Abstract
Background
The immune response has important clinical value in colorectal cancer (CRC) in both prognosis and response to immunotherapy. This study aims to explore tumour immune cell infiltration in relation to clinically well-established molecular markers of CRC.
Methods
Multiplex immunohistochemistry and multispectral imaging was used to evaluate tumour infiltration of cytotoxic T cells (CD8+), Th1 cells (T-bet+), T regulatory cells (FoxP3+), B cells (CD20+), and macrophages (CD68+) in a cohort of 257 CRC patients.
Results
We found the expected association between higher immune-cell infiltration and microsatellite instability. Also, whereas BRAF-mutated tumours displayed increased immune-cell infiltration compared to BRAF wild-type tumours, the opposite was seen for KRAS-mutated tumours, differences that were most prominent for cytotoxic T cells and Th1 cells. The opposing relationships of BRAF and KRAS mutations with tumour infiltration of cytotoxic T cells was validated in an independent cohort of 608 CRC patients. A positive prognostic importance of cytotoxic T cells was found in wild-type as well as KRAS and BRAF-mutated CRCs in both cohorts.
Conclusion
A combined evaluation of MSI status, KRAS and BRAF mutational status, and immune infiltration (cytotoxic T cells) may provide important insights to prognosis and response to immunotherapy in CRC.
Similar content being viewed by others
Background
Colorectal cancer (CRC) is one of the major malignancies in the world and the second-leading cause of cancer deaths [1], medical advances notwithstanding. The basis for curative therapy is surgical resection, but still around 40% of the patients die from metastatic disease. In addition to the TNM staging system, the tumour immune response has been found an important prognosticator in CRC, with a higher density of tumour-infiltrating lymphocytes predicting a better outcome [2]. This finding has led to a joint effort to introduce the Immunoscore, based on immunohistochemical (IHC) evaluation of T cell markers, into clinical practice [3]. T cells are known combatants in anti-tumour immunity and can inhibit tumour growth by direct killing (cytotoxic T cells) [4]. The activity of the cytotoxic T cells is, however, tightly regulated by the balance of other T cell subsets; the T helper 1 (Th1) and T helper 2 (Th2) cells, and the T regulatory cells, as well as the expression of checkpoint inhibitors, e.g., CTLA4 and PD1/PDCD1 [5].
Considering the well-established prognostic value of the tumour immune response in CRC, these patients should be promising candidates for immunotherapy. However, the clinical response to immune checkpoint inhibitors has so far been quite limited in patients with metastatic CRC, the strongest effect found in patients with microsatellite instable (MSI) tumours [6]. The Food and Drug Administration has recently approved Pembrolizumab and Nivolumab (PD1 inhibitors) for clinical use in the treatment of metastatic MSI CRC patients, but ongoing studies suggest a striking beneficial effect of immune checkpoint inhibitor therapy also in non-metastatic MSI CRC [7, 8].
Sporadic CRC is a heterogenous disease, and the heterogeneity is partly seen in its development through three major genetic pathways: The classical chromosomal instability adenoma to carcinoma pathway responsible for approximately 70% of sporadic CRCs, the MSI pathway caused by aberrations in mismatch repair genes found in around 15% of CRCs, and the epigenetic instability CpG island methylator phenotype pathway causing 30–40% of sporadic CRCs. These pathways are partly driven by mutations in tumour oncogenes and are not mutually exclusive. Some of the most common driver mutations found in sporadic CRC are activating mutations in KRAS accounting for 30–40% of all CRCs and activating mutations in BRAF which occur in 5–15 % of the tumours [9].
Many robust studies have linked immune cell infiltration to an immunogenic MSI/d-MMR tumour status [4, 10]. However, the Immunoscore has been shown to be a stronger predictor of patient survival than tumour MSI status, suggesting that there are other subgroups of CRC patients in whom immune cell infiltration has clinical value [11]. Indeed, studies by us and others have proposed that there are microsatellite stable (MSS) tumours with a similar tumour immune profile as MSI tumours, e.g., the POLE mutated CRCs [11,12,13,14]. In addition, the expression of immune checkpoint proteins such as PD1 and PD-L1/CD274 are not restricted to MSI tumours [15, 16]. More comprehensive studies on the distribution of immune cell subsets according to KRAS and BRAF mutation are rare. KRAS and BRAF mutations are mainly mutually exclusive [17]. BRAF mutation has been associated with MSI tumours, but is also found in MSS tumours [18]. BRAF-mutated tumours have also been linked to a higher immune cell infiltration and expression of immunotherapeutic targets [19], but the relative contribution of mutated BRAF and MSI to the immune response remains uncertain. The potential of extracting putative predictive information from KRAS and BRAF mutation analyses in immunotherapy would be attractive since these are often already included, in addition to MSI status, in the clinical assessment of CRC patients.
This project aims to better characterise the local immune response to CRC in association with molecular characteristics. In particular, the importance of KRAS and BRAF mutation on immune cell infiltration will be evaluated. An increased understanding of the distribution of the immune response in different molecular subgroups of CRC may lead to the identification of prognostic and predictive tools, as well as new targets for therapy.
Materials and methods
Study cohort of patients with CRC
A logistic system has been developed for collecting biological samples and data from CRC patients diagnosed at Umeå University Hospital, Umeå, Sweden. Since 2010, CRC patients are enroled in U-CAN (Uppsala-Umeå Comprehensive Cancer Consortium) [20], which longitudinally collects blood, tissue, faeces, radiological data, and clinical data from all enroled patients. The present study utilised retrospectively collected resected formalin-fixed paraffin-embedded (FFPE) primary tumour tissue specimens from a cohort of stage I–IV U-CAN patients with faecal samples collected between the years 2010–2014 (n = 257), for studies of microbial markers in relation to molecular subtypes, immune cell infiltration and prognosis [21]. The patient tumour tissue specimens of this cohort have been analysed for molecular changes including BRAFV600E mutation and KRAS mutations (codon 12 and 13), and MSI status, as previously described [22]. Also data on clinicopathological characteristics, and survival have been collected. From this cohort a tumour tissue microarray (TMA) was prepared in September 2020, from patients operated on in Umeå with available tumour tissue at that time (n = 151). The TMA blocks were constructed using a TMA GRAND Master instrument (3DHISTEC, Budapest, Hungary) by punching 1 mm cores from archived FFPE tissue samples. The TMAs used for this study, included two cores of tumour tissue taken at the tumour front and one core taken from the tumour centre for each patient.
For validation, retrospectively collected tumour specimens from CRC patients identified within a population-based cohort from northern Sweden, the Northern Sweden Health and Disease Study (NSHDS), were used. The NSHDS cohort has been described in detail previously [23]. Patients diagnosed between the years 2000–2016 with available tumour tissue for analyses of molecular traits and immune infiltration were included in this study (n = 608). Clinical patient characteristics can be found in Supplementary Table S1.
Multispectral imaging for in situ evaluations of immune cell subsets in CRC
We evaluated immune cell infiltration using the Vectra® system for multispectral quantitative automated pathology imaging (Akoya Biosciences). The multiplexed IHC staining was a modification of the OpalTM 7 Solid Tumour Immunology Kit (Akoya Biosciences). Tissue TMA slides were sequentially stained using antibodies against T-bet, CD8, CD20, FoxP3, CD68 and pan-Cytokeratin. The CD4 antibody used in the kit was exchanged by T-bet (clone H210, Santa Cruz Biotechnology Inc.). The CD8 antibody in the kit was exchanged by CD8 (clone 144b, DAKO). The final concentrations of antibodies and Opal dyes used can be found in Supplementary Table S2. The Prolong Diamond Antifade Mountant (ThermoFisher, Waltham, MA, USA) was used to mount slides. Multispectral imaging was performed using the VECTRA 3 Quantitative Pathology Imaging System (Akoya Biosciences) as previously described [24]. A spectral library was created and applied to the images for spectral unmixing together with an autofluorescence control using the inForm® software (Akoya Biosciences, Marlborough, USA). The signal intensity of each marker was adjusted to give a signal range of 5–30 ms at exposure times of 30–200 ms. For validation, multiplex images were compared to monoplex images and inspected for interference and cross-talk. The inForm® software (Akoya Biosciences) was further used to quantify the different immune cells in tissue segmented areas using machine learning algorithms, as previously described [24]. T-bet was used as a marker for T helper 1 cells, but has been reported to be expressed also in cytotoxic (CD8 positive) T cells [25]. In this study, CD8 positive cells generally showed no or low expression of T-bet and were classified as cytotoxic T cells. Remaining T-bet positive cells were defined as T helper 1 cells. Tissue was segmented into tumour epithelial area, stromal area, tumour debris and no tissue. Extracellular mucin was classified as tumour debris. Exclusions were, large part of or whole core lost, lack of tumour or stromal tissue, bad quality of staining, and heavy necrosis. Image analysis was supervised by two experienced pathologists, and the investigators were blinded from patient data during the evaluations. Cell segmentation summary data was collected for each TMA core as number of individual cell types per mm2 (tumour or stromal compartment). For patients with data on two TMA cores, data was given as an average number of cells per mm2. Data was successfully collected from tumour front of 139 patients (of which 33 had data from only one core), and from tumour centre of 137 patients. Infiltrating immune cells, as identified by the respective markers, were further divided into groups of high and low infiltration with the median number of infiltrating cells for all CRC cases used as cut-off. Patients not surgically resected for CRC and patients dying from post-operative complications within 90 days of surgery were excluded from survival analyses.
Evaluation of cytotoxic T cells by immunohistochemistry for validation
The evaluation of cytotoxic T cells (CD8+) by immunohistochemistry in the NSHDS validation cohort has been previously described [23]. In brief, CD8+ cells were stained on whole tissue sections and semi-quantitatively evaluated at the tumour front of CRC specimens (n = 608) ranging from score (1) no or sporadic, (2) moderate numbers, (3) abundant or (4) highly abundant. Patients with pre-operatively irradiated rectal tumours were excluded (n = 98), leaving in total 510 CRC tumour specimens for analyses.
Statistics
IBM SPSS Statistics v.28 (SPSS Inc.) was used for statistical analyses. The distribution of each variable and subgroup was assessed and appropriate statistical tests chosen accordingly. Comparisons of differences within continuous variables between groups were hence performed by non-parametric two-sided Mann–Whitney U tests or Kruskall-Wallis H tests. The χ2 test or Fischer´s exact test (when expected counts were less than 5) was used for comparisons of categorical variables. Cancer-specific survival was defined as death with disseminated or recurrent disease and estimated using Kaplan–Meier survival analyses. Follow-up time was from day of surgery until death or end of follow-up (October 2021 for U-CAN, May 2022 for NSHDS). Median follow-up time was 92.8 months for U-CAN and 94.5 months for NSHDS. Log-rank tests were used to estimate differences in 5-year cancer-specific survival between groups. The Cox Proportional-Hazards model was used for uni- and multivariable survival analyses. A P value < 0.05 was considered statistically significant.
Results
Distribution of different immune cells in tumour front and centre of CRC patients
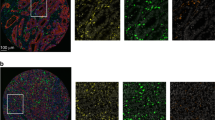
We have set up a functional assay for multiplex immunohistochemistry and evaluation of CD8+ cytotoxic T cells, T-bet+ Th1 cells, FoxP3+ T regulatory cells, CD20+ B cells, CD68+ macrophages, and cytokeratin using the VECTRA system for multispectral imaging (Fig. 1). We have evaluated this immune cell infiltration panel in surgically resected stage I–IV patients from a CRC patient study cohort (n = 257). The different immune cells were evaluated in the stromal compartment and in the tumour compartment at the tumour front and in the tumour centre. The infiltrating immune cells were shown to be quite evenly distributed between the tumour front and the tumour centre, but with higher levels in the stromal area compared to the tumour epithelial area (Table 1). Data from the tumour epithelial area for FoxP3+ T regulatory cells and CD20+ B cells was not further analysed since these immune cells were rarely present within this compartment.
Shown are high-magnification areas (20x) of representative multiplex IHC stains from (a) a TMA (1 mm core) taken at the tumour front and (b) a TMA (1 mm core) taken at the tumour centre. Left panel displays multiplex IHC images after spectral unmixing, of cytokeratin (magenta), CD8+ cytoxic T cells (red), T-bet+ Th1 cells (white), FoxP3+ T regulatory cells (orange), CD20+ B cells (yellow), and CD68+ macrophages (cyan). Right panel displays the same multiplex IHC but with tumour tissue segmentation map of stromal area (blue), tumour epithelial area (magenta), tumour debris (yellow) and no tissue (brown), and the cell phenotypes from which data was collected highlighted using the above described colours.
Analyses of immune cell infiltration according to patient clinical and pathological characteristics
The strongest associations of immune cells to clinical and pathological parameters in study patients were found at the tumour front (Table 2). A slightly higher infiltration of immune cells was found in women, especially within the tumour epithelial area. Immune cell infiltration was also strongly linked to the right-sided colon cancers, except for B cells and macrophages. All immune cell types were found linked to tumour stage, with a gradual decrease in immune cell infiltration from stage I to stage IV tumours (Table 2). Pre-operative radiotherapy of rectal cancers was strongly associated with a decreased infiltration of immune cells. Thus, for further analyses, the pre-operatively irradiated rectal cancers were excluded. The clinical and pathological associations of immune cell infiltration in the tumour centre can be found in Supplementary Table S3.
Analyses of immune cell infiltration in molecular subgroups of CRC
Infiltrating immune cells at the tumour front of study patients further exhibited several significant associations with tumour molecular subtypes (Table 3). The MSI tumours were found to be significantly more infiltrated by cytotoxic T cells, Th1 cells, and macrophages, in validation of our analyses. We found contrasting associations of immune cell infiltration in KRAS-mutated and BRAF-mutated CRC tumours. While KRAS-mutated tumours showed less infiltration of immune cells compared to wild-type KRAS tumours, in particular cytotoxic T cells and Th1 cells, BRAF-mutated tumours showed a higher infiltration of these immune cells compared to wild-type BRAF tumours. No associations to KRAS or BRAF mutation was found for T regulatory cells at the tumour front. The associations of immune cells in the tumour centre to molecular characteristics can be found in Supplementary Table S4. Similar, but weaker associations were found in the tumour centre. However, in the tumour centre KRAS mutation was significantly associated with increased infiltration of regulatory T cells (Supplementary Table S4).
To investigate the relative contribution of BRAF mutation and MSI status on immune cell infiltration, we stratified the analysis of immune cell infiltration according to BRAF mutation by MSI status (Table 3). Even though limited by sample size, BRAF mutation was found to increase infiltration of cytotoxic T cells in both MSI and MSS tumours to similar levels. In contrast, Th1 cell infiltration appeared to be more linked to MSI status than BRAF mutation. However, BRAF mutation was still linked to increased infiltration of most analysed immune cells (with the exception of regulatory T cells) in the MSS tumours.
In validation analyses, tumour infiltration of cytotoxic T cells was evaluated in an independent cohort of CRC patients, the NSHDS (n = 608). Also here, a contrasting role on immune cell infiltration by KRAS and BRAF mutation was found. The proportion of KRAS mutated tumours was found to decrease with increasing score of infiltrating cytotoxic T cells, while the proportion of BRAF mutated tumours instead was found to increase (Table 4). The proportion of MSI tumours was also found to increase with increasing immune score (Table 4). However, even though MSI status was found highly associated with a high immune score, around half of the tumours with the highest immune score were defined as MSS (Table 4). In this cohort where infiltration of cytotoxic T cells was assessed on a semi-quantitative scale (score 1-4), immune cell infiltration by BRAF mutation was found mainly linked to MSI tumours (Table 4).
Analyses of immune cell infiltration in CRC prognosis
A positive prognostic effect of immune cell infiltration in CRC tumours has been established in the literature. In this study, even though on a relatively small patient cohort, we also found a significant positive prognostic role for stromal infiltration of cytotoxic T cells, Th1 cells and T regulatory cells at the tumour front of stage I–IV CRC patients, with the strongest association found for cytotoxic T cells (P = 0.009) (Supplementary Fig. S1). The prognostic significance remained in stage I-III CRC patients for cytotoxic T cells and T regulatory cells (P = 0.047 and P = 0.014, respectively). Stratifying the prognostic role of stromal infiltration of cytotoxic T cells by KRAS or BRAF mutation status in both the study cohort and the NSHDS validation cohort, revealed a prognostic role of cytotoxic T cells independent of KRAS and BRAF mutation in CRC (Fig. 2). The independent prognostic role of cytotoxic T cells was further tested in a multivariable Cox Proportional-Hazards model in stage I-III patients from the NSHDS cohort. The positive prognostic role of cytotoxic T cells remained statistically significant in a multivariable analysis including gender, age, localisation, tumour stage, BRAF mutation, KRAS mutation, and MSI status (Supplementary Table S5).
Shown are Kaplan–Meier plots of cancer-specific survival in patients with different levels of stromal infiltration of cytotoxic T cells (Tcyt) in wild-type/wild-type tumours, KRAS mutant tumours, and BRAF mutant tumours, as indicated, in (a) the study cohort, and in (b) the NSHDS validation cohort. Pre-operatively irradiated rectal cancers were excluded. Log-rank tests were used to calculate differences in 5-year survival between groups. P < 0.05 was considered statistically significant.
Discussion
The immune response has important clinical value in both prognosis and therapy of CRC patients. Immune checkpoint inhibitors have proven very effective in some patients with metastatic immunogenic MSI tumours, but there are likely additional CRC patient groups where immunotherapy may have clinical value. CRC is a heterogeneous disease, which calls for careful investigations of the immune response in molecular subgroups of CRC to identify potential predictive markers of response to immunotherapy. Here, we investigated the infiltration of different immune cell subsets in relation to molecular characteristics of CRC, with a focus on the clinically well-established molecular markers KRAS and BRAF mutation.
We analysed the tumour infiltration of five different immune cells (cytotoxic T cells, Th1 cells, regulatory T cells, B cells and macrophages), both in the tumour centre and at the tumour front of patients with CRC. Immune cells were quite evenly distributed between the tumour centre and tumour front, however the associations to clinicopathological and tumour molecular traits were found to be stronger at the tumour front. We further investigated both the stromal area and the tumour epithelial area, noticing a higher density of immune cells in the stromal compartment, a finding in line with a recent study by Mezheyeuski et al., where they used a similar approach to characterise the immune landscape in CRC [26]. These results support the analysis of immune cell infiltration in TMAs taken at the tumour front as a suitable approach in CRC.
Right-sided colon cancers were more highly infiltrated by different subsets of T cells at the tumour front, a higher immune cell infiltration being a recognised feature of these tumours [27]. We further found a decreasing infiltration of all immune cells analysed from stage I to stage IV tumours, findings that may be related to tumour immune escape and metastasis. Pre-operatively radiated rectal tumours displayed a significantly lower infiltration of all immune cells analysed, which is in line with the reported radiosensitivity of lymphocytic cells [28]. Even though being an interesting patient group, they should likely be analysed separately as pre-operative radiotherapy may alter the anti-tumour immune response, introducing e.g., damage associated molecular patterns and rediscovery of tumour antigens [29]. Pre-operative radiotherapy has so far shown contradictory results in CRC, with both enhanced and reduced immune cell infiltration, calling for further investigation [30,31,32].
When analysing immune cell infiltration in association with known molecular parameters of CRC tumours, we found the expected and well-established associations of immune cells to MSI tumours [4, 10]. Immunogenomic studies have, however, shown that not only hypermutated tumours (MSI or POLE mutated) but also some non-hypermutated MSS tumours display high neoantigen load and tumour immune cell infiltration [33,34,35]. Interestingly, we here found an opposing role of KRAS and BRAF mutation on the immune response in CRC. While KRAS-mutated tumours displayed decreased immune cell infiltration compared to KRAS wild-type tumours, the opposite was found for BRAF-mutated tumours, differences that were most obvious for the cytotoxic T cells and the Th1 cells. For cytotoxic T cells, the findings were validated in the NSHDS cohort. A contrasting role of KRAS and BRAF mutation on immune cell infiltration in CRC tumours has been previously suggested [36, 37]. Ling et al. found a similar contrasting infiltration of Th1 cells according to KRAS and BRAF mutation in CRC [37], and Lal et al. found that KRAS- and BRAF-mutated CRCs were found in clusters with low and high expression of immune response genes, respectively [36]. KRAS-mutated CRCs have in previous studies been associated with supressed immune pathways and decreased immune cell infiltration [38,39,40]. BRAF mutation has been linked to the immunogenic MSI tumours [27], but our findings partly suggest that BRAF and MSI may to some extent have individual effects on the immune response in CRC. Further larger studies are needed to establish the role of BRAF mutation on immune cell infiltration in CRC. In a study by Cen et al., where they compared the immune microenvironment in BRAF-mutated tumours to that of BRAF wild-type tumours, an increased immune cell infiltration was found along with an increased expression of immune checkpoints, e.g., PD1, PD-L1 and CTLA4 [19]. Bolzacchini et al. further identified a gene expression based “hot/inflamed” immunoprofile in a high fraction (52%) of patients with advanced BRAF-mutant CRC, with only a partial overlap with MSI [41]. Additionally, 42% of the MSI tumours showed a “cold” immunoprofile. Barras et al. also suggested different subtypes of BRAF-mutant CRCs based on gene expression, finding differences in immune regulating genes as well as patient survival, regardless of MSI, suggesting that the BRAF-mutated tumours are not uniform [42]. Further studies dissecting the immune response to BRAF-mutated tumours in MSI as well as MSS CRCs, including also analyses of immune checkpoint molecules, are needed to find potential predictive markers for immunotherapy. For patients with BRAF-mutated MSI CRCs, combined treatment with immune checkpoint inhibitors and targeted therapies are currently under clinical evaluation. In a clinical trial, Pembrolizumab as first line treatment of advanced MSI metastatic CRC showed good efficacy regardless of BRAF mutation [43]. However, a decreased benefit of Pembrolizumab treatment was found in KRAS- or NRAS-mutated metastatic MSI patients [43]. Furthermore, combination therapy with Nivolumab and Ipilimumab (CTLA4 inhibitor), showed evident beneficial effects, especially for patients with BRAF-mutated tumours [44]. Interestingly, even though limited by few patients, an improved clinical response to Pembrolizumab was found in patients with BRAF-mutated treatment-resistant metastatic MSS CRCs [45]. Importantly, our findings show that there are highly immune infiltrated MSS tumours (being the majority of CRCs), which should be further explored in terms of possible implications for immunotherapy. One limitation of this study was that immune analyses were restricted to patients who underwent primary surgery. The potential predictive value of immune markers needs to be further assessed also in the neo-adjuvant setting using larger patient cohorts. In fact, pathological responses have been shown for neo-adjuvant immunotherapy in both MSI and MSS early-stage cancers [46].
Mechanistic insights on the regulation of immunity according to KRAS and BRAF mutation are limited. In a study by Liao et al. KRAS mutation was found to direct the tumour recruitment of myeloid derived suppressor cells through the IRF2-CXCL3-CXCR2 axis leading to immune therapy resistance in CRC [39]. In a previous in vitro study comparing the cytokine profile of KRAS mutant, BRAF mutant, and BRAF/KRAS wild-type Caco2 CRC cells, we found an increased expression of a Th1 related cytokine (CXCL10), and decreased expression of Th2 related cytokines (CCL2 and TGFB1) in BRAF-mutated cells, while the opposite pattern was shown for KRAS-mutated cells [37]. The results were confirmed in tumour tissues and linked to infiltration of Th1 cells, suggesting that these mutations may have different and direct effects on the immune response to CRC.
In this study, we further analysed the prognostic role of immune cell infiltration in CRC tumours and found a significantly improved survival for patients with a high number of infiltrating T cell subsets (the strongest effect seen with cytotoxic T cells). The prognostic importance of cytotoxic T cells and regulatory T cells remained also in stage I-III patients.
KRAS mutation has been associated with a poor prognosis in CRC [47, 48], and so has BRAF mutation, irrespective of MSI status, with BRAF-mutated MSS tumours showing the worst prognosis [18]. Here, survival analyses of infiltrating cytotoxic T cells at the tumour front showed a strong prognostic role for cytotoxic T cells in CRC independent of KRAS- and BRAF-mutational status. Further studies are needed to understand the prognostic importance of immune cell infiltration in these molecular subgroups of CRC.
The strengths of this study include the quantitative evaluation of multiple immune cells in different tumour locations of patients with CRC, comprehensive immune evaluations on a protein-based level being rare and an important complement to the immunogenomic studies. We were also able to validate our main findings in an independent larger cohort of CRC patients. Weaknesses of this study include small sample sizes in the primary patient cohort and the possibility that the use of TMAs do not fully reflect the heterogenous distribution of tumour infiltrating immune cells within the whole tumour. Furthermore, the analyses in this study were restricted to BRAFV600E mutation and KRAS mutations in codon 12 and 13. Even though covering the majority of KRAS and BRAF mutations in CRC, further studies taking additional mutations into account are needed.
Collectively, our findings suggest that combined evaluation of MSI status, KRAS and BRAF mutation, and immune cell infiltration (cytotoxic T cells) may provide added value compared to each variable alone and might have potential to help guide decisions on immunotherapy in CRC. In future management of CRC patients, individualised therapy taking both the molecular characteristics of the tumour and the tumour immune response into consideration will be of importance. An increased understanding of the actions of the immune response in molecular subgroups of CRC may lead to significant advances in personalised medicine, including the identification of important prognostic and predictive tools, as well as new targets for therapy.
Data availability
The data presented in this study are available on reasonable request from the corresponding author.
References
Morgan E, Arnold M, Gina A, Lorenzoni V, Cabasag ML, Laversanne M, et al. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut. 2023;72:228–344.
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science. 2006;313:1960–4.
Galon J, Mlecnik B, Bindea G, Angell HK, Berger A, Lagorce C, et al. Towards the introduction of the ‘Immunoscore’ in the classification of malignant tumours. J Pathol. 2014;232:199–209.
Deschoolmeester V, Baay M, Lardon F, Pauwels P, Peeters M. Immune cells in colorectal cancer: prognostic relevance and role of MSI. Cancer Microenviron. 2011;4:377–92.
Palucka AK, Coussens LM. The basis of oncoimmunology. Cell. 2016;164:1233–47.
Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357:409–13.
Poh A. A wow for Neoadjuvant ICI in dMMR colon cancer. Cancer Discov. 2022;12:2486–7.
Cercek A, Lumish M, Sinopoli J, Weiss J, Shia J, Lamendola-Essel M, et al. PD-1 blockade in mismatch repair-deficient, locally advanced rectal cancer. N. Engl J Med. 2022;386:2363–76.
Baldus SE, Schaefer KL, Engers R, Hartleb D, Stoecklein NH, Gabbert HE. Prevalence and heterogeneity of KRAS, BRAF, and PIK3CA mutations in primary colorectal adenocarcinomas and their corresponding metastases. Clin Cancer Res. 2010;16:790–9.
Randrian V, Evrard C, Tougeron D. Microsatellite instability in colorectal cancers: carcinogenesis, neo-antigens, immuno-resistance and emerging therapies. Cancers. 2021;13:3063.
Mlecnik B, Bindea G, Angell HK, Maby P, Angelova M, Tougeron D, et al. Integrative analyses of colorectal cancer show immunoscore is a stronger predictor of patient survival than microsatellite instability. Immunity. 2016;44:698–711.
Domingo E, Freeman-Mills L, Rayner E, Glaire M, Briggs S, Vermeulen L, et al. Somatic POLE proofreading domain mutation, immune response, and prognosis in colorectal cancer: a retrospective, pooled biomarker study. Lancet Gastroenterol Hepatol. 2016;1:207–16.
Li X, Ling A, Kellgren TG, Lundholm M, Lofgren-Burstrom A, Zingmark C, et al. A detailed flow cytometric analysis of immune activity profiles in molecular subtypes of colorectal cancer. Cancers. 2020;12:3440.
Nebot-Bral L, Brandao D, Verlingue L, Rouleau E, Caron O, Despras E, et al. Hypermutated tumours in the era of immunotherapy: the paradigm of personalised medicine. Eur J Cancer. 2017;84:290–303.
Ahtiainen M, Wirta EV, Kuopio T, Seppala T, Rantala J, Mecklin JP, et al. Combined prognostic value of CD274 (PD-L1)/PDCDI (PD-1) expression and immune cell infiltration in colorectal cancer as per mismatch repair status. Mod Pathol. 2019;32:866–83.
Lee LH, Cavalcanti MS, Segal NH, Hechtman JF, Weiser MR, Smith JJ, et al. Patterns and prognostic relevance of PD-1 and PD-L1 expression in colorectal carcinoma. Mod Pathol. 2016;29:1433–42.
Rajagopalan H, Bardelli A, Lengauer C, Kinzler KW, Vogelstein B, Velculescu VE. Tumorigenesis: RAF/RAS oncogenes and mismatch-repair status. Nature. 2002;418:934.
Lochhead P, Kuchiba A, Imamura Y, Liao X, Yamauchi M, Nishihara R, et al. Microsatellite instability and BRAF mutation testing in colorectal cancer prognostication. J Natl Cancer Inst. 2013;105:1151–6.
Cen S, Liu K, Zheng Y, Shan J, Jing C, Gao J, et al. BRAF mutation as a potential therapeutic target for checkpoint inhibitors: a comprehensive analysis of immune microenvironment in BRAF mutated colon cancer. Front Cell Dev Biol. 2021;9:705060.
Glimelius B, Melin B, Enblad G, Alafuzoff I, Beskow A, Ahlstrom H, et al. U-CAN: a prospective longitudinal collection of biomaterials and clinical information from adult cancer patients in Sweden. Acta Oncol. 2018;57:187–94.
Lowenmark T, Lofgren-Burstrom A, Zingmark C, Eklof V, Dahlberg M, Wai SN, et al. Parvimonas micra as a putative non-invasive faecal biomarker for colorectal cancer. Sci Rep. 2020;10:15250.
Lowenmark T, Lofgren-Burstrom A, Zingmark C, Ljuslinder I, Dahlberg M, Edin S, et al. Tumour colonisation of parvimonas micra Is associated with decreased survival in colorectal cancer patients. Cancers. 2022;14:5937.
Renman D, Gylling B, Vidman L, Boden S, Strigard K, Palmqvist R, et al. Density of CD3(+) and CD8(+) cells in the microenvironment of colorectal cancer according to prediagnostic physical activity. Cancer Epidemiol Biomark Prev. 2021;30:2317–26.
Edin S, Kaprio T, Hagstrom J, Larsson P, Mustonen H, Bockelman C, et al. The prognostic importance of CD20(+) B lymphocytes in colorectal cancer and the relation to other immune cell subsets. Sci Rep. 2019;9:19997.
Sullivan BM, Juedes A, Szabo SJ, von Herrath M, Glimcher LH. Antigen-driven effector CD8 T cell function regulated by T-bet. Proc Natl Acad Sci USA. 2003;100:15818–23.
Mezheyeuski A, Micke P, Martin-Bernabe A, Backman M, Hrynchyk I, Hammarstrom K, et al. The immune landscape of colorectal cancer. Cancers. 2021;13:5545.
Inamura K. Colorectal cancers: an update on their molecular pathology. Cancers. 2018;10:26.
Manda K, Glasow A, Paape D, Hildebrandt G. Effects of ionizing radiation on the immune system with special emphasis on the interaction of dendritic and T cells. Front Oncol. 2012;2:102.
Carvalho HA, Villar RC. Radiotherapy and immune response: the systemic effects of a local treatment. Clinics. 2018;73:e557s.
Lim SH, Chua W, Cheng C, Descallar J, Ng W, Solomon M, et al. Effect of neoadjuvant chemoradiation on tumor-infiltrating/associated lymphocytes in locally advanced rectal cancers. Anticancer Res. 2014;34:6505–13.
Matsutani S, Shibutani M, Maeda K, Nagahara H, Fukuoka T, Nakao S, et al. Significance of tumor-infiltrating lymphocytes before and after neoadjuvant therapy for rectal cancer. Cancer Sci. 2018;109:966–79.
Sakuyama N, Kojima M, Kawano S, Akimoto T, Saito N, Ito M, et al. Histological differences between preoperative chemoradiotherapy and chemotherapy for rectal cancer: a clinicopathological study. Pathol Int. 2016;66:273–80.
Angelova M, Charoentong P, Hackl H, Fischer ML, Snajder R, Krogsdam AM, et al. Characterization of the immunophenotypes and antigenomes of colorectal cancers reveals distinct tumor escape mechanisms and novel targets for immunotherapy. Genome Biol. 2015;16:64.
Giannakis M, Mu XJ, Shukla SA, Qian ZR, Cohen O, Nishihara R, et al. Genomic correlates of immune-cell infiltrates in colorectal carcinoma. Cell Rep. 2016;17:1206.
van den Bulk J, Verdegaal EME, Ruano D, Ijsselsteijn ME, Visser M, van der Breggen R, et al. Neoantigen-specific immunity in low mutation burden colorectal cancers of the consensus molecular subtype 4. Genome Med. 2019;11:87.
Lal N, Beggs AD, Willcox BE, Middleton GW. An immunogenomic stratification of colorectal cancer: implications for development of targeted immunotherapy. Oncoimmunology. 2015;4:e976052.
Ling A, Lundberg IV, Eklof V, Wikberg ML, Oberg A, Edin S, et al. The infiltration, and prognostic importance, of Th1 lymphocytes vary in molecular subgroups of colorectal cancer. J Pathol Clin Res. 2016;2:21–31.
Lal N, White BS, Goussous G, Pickles O, Mason MJ, Beggs AD, et al. KRAS mutation and consensus molecular Subtypes 2 and 3 are independently associated with reduced immune infiltration and reactivity in colorectal cancer. Clin Cancer Res. 2018;24:224–33.
Liao W, Overman MJ, Boutin AT, Shang X, Zhao D, Dey P, et al. KRAS-IRF2 axis drives immune suppression and immune therapy resistance in colorectal cancer. Cancer Cell. 2019;35:559–72.e7.
Liu J, Huang X, Liu H, Wei C, Ru H, Qin H, et al. Immune landscape and prognostic immune-related genes in KRAS-mutant colorectal cancer patients. J Transl Med. 2021;19:27.
Bolzacchini E, Libera L, Church SE, Sahnane N, Bombelli R, Digiacomo N, et al. Tumor antigenicity and a pre-existing adaptive immune response in advanced BRAF mutant colorectal cancers. Cancers. 2022;14:3951.
Barras D, Missiaglia E, Wirapati P, Sieber OM, Jorissen RN, Love C, et al. BRAF V600E mutant colorectal cancer subtypes based on gene expression. Clin Cancer Res. 2017;23:104–15.
Andre T, Shiu KK, Kim TW, Jensen BV, Jensen LH, Punt C, et al. Pembrolizumab in microsatellite-instability-high advanced colorectal cancer. N. Engl J Med. 2020;383:2207–18.
Andre T, Lonardi S, Wong KYM, Lenz HJ, Gelsomino F, Aglietta M, et al. Nivolumab plus low-dose ipilimumab in previously treated patients with microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: 4-year follow-up from CheckMate 142. Ann Oncol. 2022;33:1052–60.
Wang C, Sandhu J, Ouyang C, Ye J, Lee PP, Fakih M. Clinical response to immunotherapy targeting programmed cell death receptor 1/programmed cell death Ligand 1 in patients with treatment-resistant microsatellite stable colorectal cancer with and without liver metastases. JAMA Netw Open. 2021;4:e2118416.
Chalabi M, Fanchi LF, Dijkstra KK, Van den Berg JG, Aalbers AG, Sikorska K, et al. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat Med. 2020;26:566–76.
Imamura Y, Morikawa T, Liao X, Lochhead P, Kuchiba A, Yamauchi M, et al. Specific mutations in KRAS codons 12 and 13, and patient prognosis in 1075 BRAF wild-type colorectal cancers. Clin Cancer Res. 2012;18:4753–63.
Modest DP, Ricard I, Heinemann V, Hegewisch-Becker S, Schmiegel W, Porschen R, et al. Outcome according to KRAS-, NRAS- and BRAF-mutation as well as KRAS mutation variants: pooled analysis of five randomized trials in metastatic colorectal cancer by the AIO colorectal cancer study group. Ann Oncol. 2016;27:1746–53.
Acknowledgements
The authors are grateful to the patients who participated in the study.
Funding
This study was supported by the Swedish Cancer Society under Grant 20-1271 PjF; Lion´s Cancer Research Foundation under Grant LP 21-2265; the Cancer Research Foundation in Northern Sweden under Grant AMP 16-798; the County Council of Västerbotten under Grants Palmqvist, 2021 and Ling, 2021; the Sjöberg Foundation under Grant Palmqvist, 2021; and the Faculty of Medicine under Grant Interdisciplinary and innovative research, 2021. Open access funding provided by Umea University.
Author information
Authors and Affiliations
Contributions
Study concept and design: SE, BG, XL, BVG, IL, AL, RP; acquisition of data: SE, BG, ÅS, ALB, CZ, BVG, IL, AL, RP; data analyses: SE, BG, AL; interpretation of data: SE, BG, XL, BVG, AL, RP; draughting of the manuscript: SE, BG, AL; critical revision of the manuscript for important intellectual content: SE, BG, XL, ÅS, ALB, CZ, BVG, IL, AL, RP. All authors approved the final version of the manuscript and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was performed in accordance with the Declaration of Helsinki and the ethical committee of Umeå University/Regional Ethical Review Board has approved all parts of this application including the procedure whereby the patients gave written informed consent (dnr 2014/321-31, dnr 2016/219-31, and dnr 2021-07029-01).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Edin, S., Gylling, B., Li, X. et al. Opposing roles by KRAS and BRAF mutation on immune cell infiltration in colorectal cancer – possible implications for immunotherapy. Br J Cancer 130, 143–150 (2024). https://doi.org/10.1038/s41416-023-02483-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-023-02483-9
- Springer Nature Limited
This article is cited by
-
The long-term effectiveness and mechanism of oncolytic virotherapy combined with anti-PD-L1 antibody in colorectal cancer patient
Cancer Gene Therapy (2024)
-
First-line oxaliplatin-based chemotherapy and nivolumab for metastatic microsatellite-stable colorectal cancer—the randomised METIMMOX trial
British Journal of Cancer (2024)