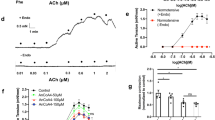
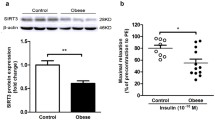
Abstract
Little is known about the contribution of the transient receptor potential canonical channel isoform 5 (TRPC5), a Ca2+-sensitive channel, to vasoconstriction in obesity. In this study, we found that the TRPC5 expression and carotid artery contraction of diet-induced obese (DIO) mice were significantly higher than those of wild-type mice. Endothelium-dependent vasocontraction was inhibited by the TRPC5 inhibitor clemizole and the knockout of TRPC5 in DIO mouse carotid arteries, while activation of TRPC5 enhanced contraction in wild-type mice. TRPC5-regulated vasocontraction can be inhibited by the ROS scavenger NAC and the COX-2 inhibitor NS-398. Our study suggested that upregulation of TRPC5 contributes to endothelium-dependent contraction, which is involved in ROS production and COX-2 expression in DIO mouse carotid arteries. From these results, we speculated that TRPC5 mediated endothelium-dependent contraction in the carotid artery of DIO mice, which was achieved by increasing the levels of ROS and COX-2 expression.






Similar content being viewed by others
References
Barton M, Baretella O, Meyer MR. Obesity and risk of vascular disease: importance of endothelium-dependent vasoconstriction. Br J Pharm. 2012;165:591–602.
Vanhoutte PM, Shimokawa H, Tang EHC, Feletou M. Endothelial dysfunction and vascular disease. Acta Physiologica. 2009;196:193–222.
Traupe T, Lang M, Goettsch W, Münter K, Morawietz H, Vetter W, et al. Obesity increases prostanoid-mediated vasoconstriction and vascular thromboxane receptor gene expression. J hypertension. 2002;20:2239–45.
Thébault S. Minireview: insights into the role of TRP channels in the retinal circulation and function. Neurosc Lett. 2021;765:136285.
Liang C, Zhang Y, Zhuo D, Lo CY, Yu L, Lau CW, et al. Endothelial cell transient receptor potential channel C5 (TRPC5) is essential for endothelium-dependent contraction in mouse carotid arteries. Biochem Pharm. 2019;159:11–24.
Li Z, Guo G, Wang H, Si X, Zhou G, Xiong Y, et al. TRPC5 channel modulates endothelial cells senescence. Eur J Pharmacol. 2017;802:27–35.
Lau OC, Shen B, Wong CO, Tjong YW, Lo CY, Wang HC, et al. TRPC5 channels participate in pressure-sensing in aortic baroreceptors. Nat Commun. 2016;7:11947.
Riccio A, Li Y, Moon J, Kim KS, Smith KS, Rudolph U, et al. Essential role for TRPC5 in amygdala function and fear-related behavior. Cell. 2009;137:761–72.
Zhang P, Sun C, Li H, Tang C, Kan H, Yang Z, et al. TRPV4 (transient receptor potential vanilloid 4) mediates endothelium-dependent contractions in the aortas of hypertensive mice. Hypertension. 2018;71:134–42.
Minard A, Bauer CC, Chuntharpursat-Bon E, Pickles IB, Wright DJ, Ludlow MJ, et al. Potent, selective, and subunit-dependent activation of TRPC5 channels by a xanthine derivative. Br J Pharm. 2019;176:3924–38.
Sun GY, Chuang DY, Zong Y, Jiang J, Lee JC, Gu Z, et al. Role of cytosolic phospholipase A2 in oxidative and inflammatory signaling pathways in different cell types in the central nervous system. Mol Neurobiol. 2014;50:6–14.
Liu Y, Xu Y, Thilo F, Friis UG, Jensen BL, Scholze A, et al. Erythropoietin increases expression and function of transient receptor potential canonical 5 channels. Hypertension. 2011;58:317–24.
Wong MS, Vanhoutte PM. COX-mediated endothelium-dependent contractions: from the past to recent discoveries. Acta Pharm Sin. 2010;31:1095–102.
Tang EH, Vanhoutte PM. Prostanoids and reactive oxygen species: team players in endothelium-dependent contractions. Pharm Ther. 2009;122:140–9.
Chen M, Xiang L, Wu G, Liao Y, Cai Y. Puerarin inhibits endothelium-dependent contractions in mouse carotid arteries. Med Sci Monit. 2020;26:e923163.
Zhou Y, Castonguay P, Sidhom E, Clark A, Dvela-Levitt M, Kim S, et al. A small-molecule inhibitor of TRPC5 ion channels suppresses progressive kidney disease in animal models. Science. 2017;358:1332–6.
Qi J, Cui J, Mi B, Yan X, Xu W, Ma H, et al. Isoliquiritigenin inhibits atherosclerosis by blocking TRPC5 channel expression. Cardiovasc Ther. 2020;2020:1926249.
Liu FF, Ma ZY, Li DL, Feng JB, Zhang K, Wang R, et al. Differential expression of TRPC channels in the left ventricle of spontaneously hypertensive rats. Mol Biol Rep. 2010;37:2645–51.
Acknowledgements
We thank Professor David E. Clapham (Harvard Medical School, USA) for giving us the TRPC5-knockout (TRPC5-/-) mice as a gift. We appreciate Dr Chunlei Tang (Jiangnan University, China) for assistance with the preparation of the AM237 compound.
Funding
This work was supported by the National Natural Science Foundation of China (81622007, 81960662, 82000291, and 82025005), the Chang Jiang Scholars Program (Q2015106), the Fundamental Research Funds for the Central Universities (JUSRP51704A), the National First-Class Discipline Program of Food Science and Technology (JUFSTR20180101), the Fundamental Research Funds for Young Scholars of Jiangnan University (JUSRP12046), and the Natural Science Foundation for Young Scholars of Jiangsu Province (BK20190596).
Author information
Authors and Affiliations
Contributions
The experimental design and operation of this study were mainly completed by YC and SW. The paper was written by YC and SW. YZ and XM guided the overall idea of the study. FY and KZ put forward valuable opinions on the writing of the article. XM provided a critical reading of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chu, Y., Wang, S., Zhu, Y. et al. TRPC5 mediates endothelium-dependent contraction in the carotid artery of diet-induced obese mice. Hypertens Res 45, 1945–1953 (2022). https://doi.org/10.1038/s41440-022-01017-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-022-01017-9
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
Endothelial cell Orai1 is essential for endothelium-dependent contraction of mouse carotid arteries in normotensive and hypertensive mice
Acta Pharmacologica Sinica (2024)
-
TRPC5 as a possible therapeutic target for vascular dysfunction associated with obesity
Hypertension Research (2022)