Abstract
T-cell acute lymphoblastic leukemia (T-ALL) is an aggressive hematological malignancy characterized by an accumulation of immature T cells. Although patient outcomes have improved, novel targeted therapies are needed to reduce the intensity of chemotherapy and improve the prognosis of high-risk patients. Interleukin-7 (IL-7) modulates the survival and proliferation of normal and malignant T cells. Targeting the IL-7 signaling pathway is thus a potentially effective therapeutic strategy. To achieve such aim, it is essential to first understand how the IL-7 signaling pathway is activated. Although IL-7 production has been observed from multiple stromal tissues, T-ALL autocrine IL-7 secretion has not yet been described. Interestingly, using T-ALL cell lines, primary and patient-derived xenotransplanted (PDX) T-ALL cells, we demonstrate that T-ALL cells produce IL-7 whereas normal T cells do not. Finally, using knock down of IL7 gene in T-ALL cells, we describe to what extent IL-7 autocrine secretion is involved in the T-ALL cells propagation in bone marrow and how it affects the number of leukemia-initiating cells in PDX mice. Together, these results demonstrate how the autocrine production of the IL-7 cytokine mediated by T-ALL cells can be involved in the oncogenic development of T-ALL and offer novel insights into T-ALL spreading.




Similar content being viewed by others
Change history
01 August 2019
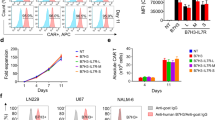
The original HTML version of this Article was updated after publication because the authors noted that panels D and E had been omitted from Figure 2. The Figure has been updated in the PDF and HTML versions of the article.
References
Girardi T, Vicente C, Cools J, De Keersmaecker K. The genetics and molecular biology of T-ALL. Blood. 2017;129:1113–23.
Hunger SP, Mullighan CG. Acute lymphoblastic leukemia in children. N Engl J Med. 2015;373:1541–52.
Marks DI, Paietta EM, Moorman AV, Richards SM, Buck G, DeWald G, et al. T-cell acute lymphoblastic leukemia in adults: clinical features, immunophenotype, cytogenetics, and outcome from the large randomized prospective trial (UKALL XII/ECOG 2993). Blood. 2009;114:5136–45.
Freyer DR, Devidas M, La M, Carroll WL, Gaynon PS, Hunger SP, et al. Postrelapse survival in childhood acute lymphoblastic leukemia is independent of initial treatment intensity: a report from the Children’s Oncology Group. Blood. 2011;117:3010–5.
Thomas X, Boiron JM, Huguet F, Dombret H, Bradstock K, Vey N, et al. Outcome of treatment in adults with acute lymphoblastic leukemia: analysis of the LALA-94 trial. J Clin Oncol. 2004;22:4075–86.
Jacobs SR, Michalek RD, Rathmell JC. IL-7 is essential for homeostatic control of T cell metabolism in vivo. J Immunol. 2010;184:3461–9.
Karawajew L, Ruppert V, Wuchter C, Kosser A, Schrappe M, Dorken B, et al. Inhibition of in vitro spontaneous apoptosis by IL-7 correlates with bcl-2 up-regulation, cortical/mature immunophenotype, and better early cytoreduction of childhood T-cell acute lymphoblastic leukemia. Blood. 2000;96:297–306.
Barata JT, Cardoso AA, Nadler LM, Boussiotis VA. Interleukin-7 promotes survival and cell cycle progression of T-cell acute lymphoblastic leukemia cells by down-regulating the cyclin-dependent kinase inhibitorp27(kip1). Blood. 2001;98:1524–31.
Scupoli MTV F, Krampera M, Vincenzi C, Nadali G, Zampieri F, Ritter MA, et al. Thymic epithelial cells promote survival of human T-cell acute lymphoblastic leukemia blasts: the role of interleukin-7. Haematologica. 2003;88:1229–37.
Barata JTS A, Brandao JG, Nadler LM, Cardoso AA, Boussiotis V. A. Activation of PI3K is indispensable for interleukin 7-mediated viability, proliferation, glucose use, and growth of T cell acute lymphoblastic leukemia cells. J Exp Med. 2004;200:659–69.
Scupoli MT, Perbellini O, Krampera M, Vinante F, Cioffi F, Pizzolo G. Interleukin 7 requirement for survival of T-cell acute lymphoblastic leukemia and human thymocytes on bone marrow stroma. Haematologica. 2007;92:264–6.
Rich BE, Campos-Torres J, Tepper RI, Moreadith RW, Leder P. Cutaneous lymphoproliferation and lymphomas in interleukin 7 transgenic mice. J Exp Med. 1993;177:305–16.
Laouar Y, Crispe IN, Flavell RA. Overexpression of IL-7R alpha provides a competitive advantage during early T-cell development. Blood. 2004;103:1985–94.
Silva AL AB, Martins LR, Cardoso BA, Demengeot J, Yunes JA, Seddon B, et al. IL-7 contributes to the progression of human T-cell acute lymphoblastic leukemias. Cancer Res. 2011;71:4780–9.
Shochat C, Tal N, Bandapalli OR, Palmi C, Ganmore I, te Kronnie G, et al. Gain-of-function mutations in interleukin-7 receptor-alpha (IL7R) in childhood acute lymphoblastic leukemias. J Exp Med. 2011;208:901–8.
Zenatti PPR D, Li W, Zuurbier L, Silva MC, Paganin M, Tritapoe J, et al. Oncogenic IL7R gain-of-function mutations in childhood T-cell acute lymphoblastic leukemia. Nat Genet. 2011;43:932–9.
Cramer SD, Aplan PD, Durum SK. Therapeutic targeting of IL-7Ralpha signaling pathways in ALL treatment. Blood. 2016;128:473–8.
Oosterwegel MA, Haks MC, Jeffry U, Murray R, Kruisbeek AM. Induction of TCR gene rearrangements in uncommitted stem cells by a subset of IL-7 producing, MHC class-II-expressing thymic stromal cells. Immunity. 1997;6:351–60.
Alves NL, Richard-Le Goff O, Huntington ND, Sousa AP, Ribeiro VS, Bordack A, et al. Characterization of the thymic IL-7 niche in vivo. Proc Natl Acad Sci USA. 2009;106:1512–7.
Funk PE, Stephan RP, Witte PL. Vascular cell adhesion molecule 1-positive reticular cells express interleukin-7 and stem cell factor in the bone marrow. Blood. 1995;86:2661–71.
Liu Y, Easton J, Shao Y, Maciaszek J, Wang Z, Wilkinson MR, et al. The genomic landscape of pediatric and young adult T-lineage acute lymphoblastic leukemia. Nat Genet. 2017;49:1211–8.
Oshima S, Nakamura T, Namiki S, Okada E, Tsuchiya K, Okamoto R, et al. Interferon regulatory factor 1 (IRF-1) and IRF-2 distinctively up-regulate gene expression and production of interleukin-7 in human intestinal epithelial cells. Mol Cell Biol. 2004;24:6298–310.
Cai YJ, Wang WS, Yang Y, Sun LH, Teitelbaum DH, Yang H. Up-regulation of intestinal epithelial cell derived IL-7 expression by keratinocyte growth factor through STAT1/IRF-1, IRF-2 pathway. PLoS ONE. 2013;8:e58647.
Deaton AM, Bird A. CpG islands and the regulation of transcription. Genes Dev. 2011;25:1010–22.
Jones PA. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat Rev Genet. 2012;13:484–92.
Richter-Pechanska P, Kunz JB, Bornhauser B, von Knebel Doeberitz C, Rausch T, Erarslan-Uysal B, et al. PDX models recapitulate the genetic and epigenetic landscape of pediatric T-cell leukemia. EMBO Mol Med. 2018;10(12):e9443.
Fry TJ, Mackall CL. Interleukin-7: from bench to clinic. Blood. 2002;99:3892–904.
Barata JTS A, Abecasis M, Carlesso N, Cumano A, Cardoso A. A molecular and functional evidence for activity of murine IL-7 on human lymphocytes. Exp Hematol. 2006;34:1133–42.
Black KL, Naqvi AS, Asnani M, Hayer KE, Yang SY, Gillespie E, et al. Aberrant splicing in B-cell acute lymphoblastic leukemia. Nucleic Acids Res. 2018;46:11357–69.
Poglio S, Cahu X, Uzan B, Besnard-Guerin C, Lapillonne H, Leblanc T, et al. Rapid childhood T-ALL growth in xenograft models correlates with mature phenotype and NF-kappaB pathway activation but not with poor prognosis. Leukemia. 2015;29:977–80.
Weng AP, Ferrando AA, Lee W, Morris JPt, Silverman LB, Sanchez-Irizarry C, et al. Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science. 2004;306:269–71.
Buffière A, Accogli T, Saint-Paul L, Lucchi G, Uzan B, Ballerini P, et al. Saracatinib impairs maintenance of human T-ALL by targeting the LCK tyrosine kinase in cells displaying high level of lipid rafts. Leukemia. 2018;32:2062–5.
Acknowledgements
The authors express their gratitude to the animal housing facility and cytometry platform staff at the University of Burgundy (Dijon, France), especially Valérie Saint-Giorgio, David Lheraud, Sébastien Lapipe, Anabelle Sequeira, and Serge Monier, for their excellent suggestions and valuable technical support. The authors also thank Julien Guy for providing information on T-ALL immune data that helped in the EGIL characterization, as well as Antonio Vitobello for providing insight about DNA methylation. The authors are also grateful to Suzanne Rankin (CHU Dijon) for editing and proofreading this paper. This study was supported by grants from the Association Laurette Fugain (RQ), the Conférence de Coordination Interrégionale du Grand Est – Bourgogne Franche-Comté (CCIRGE-BFC) de la Ligue contre le Cancer (RQ), by the National Research Agency under the program “Investissements d’Avenir” (reference ANR-11-LABX-0021, LipSTIC Labex), by the Conseil Régional de Bourgogne through the plan d’action régional pour l’innovation (PARI), and the European Union through the PO FEDER-FSE Bourgogne 2014/2020 programs. AB was supported by fellowships from the French Ministère de la Recherche et de l’Enseignement Supérieur (MRES) and the Ligue contre le Cancer. FP and BU have grants from LNCC, INSERM, CEA, and Université Paris Diderot.
Author information
Authors and Affiliations
Contributions
AB and RQ conceived the study, performed experiments, analyzed data, and wrote the paper; BU, RA, FH, MM, SN, AA, VC, and BT helped with experiments; OB provided important material; PB performed cytogenetic; JTB, JNB, and LD discussed the data; FP provided human T-ALL primary samples and xenografts, advices in the usage of such models, discussed the data, and helped with the writing of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Buffière, A., Uzan, B., Aucagne, R. et al. T-cell acute lymphoblastic leukemia displays autocrine production of Interleukin-7. Oncogene 38, 7357–7365 (2019). https://doi.org/10.1038/s41388-019-0921-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-019-0921-4
- Springer Nature Limited