modifications is significantly correlated with the level of inflammatory activity of RA measured by Anti-CCP level.
Our study is relevant also in the Public Health perspective. Indeed, the quest towards sustainability of increased aging in western societies is calling for strategies of prevention, early disease detection and more efficient therapy40,41,42. In particular, chronic diseases represent an important challenge to tackle, for their global burden in terms of disability43. Among these, RA brings in a major burden for polytherapy, comorbidity, hospital accesses and increased disability44. In particular, the association of increased risk of cardiovascular events in RA is a major problem with still unanswered questions45. In this context, the identification of early marker of cardiovascular complications may allow early diagnosis and therapy. Previous study has indeed identified presence of diastolic dysfunction in RA population without clinical evidence of cardiovascular disease, firstly advancing the idea that prevention of cardiovascular complications in RA can be favored by early assessment of RA patients by CUS14. We provide a reliable, easily assessed parameter of CUS that can be performed without the assistance of specialized CUS laboratory.
In conclusion, our study suggests that the diagnostic path of RA patients should be modified flanking the rheumatology visit with a cardiologic examination as soon as possible after the first contact. Moreover, it is necessary to include CUS in the diagnostic path and risk assessment, given the evidence that cardiovascular risk for patients with RA is independent and it can be easily detected with the evaluation of RWT.
Significance
-
(1)
Cardiac eccentric remodeling is significantly correlated with the level of inflammatory activity of RA.
-
(2)
RWT is an easy and accessible method to predict progression towards HF in all RA patients.
-
(3)
Early clinical and echocardiographic evaluation of patients with RA can prevent development of HF.
References
Riise, T., Jacobsen, B. K. & Gran, J. T. High mortality in patients with rheumatoid arthritis and atlantoaxial subluxation. The Journal of rheumatology 28, 2425–2429 (2001).
Young, A. et al. Mortality in rheumatoid arthritis. Increased in the early course of disease, in ischaemic heart disease and in pulmonary fibrosis. Rheumatology 46, 350–357 (2007).
Neri Serneri, G. G. & Von, B. [Heredo-pathology of reactive mesenchymal diseases (so-called collagen diseases). II. Researches on hereditary factors of chronic primary rheumatism]. Acta geneticae medicae et gemellologiae 5, 402–425 (1956).
Einaudi, G. [Investigations of the Diffusion and Characteristics of Cardio-Rheumatological Diseases in the Total Population of a Region of the Province of Turin]. Minerva medica 54, 2472–2483 (1963).
van de Sande, M. G. & Baeten, D. L. Immunopathology of synovitis: from histology to molecular pathways. Rheumatology 55, 599–606 (2016).
Smolen, J. S., Aletaha, D. & McInnes, I. B. Rheumatoid arthritis. Lancet, (2016).
López-Mejías R, et al risk assessment in patients with rheumatoid arthritis: The relevance of clinical, genetic and serological markers.
Wolfe, F., Freundlich, B. & Straus, W. L. Increase in cardiovascular and cerebrovascular disease prevalence in rheumatoid arthritis. The Journal of rheumatology 30, 36–40 (2003).
Joseph, P. et al. Reducing the Global Burden of Cardiovascular Disease, Part 1: The Epidemiology and Risk Factors. Circ Res 121, 677–694 (2017).
Dzau, V. J. et al. The cardiovascular disease continuum validated: clinical evidence of improved patient outcomes: part I: Pathophysiology and clinical trial evidence (risk factors through stable coronary artery disease). Circulation 114, 2850–2870 (2006).
Marwick, T. H. et al. Recommendations on the use of echocardiography in adult hypertension: a report from the European Association of Cardiovascular Imaging (EACVI) and the American Society of Echocardiography (ASE)dagger. European heart journal cardiovascular Imaging 16, 577–605 (2015).
Krumholz, H. M., Larson, M. & Levy, D. Prognosis of left ventricular geometric patterns in the Framingham Heart Study. Journal of the American College of Cardiology 25, 879–884 (1995).
Verdecchia, P. et al. Impact of Chamber Dilatation on the Prognostic Value of Left Ventricular Geometry in Hypertension. J Am Heart Assoc 6, (2017).
Gonzalez-Juanatey, C. et al. Echocardiographic and Doppler findings in long-term treated rheumatoid arthritis patients without clinically evident cardiovascular disease. Seminars in arthritis and rheumatism 33, 231–238 (2004).
Rexhepaj, N. et al. Left and right ventricular diastolic functions in patients with rheumatoid arthritis without clinically evident cardiovascular disease. International journal of clinical practice 60, 683–688 (2006).
Meune, C. et al. Myocardial dysfunction in rheumatoid arthritis: a controlled tissue-Doppler echocardiography study. The Journal of rheumatology 34, 2005–2009 (2007).
Arslan, S., Bozkurt, E., Sari, R. A. & Erol, M. K. Diastolic function abnormalities in active rheumatoid arthritis evaluation by conventional Doppler and tissue Doppler: relation with duration of disease. Clinical rheumatology 25, 294–299 (2006).
Cader, M. Z. et al. Performance of the 2010 ACR/EULAR criteria for rheumatoid arthritis: comparison with 1987 ACR criteria in a very early synovitis cohort. Annals of the rheumatic diseases 70 949–955, (2011).
Lee, K. E. et al. HAQ score is an independent predictor of sustained remission in patients with rheumatoid arthritis. Rheumatol Int, (2017).
de Simone, G. et al. Evaluation of concentric left ventricular geometry in humans: evidence for age-related systematic underestimation. Hypertension 45, 64–68 (2005).
Predeteanu, D. et al. Anti-cyclic citrullinated peptide antibodies–activity markers in rheumatoid arthritis. Journal of medicine and life 2, 36–41 (2009).
Rezus, E., Grigoriu, A. & Rezus, C. [Aggressive nature of rheumatic arthritis with citrullinated cyclic peptide antibodies]. Revista medico-chirurgicala a Societatii de Medici si Naturalisti din Iasi 113, 73–78 (2009).
Fine, N. M. et al. Evaluation of myocardial function in patients with rheumatoid arthritis using strain imaging by speckle-tracking echocardiography. Annals of the rheumatic diseases 73, 1833–1839 (2014).
Giles, J. T., Fernandes, V., Lima, J. A. & Bathon, J. M. Myocardial dysfunction in rheumatoid arthritis: epidemiology and pathogenesis. Arthritis research & therapy 7, 195–207 (2005).
Svensson, A. L. et al. Multifactorial intervention to prevent cardiovascular disease in patients with early rheumatoid arthritis: protocol for a multicentre randomised controlled trial. BMJ open 6, e009134 (2016).
Vasan, R. S., Larson, M. G., Benjamin, E. J., Evans, J. C. & Levy, D. Left ventricular dilatation and the risk of congestive heart failure in people without myocardial infarction. The New England journal of medicine 336, 1350–1355 (1997).
Drazner, M. H. et al. Increased left ventricular mass is a risk factor for the development of a depressed left ventricular ejection fraction within five years: the Cardiovascular Health Study. Journal of the American College of Cardiology 43, 2207–2215 (2004).
Lauer, M. S., Evans, J. C. & Levy, D. Prognostic implications of subclinical left ventricular dilatation and systolic dysfunction in men free of overt cardiovascular disease (the Framingham Heart Study). The American journal of cardiology 70, 1180–1184 (1992).
Vasan, R. S. et al. Congestive heart failure in subjects with normal versus reduced left ventricular ejection fraction: prevalence and mortality in a population-based cohort. Journal of the American College of Cardiology 33, 1948–1955 (1999).
Zile, M. R. & Brutsaert, D. L. New concepts in diastolic dysfunction and diastolic heart failure: Part II: causal mechanisms and treatment. Circulation 105, 1503–1508 (2002).
Deswal, A. et al. Cytokines and cytokine receptors in advanced heart failure: an analysis of the cytokine database from the Vesnarinone trial (VEST). Circulation 103, 2055–2059 (2001).
Kubota, T. et al. Expression of proinflammatory cytokines in the failing human heart: comparison of recent-onset and end-stage congestive heart failure. The Journal of heart and lung transplantation: the official publication of the International Society for Heart Transplantation 19, 819–824 (2000).
Levine, B., Kalman, J., Mayer, L., Fillit, H. M. & Packer, M. Elevated circulating levels of tumor necrosis factor in severe chronic heart failure. The New England journal of medicine 323, 236–241 (1990).
Rauchhaus, M. et al. Plasma cytokine parameters and mortality in patients with chronic heart failure. Circulation 102, 3060–3067 (2000).
Torre-Amione, G. et al. Proinflammatory cytokine levels in patients with depressed left ventricular ejection fraction: a report from the Studies of Left Ventricular Dysfunction (SOLVD). Journal of the American College of Cardiology 27, 1201–1206 (1996).
Ridker, P. M. et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. The New England journal of medicine 377, 1119–1131 (2017).
Sivasubramanian, N. et al. Left ventricular remodeling in transgenic mice with cardiac restricted overexpression of tumor necrosis factor. Circulation 104, 826–831 (2001).
Berry, M. F. et al. Administration of a tumor necrosis factor inhibitor at the time of myocardial infarction attenuates subsequent ventricular remodeling. The Journal of heart and lung transplantation: the official publication of the International Society for Heart Transplantation 23, 1061–1068 (2004).
Kubota, T. et al. Soluble tumor necrosis factor receptor abrogates myocardial inflammation but not hypertrophy in cytokine-induced cardiomyopathy. Circulation 101, 2518–2525 (2000).
Illario, M., Iaccarino, G., Piazza, O., Menditto, E. & Coscioni, E. Proceedings of the EIP on AHA: A3 Action Group on Frailty. Transl Med UniSa 13, 1–3, (2015).
Liotta, G. et al. The European Innovation Partnership on Active and Healthy Ageing Synergies: Protocol for a Prospective Observational Study to Measure the Impact of a Community-Based Program on Prevention and Mitigation of Frailty (ICP - PMF) in Community-Dwelling Older Adults. Transl Med UniSa 15, 53–66 (2016).
Illario, M. et al. The Italian reference sites of the European innovation partnership on active and healthy ageing: Progetto Mattone Internazionale as an enabling factor. Ann Ist Super Sanita 53, 60–69 (2017).
Disease, G. B. D., Injury, I. & Prevalence, C. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 390, 1211–1259 (2017).
Filkova, M. et al. Polypharmacy and Unplanned Hospitalizations in Patients with Rheumatoid Arthritis. The Journal of rheumatology, (2017).
Symmons, D. P. & Gabriel, S. E. Epidemiology of CVD in rheumatic disease, with a focus on RA and SLE. Nat Rev Rheumatol 7, 399–408 (2011).
Acknowledgements
This work was in part supported by a grants from Italian Ministry of Research (PRIN 2009 and PON Campania Bioscience PON03PE0006008 to GI), POR MOVIE from Regione Campania Rete di Biotecnologie (GI), University of Salerno’s Funds for Basic Research (FARB to GI and MC), and Italian Society of Hypertension (SIIA to GI).
Author information
Authors and Affiliations
Contributions
V.P., R.F., R.G. performed data collection and prepared manuscript and figures; E.C., D.C., P.M. managed patients and database; G.I. and M.C. designed and coordinated the study, performed analysis and corrected the manuscript; R.I. and F.R. revised and critically read the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pascale, V., Finelli, R., Giannotti, R. et al. Cardiac eccentric remodeling in patients with rheumatoid arthritis. Sci Rep 8, 5867 (2018). https://doi.org/10.1038/s41598-018-24323-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24323-0
- Springer Nature Limited
This article is cited by
-
Left ventricular remodeling in rheumatoid arthritis patients without clinical heart failure
Arthritis Research & Therapy (2023)