Abstract
Background:
Study compliance is crucial when the study outcome is determined by an invasive procedure, such as prostate biopsy. To investigate predictors of compliance in study-mandated prostate biopsies, we analyzed demographic, clinical and reported lifestyle data from the REDUCE trial.
Methods:
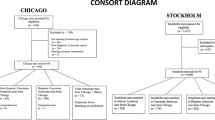
We retrospectively identified 8025 men from REDUCE with at least 2 years of follow-up, and used multivariable logistic regression to test the association between baseline demographic and clinical characteristics and undergoing the study-mandated prostate biopsy at 2 years. We then examined whether missing any of these data was associated with undergoing a biopsy.
Results:
In REDUCE, 22% of men did not undergo a 2-year biopsy. On multivariable analysis, the non-North American region was predictive of 42–44% increased likelihood of undergoing a 2-year biopsy (P⩽0.001). Being enrolled at a center that enrolled >10 subjects (2nd and 3rd tertile) was associated with a 42–48% increased likelihood of undergoing a 2-year biopsy (P<0.001). In addition, black race predicted 44% lower rate of on-study 2-year biopsy (odds ratio (OR)=0.56; P=0.001). Finally, missing one or more baseline variables was associated with a 32% decreased likelihood of undergoing a 2-year biopsy (OR=0.68; P<0.001).
Conclusions:
In REDUCE, men outside North America, those at higher volume centers and those with complete baseline data were more likely to undergo study-mandated 2-year biopsies. Given prostate biopsy is becoming increasingly utilized as an endpoint in trials that are often multi-national, regional differences in compliance should be considered when designing future trials. Likewise, efforts are needed to ensure compliance in low-volume centers or among subjects missing baseline data.

Similar content being viewed by others
References
Osterberg L, Blaschke T . Adherence to medication. N Engl J Med 2005; 353: 487–497.
Haynes RB, Ackloo E, Sahota N, McDonald HP, Yao X . Interventions for enhancing medication adherence. Cochrane Database Syst Rev 2008; (2):CD000011.
Andriole GL, Crawford ED, Grubb RL 3rd, Buys SS, Chia D, Church TR et al. Prostate cancer screening in the randomized Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial: mortality results after 13 years of follow-up. J Natl Cancer Inst 2012; 104: 125–132.
Avery KN, Metcalfe C, Vedhara K, Lane JA, Davis M, Neal DE et al. Predictors of attendance for prostate-specific antigen screening tests and prostate biopsy. Eur Urol 2012; 62: 649–655.
Gritz ER, Arnold KB, Moinpour CM, Burton-Chase AM, Tangen CM, Probstfield JF et al. Factors associated with adherence to an end-of-study biopsy: lessons from the prostate cancer prevention trial (SWOG-Coordinated Intergroup Study S9217). Cancer Epidemiol Biomarkers Prev 2014; 23: 1638–1648.
Marcus PM, Ogden SL, Gren LH, Childs JC, Pretzel SM, Lamerato LE et al. Non-compliance with the initial screening exam visit in the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial. Prev Med 2014; 67: 82–88.
Bill-Axelson A, Holmberg L, Garmo H, Rider JR, Taari K, Busch C et al. Radical prostatectomy or watchful waiting in early prostate cancer. N Engl J Med 2014; 370: 932–942.
Schroder FH, Hugosson J, Roobol MJ, Tammela TL, Ciatto S, Nelen V et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med 2009; 360: 1320–1328.
Bul M, Zhu X, Valdagni R, Pickles T, Kakehi Y, Rannikko A et al. Active surveillance for low-risk prostate cancer worldwide: the PRIAS study. Eur Urol 2013; 63: 597–603.
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T et al. EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 2014; 65: 124–137.
Carter HB, Albertsen PC, Barry MJ, Etzioni R, Freedland SJ, Greene KL et al. Early detection of prostate cancer: AUA Guideline. J Urol 2013; 190: 419–426.
Andriole GL, Bostwick DG, Brawley OW, Gomella LG, Marberger M, Montorsi F et al. Effect of dutasteride on the risk of prostate cancer. N Engl J Med 2010; 362: 1192–1202.
Fowke JH, Howard L, Andriole GL, Freedland SJ . Alcohol intake increases high-grade prostate cancer risk among men taking dutasteride in the REDUCE Trial. Eur Urol 2014; 66: 1133–1138.
Nelson JB, Love W, Chin JL, Saad F, Schulman CC, Sleep DJ et al. Phase 3, randomized, controlled trial of atrasentan in patients with nonmetastatic, hormone-refractory prostate cancer. Cancer 2008; 113: 2478–2487.
Kwon ED, Drake CG, Scher HI, Fizazi K, Bossi A, van den Eertwegh AJ et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol 2014; 15: 700–712.
Cramer J, Rosenheck R, Kirk G, Krol W, Krystal J . VA Naltrexone Study Group 425. Medication compliance feedback and monitoring in a clinical trial: predictors and outcomes. Value Health 2003; 6: 566–573.
Besch CL . Compliance in clinical trials. AIDS 1995; 9: 1–10.
Vermeire E, Hearnshaw H, Van Royen P, Denekens J . Patient adherence to treatment: three decades of research. A comprehensive review. J Clin Pharm Ther 2001; 26: 331–342.
Fouad MN, Johnson RE, Nagy MC, Person SD, Partridge EE . Adherence and retention in clinical trials: a community-based approach. Cancer 2014; 120 (Suppl 7): 1106–1112.
Prorok PC, Andriole GL, Bresalier RS, Buys SS, Chia D, Crawford ED et al. Design of the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial. Control Clin Trials 2000; 21 (6 Suppl): 273S–309S.
Baron JA, Cole BF, Sandler RS, Haile RW, Ahnen D, Bresalier R et al. A randomized trial of aspirin to prevent colorectal adenomas. N Engl J Med 2003; 348: 891–899.
Arber N, Eagle CJ, Spicak J, Racz I, Dite P, Hajer J et al. Celecoxib for the prevention of colorectal adenomatous polyps. N Engl J Med 2006; 355: 885–895.
Okura Y, Urban LH, Mahoney DW, Jacobsen SJ, Rodeheffer RJ . Agreement between self-report questionnaires and medical record data was substantial for diabetes, hypertension, myocardial infarction and stroke but not for heart failure. J Clin Epidemiol 2004; 57: 1096–1103.
Acknowledgements
This study was supported by GlaxoSmithKline and NIH 1K24CA160653. ClinicalTrials.gov Identifier: NCT00056407.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
This study was supported by GlaxoSmithKline (GSK). Dr Andriole is a consultant to GSK. Dr Castro-Santamaria is an employee of GSK. Dr Freedland received research support from GSK and NIH K24CA160653. The remaining authors declare no conflicts of interest.
Additional information
Supplementary Information accompanies the paper on the Prostate Cancer and Prostatic Diseases website
Supplementary information
Rights and permissions
About this article
Cite this article
Fischer, S., Sun, S., Howard, L. et al. Baseline subject characteristics predictive of compliance with study-mandated prostate biopsy in men at risk of prostate cancer: results from REDUCE. Prostate Cancer Prostatic Dis 19, 202–208 (2016). https://doi.org/10.1038/pcan.2016.5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2016.5
- Springer Nature Limited
This article is cited by
-
The impact of complications after initial prostate biopsy on repeat protocol biopsy acceptance rate. Results from the Prostate Cancer Research International: Active Surveillance JAPAN study
International Journal of Clinical Oncology (2020)
-
Serum cholesterol and risk of high-grade prostate cancer: results from the REDUCE study
Prostate Cancer and Prostatic Diseases (2018)
-
Are intensive data collection methods in pain research feasible in those with physical disability? A study in persons with chronic pain and spinal cord injury
Quality of Life Research (2017)