Abstract
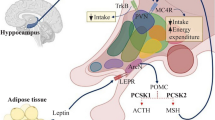
Genome-wide association studies (GWAS) have identified >250 loci for body mass index (BMI), implicating pathways related to neuronal biology. Most GWAS loci represent clusters of common, noncoding variants from which pinpointing causal genes remains challenging. Here we combined data from 718,734 individuals to discover rare and low-frequency (minor allele frequency (MAF) < 5%) coding variants associated with BMI. We identified 14 coding variants in 13 genes, of which 8 variants were in genes (ZBTB7B, ACHE, RAPGEF3, RAB21, ZFHX3, ENTPD6, ZFR2 and ZNF169) newly implicated in human obesity, 2 variants were in genes (MC4R and KSR2) previously observed to be mutated in extreme obesity and 2 variants were in GIPR. The effect sizes of rare variants are ~10 times larger than those of common variants, with the largest effect observed in carriers of an MC4R mutation introducing a stop codon (p.Tyr35Ter, MAF = 0.01%), who weighed ~7 kg more than non-carriers. Pathway analyses based on the variants associated with BMI confirm enrichment of neuronal genes and provide new evidence for adipocyte and energy expenditure biology, widening the potential of genetically supported therapeutic targets in obesity.


Similar content being viewed by others
Change history
03 June 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
16 March 2018
A Correction to this paper has been published: https://doi.org/10.1038/s41588-018-0050-y
References
Bray, G. A. & Ryan, D. H. Update on obesity pharmacotherapy. Ann. NY Acad. Sci. 1311, 1–13 (2014).
Bray, G. A., Frühbeck, G., Ryan, D. H. & Wilding, J. P. Management of obesity. Lancet 387, 1947–1956 (2016).
Monda, K. L. et al. A meta-analysis identifies new loci associated with body mass index in individuals of African ancestry. Nat. Genet. 45, 690–696 (2013).
Wen, W. et al. Meta-analysis of genome-wide association studies in East Asian-ancestry populations identifies four new loci for body mass index. Hum. Mol. Genet. 23, 5492–5504 (2014).
Locke, A. E. et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206 (2015).
Winkler, T. W. et al. The influence of age and sex on genetic associations with adult body size and shape: a large-scale genome-wide interaction study. PLoS Genet. 11, e1005378 (2015).
Akiyama, M. et al. Genome-wide association study identifies 112 new loci for body mass index in the Japanese population. Nat. Genet. 49, 1458–1467 (2017).
van der Klaauw, A. A. & Farooqi, I. S. The hunger genes: pathways to obesity. Cell 161, 119–132 (2015).
Edwards, S. L., Beesley, J., French, J. D. & Dunning, A. M. Beyond GWASs: illuminating the dark road from association to function. Am. J. Hum. Genet. 93, 779–797 (2013).
Stratigopoulos, G. et al. Hypomorphism of Fto and Rpgrip1l causes obesity in mice. J. Clin. Invest. 126, 1897–1910 (2016).
Stratigopoulos, G., LeDuc, C. A., Cremona, M. L., Chung, W. K. & Leibel, R. L. Cut-like homeobox 1 (CUX1) regulates expression of the fat mass and obesity-associated and retinitis pigmentosa GTPase regulator–interacting protein-1-like (RPGRIP1L) genes and coordinates leptin receptor signaling. J. Biol. Chem. 286, 2155–2170 (2011).
Stratigopoulos, G. et al. Hypomorphism for RPGRIP1L, a ciliary gene vicinal to the FTO locus, causes increased adiposity in mice. Cell Metab. 19, 767–779 (2014).
Claussnitzer, M. et al. FTO obesity variant circuitry and adipocyte browning in humans. N. Engl. J. Med. 373, 895–907 (2015).
Smemo, S. et al. Obesity-associated variants within FTO form long-range functional connections with IRX3. Nature 507, 371–375 (2014).
Marouli, E. et al. Rare and low-frequency coding variants alter human adult height. Nature 542, 186–190 (2017).
Fuchsberger, C. et al. The genetic architecture of type 2 diabetes. Nature 536, 41–47 (2016).
Sina, M. et al. Phenotypes in three pedigrees with autosomal dominant obesity caused by haploinsufficiency mutations in the melanocortin-4 receptor gene. Am. J. Hum. Genet. 65, 1501–1507 (1999).
Pearce, L. R. et al. KSR2 mutations are associated with obesity, insulin resistance, and impaired cellular fuel oxidation. Cell 155, 765–777 (2013).
Hinney, A. et al. Several mutations in the melanocortin-4 receptor gene including a nonsense and a frameshift mutation associated with dominantly inherited obesity in humans. J. Clin. Endocrinol. Metab. 84, 1483–1486 (1999).
Purcell, S. M. et al. A polygenic burden of rare disruptive mutations in schizophrenia. Nature 506, 185–190 (2014).
van den Berg, L. et al. Melanocortin-4 receptor gene mutations in a Dutch cohort of obese children. Obesity 19, 604–611 (2011).
Surendran, P. et al. Trans-ancestry meta-analyses identify rare and common variants associated with blood pressure and hypertension. Nat. Genet. 48, 1151–1161 (2016).
Lunetta, K. L. et al. Rare coding variants and X-linked loci associated with age at menarche. Nat. Commun. 6, 7756 (2015).
Zhou, Z. et al. Cidea-deficient mice have lean phenotype and are resistant to obesity. Nat. Genet. 35, 49–56 (2003).
Tews, D. et al. Comparative gene array analysis of progenitor cells from human paired deep neck and subcutaneous adipose tissue. Mol. Cell. Endocrinol. 395, 41–50 (2014).
Stutzmann, F. et al. Non-synonymous polymorphisms in melanocortin-4 receptor protect against obesity: the two facets of a Janus obesity gene. Hum. Mol. Genet. 16, 1837–1844 (2007).
Lin, H. Q., Wang, Y., Chan, K. L., Ip, T. M. & Wan, C. C. Differential regulation of lipid metabolism genes in the brain of acetylcholinesterase knockout mice. J. Mol. Neurosci. 53, 397–408 (2014).
Vignaud, A. et al. Genetic ablation of acetylcholinesterase alters muscle function in mice. Chem. Biol. Interact. 175, 129–130 (2008).
Ji, Z., Mei, F. C. & Cheng, X. Epac, not PKA catalytic subunit, is required for 3T3-L1 preadipocyte differentiation. Front. Biosci. 2, 392–398 (2010).
Yan, J. et al. Enhanced leptin sensitivity, reduced adiposity, and improved glucose homeostasis in mice lacking exchange protein directly activated by cyclic AMP isoform 1. Mol. Cell. Biol. 33, 918–926 (2013).
Almahariq, M., Mei, F. C. & Cheng, X. Cyclic AMP sensor EPAC proteins and energy homeostasis. Trends Endocrinol. Metab. 25, 60–71 (2014).
Kai, A. K. et al. Exchange protein activated by cAMP 1 (Epac1)-deficient mice develop β-cell dysfunction and metabolic syndrome. FASEB J. 27, 4122–4135 (2013).
Hardie, D. G., Ross, F. A. & Hawley, S. A. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 13, 251–262 (2012).
Hardie, D. G. & Ashford, M. L. AMPK: regulating energy balance at the cellular and whole body levels. Physiology 29, 99–107 (2014).
López, M., Nogueiras, R., Tena-Sempere, M. & Diéguez, C. Hypothalamic AMPK: a canonical regulator of whole-body energy balance. Nat. Rev. Endocrinol. 12, 421–432 (2016).
Minokoshi, Y. et al. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 428, 569–574 (2004).
Viollet, B. et al. The AMP-activated protein kinase α2 catalytic subunit controls whole-body insulin sensitivity. J. Clin. Invest. 111, 91–98 (2003).
Xue, B. et al. Neuronal protein tyrosine phosphatase 1B deficiency results in inhibition of hypothalamic AMPK and isoform-specific activation of AMPK in peripheral tissues. Mol. Cell. Biol. 29, 4563–4573 (2009).
Warren, H. R. et al. Genome-wide association analysis identifies novel blood pressure loci and offers biological insights into cardiovascular risk. Nat. Genet. 49, 403–415 (2017).
Chami, N. et al. Exome genotyping identifies pleiotropic variants associated with red blood cell traits. Am. J. Hum. Genet. 99, 8–21 (2016).
Li, M. et al. SOS2 and ACP1 loci identified through large-scale exome chip analysis regulate kidney development and function. J. Am. Soc. Nephrol. 28, 981–994 (2017).
Liu, C. et al. Meta-analysis identifies common and rare variants influencing blood pressure and overlapping with metabolic trait loci. Nat. Genet. 48, 1162–1170 (2016).
Schwartz, M. W., Woods, S. C., Porte, D. Jr, Seeley, R. J. & Baskin, D. G. Central nervous system control of food intake. Nature 404, 661–671 (2000).
Garfield, A. S. et al. A neural basis for melanocortin-4 receptor–regulated appetite. Nat. Neurosci. 18, 863–871 (2015).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Fan, W., Boston, B. A., Kesterson, R. A., Hruby, V. J. & Cone, R. D. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature 385, 165–168 (1997).
Yeo, G. S. H. et al. A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nat. Genet. 20, 111–112 (1998).
Vaisse, C., Clement, K., Guy-Grand, B. & Froguel, P. A frameshift mutation in human MC4R is associated with a dominant form of obesity. Nat. Genet. 20, 113–114 (1998).
Lubrano-Berthelier, C. et al. Melanocortin 4 receptor mutations in a large cohort of severely obese adults: prevalence, functional classification, genotype–phenotype relationship, and lack of association with binge eating. J. Clin. Endocrinol. Metab. 91, 1811–1818 (2006).
Farooqi, I. S. et al. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N. Engl. J. Med. 348, 1085–1095 (2003).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 536, 285–291 (2016).
Hinney, A. et al. Melanocortin-4 receptor gene: case–control study and transmission disequilibrium test confirm that functionally relevant mutations are compatible with a major gene effect for extreme obesity. J. Clin. Endocrinol. Metab. 88, 4258–4267 (2003).
Speliotes, E. K. et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat. Genet. 42, 937–948 (2010).
Saxena, R. et al. Genetic variation in GIPR influences the glucose and insulin responses to an oral glucose challenge. Nat. Genet. 42, 142–148 (2010).
Miyawaki, K. et al. Inhibition of gastric inhibitory polypeptide signaling prevents obesity. Nat. Med. 8, 738–742 (2002).
Hansotia, T. et al. Extrapancreatic incretin receptors modulate glucose homeostasis, body weight, and energy expenditure. J. Clin. Invest. 117, 143–152 (2007).
Fulurija, A. et al. Vaccination against GIP for the treatment of obesity. PLoS One 3, e3163 (2008).
Finan, B. et al. Reappraisal of GIP pharmacology for metabolic diseases. Trends Mol. Med. 22, 359–376 (2016).
Irwin, N. & Flatt, P. R. Evidence for beneficial effects of compromised gastric inhibitory polypeptide action in obesity-related diabetes and possible therapeutic implications. Diabetologia 52, 1724–1731 (2009).
Revelli, J. P. et al. Profound obesity secondary to hyperphagia in mice lacking kinase suppressor of ras 2. Obesity 19, 1010–1018 (2011).
Costanzo-Garvey, D. L. et al. KSR2 is an essential regulator of AMP kinase, energy expenditure, and insulin sensitivity. Cell Metab. 10, 366–378 (2009).
Brommage, R. et al. High-throughput screening of mouse knockout lines identifies true lean and obese phenotypes. Obesity 16, 2362–2367 (2008).
Liu, L. et al. Proteomic characterization of the dynamic KSR-2 interactome, a signaling scaffold complex in MAPK pathway. Biochim. Biophys. Acta 1794, 1485–1495 (2009).
Kühnen, P. et al. Proopiomelanocortin deficiency treated with a melanocortin-4 receptor agonist. N. Engl. J. Med. 375, 240–246 (2016).
Xiang, Y. Y., Dong, H., Yang, B. B., Macdonald, J. F. & Lu, W. Y. Interaction of acetylcholinesterase with neurexin-1β regulates glutamatergic synaptic stability in hippocampal neurons. Mol. Brain 7, 15 (2014).
Bartels, C. F., Zelinski, T. & Lockridge, O. Mutation at codon 322 in the human acetylcholinesterase (ACHE) gene accounts for YT blood group polymorphism. Am. J. Hum. Genet. 52, 928–936 (1993).
Farlow, M. R. et al. Effectiveness and tolerability of high-dose (23 mg/d) versus standard-dose (10 mg/d) donepezil in moderate to severe Alzheimer’s disease: a 24-week, randomized, double-blind study. Clin. Ther. 32, 1234–1251 (2010).
Farlow, M. et al. Safety and tolerability of donepezil 23 mg in moderate to severe Alzheimer’s disease. BMC Neurol. 11, 57 (2011).
Tariot, P., Salloway, S., Yardley, J., Mackell, J. & Moline, M. Long-term safety and tolerability of donepezil 23 mg in patients with moderate to severe Alzheimer’s disease. BMC Res. Notes 5, 283 (2012).
Hu, Y. et al. Role of exchange protein directly activated by cyclic AMP isoform 1 in energy homeostasis: regulation of leptin expression and secretion in white adipose tissue. Mol. Cell. Biol. 36, 2440–2450 (2016).
Altarejos, J. Y. et al. The Creb1 coactivator Crtc1 is required for energy balance and fertility. Nat. Med. 14, 1112–1117 (2008).
Winnier, D. A. et al. Transcriptomic identification of ADH1B as a novel candidate gene for obesity and insulin resistance in human adipose tissue in Mexican Americans from the Veterans Administration Genetic Epidemiology Study (VAGES). PLoS One 10, e0119941 (2015).
Molotkov, A., Deltour, L., Foglio, M. H., Cuenca, A. E. & Duester, G. Distinct retinoid metabolic functions for alcohol dehydrogenase genes Adh1 and Adh4 in protection against vitamin A toxicity or deficiency revealed in double null mutant mice. J. Biol. Chem. 277, 13804–13811 (2002).
GTEx Consortium. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
Volpicelli-Daley, L. A., Duysen, E. G., Lockridge, O. & Levey, A. I. Altered hippocampal muscarinic receptors in acetylcholinesterase-deficient mice. Ann. Neurol. 53, 788–796 (2003).
Ivanenkov, V. V., Murphy-Piedmonte, D. M. & Kirley, T. L. Bacterial expression, characterization, and disulfide bond determination of soluble human NTPDase6 (CD39L2) nucleotidase: implications for structure and function. Biochemistry 42, 11726–11735 (2003).
Jain, R. N. et al. Hip1r is expressed in gastric parietal cells and is required for tubulovesicle formation and cell survival in mice. J. Clin. Invest. 118, 2459–2470 (2008).
Engqvist-Goldstein, A. E. et al. RNAi-mediated Hip1R silencing results in stable association between the endocytic machinery and the actin assembly machinery. Mol. Biol. Cell 15, 1666–1679 (2004).
Tao, Y. X. The melanocortin-4 receptor: physiology, pharmacology, and pathophysiology. Endocr. Rev. 31, 506–543 (2010).
Stutzmann, F. et al. Prevalence of melanocortin-4 receptor deficiency in Europeans and their age-dependent penetrance in multigenerational pedigrees. Diabetes 57, 2511–2518 (2008).
Vaisse, C. et al. Melanocortin-4 receptor mutations are a frequent and heterogeneous cause of morbid obesity. J. Clin. Invest. 106, 253–262 (2000).
Schönke, M., Myers, M. G. Jr., Zierath, J. R. & Björnholm, M. Skeletal muscle AMP-activated protein kinase γ1H151R overexpression enhances whole body energy homeostasis and insulin sensitivity. Am. J. Physiol. Endocrinol. Metab. 309, E679–E690 (2015).
Pellinen, T. et al. Small GTPase Rab21 regulates cell adhesion and controls endosomal traffic of β1-integrins. J. Cell Biol. 173, 767–780 (2006).
Banerjee, U. & Cheng, X. Exchange protein directly activated by cAMP encoded by the mammalian rapgef3 gene: structure, function and therapeutics. Gene 570, 157–167 (2015).
Rippey, C. et al. Formation of chimeric genes by copy-number variation as a mutational mechanism in schizophrenia. Am. J. Hum. Genet. 93, 697–710 (2013).
Schmitz, C., Kinge, P. & Hutter, H. Axon guidance genes identified in a large-scale RNAi screen using the RNAi-hypersensitive Caenorhabditis elegans strain nre-1(hd20) lin-15b(hd126). Proc. Natl. Acad. Sci. USA 104, 834–839 (2007).
Setoguchi, R. et al. Repression of the transcription factor Th-POK by Runx complexes in cytotoxic T cell development. Science 319, 822–825 (2008).
Widom, R. L., Culic, I., Lee, J. Y. & Korn, J. H. Cloning and characterization of hcKrox, a transcriptional regulator of extracellular matrix gene expression. Gene 198, 407–420 (1997).
Sun, X. et al. Deletion of Atbf1/Zfhx3 in mouse prostate causes neoplastic lesions, likely by attenuation of membrane and secretory proteins and multiple signaling pathways. Neoplasia 16, 377–389 (2014).
Parsons, M. J. et al. The regulatory factor ZFHX3 modifies circadian function in SCN via an AT motif–driven axis. Cell 162, 607–621 (2015).
Balzani, E. et al. The Zfhx3-mediated axis regulates sleep and interval timing in mice. Cell Rep. 16, 615–621 (2016).
Kao, Y. H. et al. ZFHX3 knockdown increases arrhythmogenesis and dysregulates calcium homeostasis in HL-1 atrial myocytes. Int. J. Cardiol. 210, 85–92 (2016).
Auer, P. L., Reiner, A. P. & Leal, S. M. The effect of phenotypic outliers and non-normality on rare-variant association testing. Eur. J. Hum. Genet. 24, 1188–1194 (2016).
Goldstein, J. I. et al. zCall: a rare variant caller for array-based genotyping: genetics and population analysis. Bioinformatics 28, 2543–2545 (2012).
Grove, M. L. et al. Best practices and joint calling of the HumanExome BeadChip: the CHARGE Consortium. PLoS One 8, e68095 (2013).
Zhan, X., Hu, Y., Li, B., Abecasis, G. R. & Liu, D. J. RVTESTS: an efficient and comprehensive tool for rare variant association analysis using sequence data. Bioinformatics 32, 1423–1426 (2016).
Liu, D. J. et al. Meta-analysis of gene-level tests for rare variant association. Nat. Genet. 46, 200–204 (2014).
Winkler, T. W. et al. Quality control and conduct of genome-wide association meta-analyses. Nat. Protoc. 9, 1192–1212 (2014).
Winkler, T. W. et al. EasyStrata: evaluation and visualization of stratified genome-wide association meta-analysis data. Bioinformatics 31, 259–261 (2015).
Wu, M. C. et al. Rare-variant association testing for sequencing data with the sequence kernel association test. Am. J. Hum. Genet. 89, 82–93 (2011).
Price, A. L. et al. Pooled association tests for rare variants in exon-resequencing studies. Am. J. Hum. Genet. 86, 832–838 (2010).
Kiezun, A. et al. Exome sequencing and the genetic basis of complex traits. Nat. Genet. 44, 623–630 (2012).
Feng, S., Liu, D., Zhan, X., Wing, M. K. & Abecasis, G. R. RAREMETAL: fast and powerful meta-analysis for rare variants. Bioinformatics 30, 2828–2829 (2014).
Yang, J. et al. Genomic inflation factors under polygenic inheritance. Eur. J. Hum. Genet. 19, 807–812 (2011).
Willer, C. J., Li, Y. & Abecasis, G. R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010).
Thorleifsson, G. et al. Genome-wide association yields new sequence variants at seven loci that associate with measures of obesity. Nat. Genet. 41, 18–24 (2009).
Denny, J. C. et al. PheWAS: demonstrating the feasibility of a phenome-wide scan to discover gene–disease associations. Bioinformatics 26, 1205–1210 (2010).
Pers, T. H. et al. Biological interpretation of genome-wide association studies using predicted gene functions. Nat. Commun. 6, 5890 (2015).
Acknowledgements
A.P.R. was supported by R01DK089256. A.W.H. is supported by an NHMRC Practitioner Fellowship (APP1103329). A.K.M. received funding from NIH/NIDDK K01DK107836. A.T.H. is a Wellcome Trust Senior Investigator (WT098395) and an NIH Research Senior Investigator. A.P.M. is a Wellcome Trust Senior Fellow in Basic Biomedical Science (WT098017). A.R.W. is supported by the European Research Council (SZ-245 50371-GLUCOSEGENES-FP7-IDEAS-ERC). A.U.J. is supported by the American Heart Association (13POST16500011) and the NIH (R01DK089256, R01DK101855, K99HL130580). B.K. and E.K.S. were supported by the Doris Duke Medical Foundation, the NIH (R01DK106621), the University of Michigan Internal Medicine Department, Division of Gastroenterology, the University of Michigan Biological Sciences Scholars Program and the Central Society for Clinical Research. C.J.W. is supported by the NIH (HL094535, HL109946). D.J.L. is supported by R01HG008983 and R21DA040177. D.R.W. is supported by the Danish Diabetes Academy, which is funded by the Novo Nordisk Foundation. V. Salomaa has been supported by the Finnish Foundation for Cardiovascular Research. F.W.A. is supported by Dekker scholarship–Junior Staff Member 2014T001 Netherlands Heart Foundation and the UCL Hospitals NIHR Biomedical Research Centre. F.D. is supported by the UK MRC (MC_UU_12013/1-9). G.C.-P. received scholarship support from the University of Queensland and QIMR Berghofer. G.L. is funded by the Montreal Heart Institute Foundation and the Canada Research Chair program. H.Y. and T.M.F. are supported by the European Research Council (323195; SZ-245 50371-GLUCOSEGENES-FP7-IDEAS-ERC). I.M.H. is supported by BMBF (01ER1206) and BMBF (01ER1507m), the NIH and the Max Planck Society. J. Haessler was supported by NHLBI R21HL121422. J.N.H. is supported by NIH R01DK075787. K.E.N. was supported by the NIH (R01DK089256, R01HD057194, U01HG007416, R01DK101855) and the American Heart Association (13GRNT16490017). M.A.R. is supported by the Nuffield Department of Clinical Medicine Award, Clarendon Scholarship. M.I.M. is a Wellcome Trust Senior Investigator (WT098381) and an NIH Research Senior Investigator. M.D. is supported by the NCI (R25CA94880, P30CA008748). P.R.N. is supported by the European Research Council (AdG; 293574), the Research Council of Norway, the University of Bergen, the KG Jebsen Foundation and the Helse Vest, Norwegian Diabetes Association. P.T.E. is supported by the NIH (1R01HL092577, R01HL128914, K24HL105780), by an Established Investigator Award from the American Heart Association (13EIA14220013) and by the Foundation Leducq (14CVD01). P.L.A. was supported by NHLBI R21HL121422 and R01DK089256. P.L.H. is supported by the NIH (NS33335, HL57818). R.S.F. is supported by the NIH (T32GM096911). R.J.F.L. is supported by the NIH (R01DK110113, U01HG007417, R01DK101855, R01DK107786). S.A.L. is supported by the NIH (K23HL114724) and a Doris Duke Charitable Foundation Clinical Scientist Development Award. T.D.S. holds an ERC Advanced Principal Investigator award. T.A.M. is supported by an NHMRC Fellowship (APP1042255). T.H.P. received Lundbeck Foundation and Benzon Foundation support. V.T. is supported by a postdoctoral fellowship from the Canadian Institutes of Health Research (CIHR). Z.K. is supported by the Leenaards Foundation, the Swiss National Science Foundation (31003A-143914) and SystemsX.ch (51RTP0_151019). Part of this work was conducted using the UK Biobank resource (project numbers 1251 and 9072). A full list of acknowledgments appears in the Supplementary Note.
Author information
Authors and Affiliations
Consortia
Contributions
Writing group (wrote and edited the manuscript): A.E.J., A.E.L., C.M.L., C.S., G.L., H.M.H., J.N.H., K.E.N., K.L.Y., M.F.F., M. Graff, P.D., R.J.F.L., T.M.F., V.T., Y. Lu. Data preparation group (program development and quality control of data from contributing cohorts for meta-analyses): A.E.J., A.E.L., C.M.-G., C.S., D.J.L., E.M., H.M.H., I.B.B., K.L.Y., K.E.S., K.S.L., M.A.R., M.C.Y.N., M. Graff, N.G.D.M., P. Mudgal, R.J.F.L., S. Feng, S.M.W., S.S., S.V., T.A., T.E., T. Karaderi, T.W.W., V.T., X.Z., Y. Lu. BMI meta-analyses (discovery and follow-up, single variant and gene based): A.E.J., C.S., C.-T.L., D.J.L., H.M.H., I.B.B., J.N.H., K.L.Y., R.J.F.L., T.M.F., V.T., Y. Lu. Childhood data (analyses and interpretation): A.E.H., G.M., H.H., I. Barroso, I.S.F., J.P.B., S.F.A.G., V.M. Pleiotropy working group: A.M., C.J.W., C.M.L., D.J.L., E.M., F.D., G.A., G.M., G.M.P., H. Kitajima, H.M.H., J.C.F., J.P.C., J.R.B.P., J.W., K.S.R., M. Boehnke, M.I.M., P.B.M., P.D., R.J.F.L., S. Kathiresan, S.M.W., S.W., T.F.V., X.S. Phenome-wide association studies: A.G., A.M., J.C.D., L. Bastarache, M.I.M., T.L.E. Gene set enrichment analyses: D.L., J.N.H., R.S.F., S.B., T.H.P., Z.K. Monogenic and syndromic gene enrichment analyses: A.K.M., H.M.H. Fly obesity screen: A. Lempradl, J.A. Pospisilik. Oversight of contributing studies: A. Linneberg, A. Peters, A. Tönjes, A.C.H., A.D.M., A.G.U., A.I.d.H., A.J.L., A.M.D., A.P.M., A.P.R., A.S.B., A.T.H., A.W.H., B.B., B.G.N., C.A.B., C.C., C.E.P., C.H., C.J.W., C.L., C.M.L., C.N.A.P., D.F.E., D.F.R., D.I.C., D.M.W., D.O.M.-K., D.R.N., D.R.W., D.S., D.W.B., E.B., E.B.L., E.D.A., E.F., E.I., E.K.S., E.P.B., E.Z., F. Karpe, F. Kee, F. Renström, F.W.A., G. Dedoussis, G. Tromp, G.B., G.B.J., G.K.H., G.L., G.P., G.P.J., G.W.M., H. Kuivaniemi, H.B., H.D.W., H.H., H.-J.G., H.M.d.R., H.R.W., I. Barroso, I. Brandslund, I.B.B., I.F., I.J.D., I.M.H., I.R., I.S.F., J. Kaprio, J.C.C., J.-C.T., J.D., J.D.R., J.F., J.G.W., J.I.R., J.M.M.H., J.M.S., J.R.O’C., J.S.K., J.T., K. Stefansson, K. Strauch, K.E.N., K.K., K.K.A., K.L.M., K.M.H., K.N., K.R.O., K.T.Z., L.E.W., L.L., L.W., M. Blüher, M. Kähönen, M.A.I., M.A.R., M.B.S., M.C.H.d.G., M.d.H., M.E.J., M.F., M.H.B., M.I.M., M.L.O’D., M.M., M.P., M.-P.D., M.S., M.U., M.V., M.W., N.D.P., N.J.S., N.J.W., N.S., N.v.L., O. Pedersen, O. Polasek, O.T.R., P. Kovacs, P.A., P.A.P., P.B.M., P.D., P.E., P.G., P.G.-L., P.J.S., P.L.A., P.L.H., P.L.P., P.M.R., P.R.N., P.T.C., P.W.F., R.A.O., R.A.S., R.C., R.E.S., R.J.F.L., R.V., S. Fauser, S. Kathiresan, S.E.M., S.F.A.G., S.J., S.L.R.K., S.M., S.P., S.H.S., T.A.M., T.B.H., T.D.S., T.E., T.H., T.J., T.L., T.L.E., T.M.F., U.T., V. Gudnason, V. Salomaa, V.V., W.H.-H.S., X.G., X.L., Y. Liu. Genotyping of contributing studies: A. Loukola, A.T.-H., A.S.B., A.D’E., A.G.U., A.I.d.H., A.J.L., A.L.M., A.M., A.M.D., A.P.G., A.P.M., A.P.R., A.R.H., A.S.B., A.V., A.W.H., A.Y.C., B.G.N., C.A.B., C.E.P., C.H., C.J.P., C.K., C.L., C.M., C.M.-G., C.M.L., C.M.v.D., C.N.A.P., C.S., D.F.R., D.I.C., D.J.C., D.J.R., D.M.W., D.R.N., E.D.A., E.E., E.I., E.K., E.M.L., E.P.B., E.W.D., F. Karpe, F. Rivadeneira, F.S.C., G. Davies, G. Tromp, G.P.J., G.W.M., H. Kuivaniemi, H.H., H.L.G., H. Li, H.V., I.G., J. Kuusisto, J.A. Perry, J.B.-J., J.C.C., J.D., J.D.F., J.G.D., J.I.R., J.M.M.H., J.S.K., J.T., K. Strauch, K.D.T., K.E.S., K.M., L. Milani, L. Southam, L.A.L., L.A.K., L.M.Y.-A., L.-P.L., M. Benn, M. Boehnke, M. Gorski, M. Kähönen, M.B.S., M.C.H.d.G., M.F., M.H.B., M.I.M., M.L., M.L.B., M.L.G., M.L.O’D., M.M.-N., M.-P.D., M.P.S.-L., N.D.P., N.G., N.J.S., N.J.W., N.v.L., Ø.H., P.B.M., P.G.-L., P.I.W.d.B., P.T.C., P.W.F., R.A.O., R.A.S., R.E.S., R.F.-S., R.J.F.L., R.L.-G., R.R., R.Y., S. Kanoni, S. Kathiresan, S.C., S.F.A.G., S.F.N., S.H.V., S.L.R.K., S.S., S.W.v.d.L., T.A.L., T.B.H., T.E., T.H., T.L., U.V., V.V., Wei Zhao, X.L., Y. Lu, Y.-D.I.C., Y.H., Y. Liu, Y. Wang. Phenotyping of contributing studies: A.A.B., A.-E.F., A.F., A.J. Swift, A. Pattie, A. Peters, A.R.H., A. Robino, A.S.B., A.T.H., A. Tönjes, A.T.-H., A.U.J., A.V., A.W.H., B.B., B.G.N., B.H.T., B.K., C.A.B., C.E.L., C.E.P., C.H., C.J.P., C.K., C.M., C.M.-G., C.M.L., C.N.A.P., C.S., D.E., D.F.R., D.I.C., D.J.R., D.R.N., D.R.V.E., D.R.W., E.C., E.D.A., E.E., E.F., E.I., E.P.B., E.R.B.P., E.T., E.W.D., F. Karpe, F. Kee, F. Renström, F. Rivadeneira, F.W., G.B.J., G.L., G.P.J., G. Tromp, G.W.M., H.B., H.D.W., H.H., H.L.G., H. Li, I.J.D., I.R., J.C., J.C.C., J.D., J.D.F., J.F., J.-H.J., J. Kaprio, J. Lindström, J.M.M.H., J.M.S., J.P.B., J.S.K., K.E.N., K.-H.L., K.K., K.K.A., K.M.H., K.N., K.R.O., K.S.L., K.S.S., K.T.Z., L.A.K., L.E.B., L.E.W., L.L., L.M.Y.-A., L. Southam, L. Sun, L.W., M.A., M.A.I., M. Blüher, M. Brumat, M.C.H.d.G., M.F.F., M.I.M., M.J.C., M. Kähönen, M. Karaleftheri, M.L.B., M.M., M.M.-N., M.N., M.R., M.S., N.D.P., N.F., N.G., N.J.S., N.J.W., N.N., N.R.L., Ø.H., O.H.F., O.L.H., O. Polasek, O.R., O.T.R., P.A., P.G.-L., P. Komulainen, P. Kovacs, P.L.P., P.M.R., P. Mitchell, P.R.K., P.R.N., P.T.C., P.T.E., R.d.M., R.E.S., R.F.-S., R.M.-C., R.R., R.S.K., R.V., R.Y., S.A.L., S.E.M., S.F.A.G., S. Fauser, S.H.S., S.H.V., S.L.R.K., S.M., S.S., S.T., T.A.L., T.A.M., T.B.H., T.D.S., T.E.G., T.J., T.J.P., T.L., T.L.E., T.N.P., V. Giedraitis, V. Salomaa, V.T., W.H.-H.S., X.L., X.S., Y. Liu, Y. Lu. Data analysis of contributing studies: A.E.H., A.E.J., A.E.L., A.G., A.J.C., A.J. Slater, A. Lophatananon, A.M., A.P.M., A.P.P., A. Pirie, A.R.W., A. Rasheed, A. Robino, A.S.B., A. Teumer, A.V.S., A.Y.C., B.K., C.A.B., C.A.W., C.H., C.M.-G., C.P.N., C.S., C.T.H., C.-T.L., D.F.R., D.I.C., D.J.T., D.M.W., D.S.A., D.S.C., D.V., E.B.W., E.E., F. Rivadeneira, G.C.-P., G. Davies, G.L., G.M., G. Thorleifsson, G. Tromp, G.V., H. Li, H. Lin, H.M.S., H.P., H.R.W., H.T., H.Y., I.F., I.G., J.A. Perry, J.B.-J., J.C.C., J.C.G., J.E.H., J.G., J.G.D., J.H.Z., J. Haessler, J. Hernesniemi, J.I.R., J. Kuusisto, J. Li, J. Luan, J.M.M.H., J.P.B., J.P.T., J.R.O’C., J.S.K., J.v.S., J.W.J., J.Y., K.E.N., K.E.S., K.E.T., K.L.Y., K.M., K.S.L., L.-A.L., L.A.L., L. Broer, L.F.B., L.H., L.M.O.L., L.M.Y.-A., L. Moilanen, L.-P.L., L. Southam, M.A.N., M.C., M.C.H.d.G., M.C.Y.N., M.D., M.F., M.F.F., M. Gorski, M. Graff, M.L., M.P.S.-L., M.R., M.U., M.V., N.D.P., N.G., N.G.D.M., N.J.S., N.L.H.-C., N.R.R., N.v.L., Ø.H., P.L.A., P. Mudgal, P.S., P.Y., R.A.S., R.C., R.L.-G., R.U., R.Y., S.A.L., S.E.A., S.G., S.J., S.M., S.M.W., S.P., S.S., S.V., S.W.v.d.L., T.E., T. Karaderi, T. Korhonen, T.L.E., T.M.F., T.V.V., T.W.W., V.M., V. Steinthorsdottir, V.T., V.V., W.G., W. Zhang, W. Zhou, Wei Zhao, X.G., X.L., X.S., Y.H., Y.J., Y. Lu, Y.S., Y. Wu.
Corresponding authors
Ethics declarations
Competing interests
G. Thorleifsson, V. Steinthorsdottir, U.T. and K. Stefansson are employed by deCODE Genetics/Amgen, Inc.I. Barroso and spouse own stock in GlaxoSmithKline and Incyte, Ltd.S. Kathiresan has received grant support from Bayer Healthcare and Amarin; holds equity in San Therapeutics and Catabasis; and has received personal fees for participation in scientific advisory boards for Bayer Healthcare, Catabasis, Regeneron Genetics Center, Merck, Celera, Genomics PLC, Novartis, Sanofi, AstraZeneca, Alnylam, Eli Lilly Company, Leerink Partners, Noble Insights and Ionis Pharmaceuticals. A.Y.C., D.F.R. and T.F.V. are employees of Merck Sharp Dohme Corp., New Jersey, USA. D.I.C. receives genotyping and collaborative scientific support from Amgen and receives support for genetic analysis from AstraZeneca. P.M.R. receives genotyping and collaborative scientific support from Amgen and receives support for genetic analysis from AstraZeneca. M.J.C. is Chief Scientist for Genomics England, a UK government company.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–12 and Supplementary Note.
Supplementary Tables 1–26
Supplementary Tables 1–26.
Rights and permissions
About this article
Cite this article
Turcot, V., Lu, Y., Highland, H.M. et al. Protein-altering variants associated with body mass index implicate pathways that control energy intake and expenditure in obesity. Nat Genet 50, 26–41 (2018). https://doi.org/10.1038/s41588-017-0011-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-017-0011-x
- Springer Nature America, Inc.
This article is cited by
-
Incorporating functional annotation with bilevel continuous shrinkage for polygenic risk prediction
BMC Bioinformatics (2024)
-
Maternal dietary fat during lactation shapes single nucleus transcriptomic profile of postnatal offspring hypothalamus in a sexually dimorphic manner in mice
Nature Communications (2024)
-
EPAC1 enhances brown fat growth and beige adipogenesis
Nature Cell Biology (2024)
-
Socio-economic differences in body mass index: the contribution of genetic factors
International Journal of Obesity (2024)
-
Gene-SGAN: discovering disease subtypes with imaging and genetic signatures via multi-view weakly-supervised deep clustering
Nature Communications (2024)