Abstract
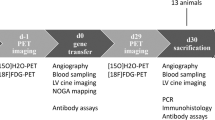
This study aimed to assess safety and therapeutic potential of gene electrotransfer (GET) as a method for delivery of plasmid encoding vascular endothelial growth factor A (VEGF-A) to ischemic myocardium in a porcine model. Myocardial ischemia was induced by surgically occluding the left anterior descending coronary artery in swine. GET following plasmid encoding VEGF-A injection was performed at four sites in the ischemic region. Control groups either received injections of the plasmid without electrotransfer or injections of the saline vehicle. Animals were monitored for 7 weeks and the hearts were evaluated for angiogenesis, myocardial infarct size and left ventricular contractility. Arteriograms suggest growth of new arteries as early as 2 weeks after treatment in electrotransfer animals. There is a significant reduction of infarct area and left ventricular contractility is improved in GET-treated group compared with controls. There was no significant difference in mortality of animals treated with GET of plasmid encoding VEGF-A from the control groups. Gene delivery of plasmid encoding VEGF-A to ischemic myocardium in a porcine model can be accomplished safely with potential for myocardial repair and regeneration.




Similar content being viewed by others
References
Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB et al. Heart disease and stroke statistics—2013 update: a report from the American Heart Association. Circulation 2013; 127: e6–e245.
Mussolino M, Shurin SB . 2012 NHLBI Morbidity and Mortality Chart Book. 2013. NHLBI: Bethesda, MD, USA, 2014. Available from http://www.nhlbi.nih.gov/files/docs/research/2012_ChartBook_508.pdf.
Krumholz HM, Merrill AR, Schone EM, Schreiner GC, Chen J, Bradley EH et al. Patterns of hospital performance in acute myocardial infarction and heart failure 30-day mortality and readmission. Circ Cardiovasc Qual Outcomes 2009; 2: 407–413.
Ross R . The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature 1993; 362: 801–809.
Mukherjee D, Bhatt DL, Roe MT, Patel V, Ellis SG . Direct myocardial revascularization and angiogenesis—how many patients might be eligible? Am J Cardiol 1999; 84: 598–600; A8.
Wolfram JA, Donahue JK . Gene therapy to treat cardiovascular disease. J Am Heart Assoc 2013; 2: e000119.
Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J et al. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med 2001; 7: 430–436.
Mercola M, Ruiz-Lozano P, Schneider MD . Cardiac muscle regeneration: lessons from development. Genes Dev 2011; 25: 299–309.
Ferrara N, Gerber HP, LeCouter J . The biology of VEGF and its receptors. Nat Med 2003; 9: 669–676.
Liu W, Xu J, Wang M, Wang Q, Bi Y, Han M . Tumor-derived vascular endothelial growth factor (VEGF)-A facilitates tumor metastasis through the VEGF-VEGFR1 signaling pathway. Int J Oncol 2011; 39: 1213–1220.
Folkman J . Tumor angiogenesis: therapeutic implications. N Engl J Med 1971; 285: 1182–1186.
Tang JM, Wang JN, Zhang L, Zheng F, Yang JY, Kong X et al. VEGF/SDF-1 promotes cardiac stem cell mobilization and myocardial repair in the infarcted heart. Cardiovasc Res 2011; 91: 402–411.
Tao Z, Chen B, Tan X, Zhao Y, Wang L, Zhu T et al. Coexpression of VEGF and angiopoietin-1 promotes angiogenesis and cardiomyocyte proliferation reduces apoptosis in porcine myocardial infarction (MI) heart. Proc Natl Acad Sci USA 2011; 108: 2064–2069.
Chen Y, Amende I, Hampton TG, Yang Y, Ke Q, Min JY et al. Vascular endothelial growth factor promotes cardiomyocyte differentiation of embryonic stem cells. Am J Physiol Heart Circ Physiol 2006; 291: H1653–H1658.
Urbich C, Aicher A, Heeschen C, Dernbach E, Hofmann WK, Zeiher AM et al. Soluble factors released by endothelial progenitor cells promote migration of endothelial cells and cardiac resident progenitor cells. J Mol Cell Cardiol 2005; 39: 733–742.
Muller OJ, Katus HA, Bekeredjian R . Targeting the heart with gene therapy-optimized gene delivery methods. Cardiovasc Res 2007; 73: 453–462.
Yla-Herttuala S, Alitalo K . Gene transfer as a tool to induce therapeutic vascular growth. Nat Med 2003; 9: 694–701.
Ayuni EL, Gazdhar A, Giraud MN, Kadner A, Gugger M, Cecchini M et al. In vivo electroporation mediated gene delivery to the beating heart. PLoS One 2010; 5: e14467.
Tevaearai HT, Gazdhar A, Giraud MN, Fluck M . In vivo electroporation-mediated gene delivery to the beating heart. Methods Mol Biol 2014; 1121: 223–229.
Hargrave B, Downey H, Strange R Jr, Murray L, Cinnamond C, Lundberg C et al. Electroporation-mediated gene transfer directly to the swine heart. Gene Therapy 2013; 20: 151–157.
Hargrave B, Strange R Jr, Navare S, Stratton M, Burcus N, Murray L et al. Gene electro transfer of plasmid encoding vascular endothelial growth factor for enhanced expression and perfusion in the ischemic Swine heart. PLoS One 2014; 9: e115235.
Turillazzi E, Di Paolo M, Neri M, Riezzo I, Fineschi V . A theoretical timeline for myocardial infarction: immunohistochemical evaluation and western blot quantification for Interleukin-15 and Monocyte chemotactic protein-1 as very early markers. J Transl Med 2014; 12: 188.
Logsdon EA, Finley SD, Popel AS, Mac Gabhann F . A systems biology view of blood vessel growth and remodelling. J Cell Mol Med 2014; 18: 1491–1508.
[Anonymous]. The Thrombolysis in Myocardial Infarction (TIMI) trial. Phase I findings. TIMI Study Group. N Engl J Med 1985; 312: 932–936.
Abraham J, Abraham TP . The role of echocardiography in hemodynamic assessment in heart failure. Heart Fail Clin 2009; 5: 191–208.
Teichholz LE, Kreulen T, Herman MV, Gorlin R . Problems in echocardiographic volume determinations: echocardiographic-angiographic correlations in the presence of absence of asynergy. Am J Cardiol 1976; 37: 7–11.
Dib N, Diethrich EB, Campbell A, Gahremanpour A, McGarry M, Opie SR . A percutaneous swine model of myocardial infarction. J Pharmacol Toxicol Methods 2006; 53: 256–263.
Acknowledgements
We thank Dr Mark Jaroszeski of University of South Florida, Tampa, FL, USA for design and construction of the electrode arrays used for gene electrotransfer. We also thank RaShae Cooke, Danielle Tajirian, Ben Cuffee and Ann Nix for providing excellent veterinary care to the animals throughout this study. We also thank Dr Robert Strange Jr of Portsmouth Naval Facility, Portsmouth, VA, USA and Dr Paul D Mahoney of Sentara Heart Hospital, Norfolk, VA, USA for surgical and arteriogram acquisition training. Funding for this work came from the National Institutes of Health R33 HL005441. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Bulysheva, A., Hargrave, B., Burcus, N. et al. Vascular endothelial growth factor-A gene electrotransfer promotes angiogenesis in a porcine model of cardiac ischemia. Gene Ther 23, 649–656 (2016). https://doi.org/10.1038/gt.2016.35
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2016.35
- Springer Nature Limited
This article is cited by
-
Genetically engineered HEK cells as a valuable tool for studying electroporation in excitable cells
Scientific Reports (2024)
-
HiPS-Cardiac Trilineage Cell Generation and Transplantation: a Novel Therapy for Myocardial Infarction
Journal of Cardiovascular Translational Research (2020)
-
Long-term Intake of Pasta Containing Barley (1–3)Beta-D-Glucan Increases Neovascularization-mediated Cardioprotection through Endothelial Upregulation of Vascular Endothelial Growth Factor and Parkin
Scientific Reports (2017)