Abstract
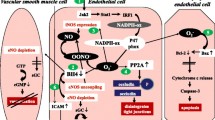
Background: Sepsis initiates an inflammatory response that causes widespread injury, and candidates for related myocardial depressant factors include cytokines and nitric oxide (NO). Nuclear factor kappa-B (NF-KB) stimulated by toll-like receptor 4 activation in sepsis mediates the transcription of multiple proinflammatory genes. These inflammatory mediators can cause myocardial dysfunction, which may deteriorate sepsis outcomes. To address this risk, we investigated the potential beneficial effects of a novel isoquinolines derivative, CYY054c, in LPS-induced inflammatory response leading to endotoxemia.
Methods: The effects of CYY054c on cytokine and inflammatory-related protein production were evaluated in lipopolysaccharide (LPS)-stimulated macrophages. To determine whether CYY054c alleviates inflammatory storm-induced myocardial dysfunction in vivo, LPS was injected in rats, and cardiac function was measured by a pressure-volume loop.
Results: CYY054c inhibited LPS-induced NF-KB expression in macrophages and reduced the release of tumor necrosis factor-alpha (TNF-1α), interleukin-1β (IL-1β), and interleukin-6 (IL-6), as well as the expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2). In the animal studies, CYY054c alleviated LPS-upregulated plasma TNF-ct, IL-1β, IL-6, and NO concentrations, as well as cardiac monocyte chemotactic protein-1, iNOS, and COX-2 expression in rats, contributing to the improvement of cardiac function during endotoxemia.
Conclusions: The reduction of NF-KB-mediated inflammatory mediators and the maintenance of hemodynamic performance by CYY054c improved the outcomes during endotoxemia. CYY054c may be a potential therapeutic agent for sepsis.
Similar content being viewed by others
References
Nduka OO, Parrillo JE. The pathophysiology of septic shock. Crit Care Clin 2009;25:677–702.vii.
Janeway Jr CA, Medzhitov R. Innate immune recognition. Annu Rev Immunol 2002;20:197–216.
Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity Cell 2006;124:783–801.
Kawai T, Akira S. Signaling to NF-kappaB by toll-like receptors. Trends Mol Med 2007;13:460–9.
Bosmann M, Ward PA. The inflammatory response in sepsis. Trends Immunol 2013;34:129–36.
Schulte W, Bernhagen J, Bucala R. Cytokines in sepsis: potent immunoregulators and potential therapeutic targets—an updated view. Mediators Inflamm 2013;2013:165974.
Aziz M, Jacob A, Yang WL, Matsuda A, Wang P. Current trends in inflammatory and immunomodulatory mediators in sepsis. J Leukoc Biol 2013;93:329–42.
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, et al. 2001 SCCM/ ESICM/ACCP/ATS/SIS international sepsis definitions conference. Intensive Care Med 2003;29:530–8.
Bonizzi G, Karin M. The two NF-KB activation pathways and their role in innate and adaptive immunity. Trends Immunol 2004;25:280–8.
Liu SF, Malik AB. NF-KB activation as a pathological mechanism of septic shock and inflammation. Am J Physiol Lung Cell Mol Physiol 2006;290:L622–45.
Yang Y, Lv J, Jiang S, Ma Z, Wang D, Hu W, et al. The emerging role of Toll-like receptor 4 in myocardial inflammation. Cell Death Dis 2016;7:e2234.
Hochstadt A, Meroz Y, Landesberg G. Myocardial dysfunction in severe sepsis and septic shock: more questions than answers? J Cardiothoracic Vase Anesth 2011;25:526–35.
Blaser H, Dostert C, Mak TW, Brenner D. TNF and ROS crosstalk in inflammation. Trends Cell Biol 2016;26:249–61.
Andrades ME, Morina A, Spasic S, Spasojevic I. Bench-to-bedside review: sepsis - from the redox point of view. Crit Care 2011;15:230.
Landesberg G, Levin PD, Gilon D, Goodman S, Georgieva M, Weissman C, et al. Myocardial dysfunction in severe sepsis and septic shock. Chest 2015;148:93–102.
Li X, Su J, Cui X, Li Y, Barochia A, Eichacker PQ. Can we predict the effects of NF-KB inhibition in sepsis? Studies with parthenolide and ethyl pyruvate. Expert Opin Investig Drugs 2009; 18(8):1047–60.
Cheng B, Hou Y, Wang L, Dong L, Peng J, Bai G. Dual-bioactivity-based liquid chromatography-coupled quadrupole time-of-flight mass spectrometry for NF-kappaB inhibitors and beta2AR agonists identification in Chinese Medicinal Preparation Qingfei Xiaoyan Wan. Anal Bioanal Chem 2012;404:2445–52.
Chen WP, Tzeng HJ, Ku HC, Ho YJ, Lee SS, Su MJ. Thaliporphine ameliorates cardiac depression in endotoxemic rats through attenuating TLR4 signaling in the downstream of TAK-1 phosphorylation and NF-kappaB signaling. Naunyn Schmiedebergs Arch Pharmacol 2010;382:441–53.
Lee AS, Chen WP, Kuo YL, Ho YJ, Lee SS, Su MJ. Thaliporphine preserves cardiac function of endotoxemic rabbits by both directly and indirectly attenuating NFkappaB signaling pathway. PLoS One 2012;7:e39174.
Ohmori Y, Tebo J, Nedospasov S, Hamilton TA. Kappa B binding activity in a murine macrophage-like cell line. Sequence-specific differences in kappa B binding and transcriptional activation functions. J Biol Chem 1994;269:17684–90.
Fullerton JN, Segre E, De Maeyer RP, Maini AA, Gilroy DW. Intravenous endotoxin challenge in healthy humans: an experimental platform to investigate and modulate systemic inflammation. J Vis Exp 2016(111)53913.
Thomas RC, Bath MF, Stover CM, Lambert DG, Thompson JP. Exploring LPS- induced sepsis in rats and mice as a model to study potential protective effects of the nociceptin/orphanin FQ system. Peptides 2014;61:56–60.
Ku HC, Lee SY, Chen CH, Wang YH, Lin CT, Lee SS, et al. TM-1-1DP exerts protective effect against myocardial ischemia reperfusion injury via AKT-eNOS pathway. Naunyn Schmiedebergs Arch Pharmacol 2015;388:539–48.
Ku HC, Chen WP, Su MJ. GLP-1 signaling preserves cardiac function in endotoxemic Fischer 344 and DPP4-deficient rats. Naunyn Schmiedebergs Arch Pharmacol 2010;382:463–74.
Ku HC, Chen WP, Su MJ. DPP4 deficiency preserves cardiac function via GLP-1 signaling in rats subjected to myocardial ischemia/reperfusion. Naunyn Schmiedebergs Arch Pharmacol 2011;384:197–207.
Ku HC, Lee SY, Yang KC, Kuo YH, Su MJ. Modification of caffeic acid with pyrrolidine enhances antioxidant ability by activating AKT/HO-1 pathway in heart. PLoS One 2016;11:e0148545.
Fang H, Pengal RA, Cao X, Ganesan LP, Wewers MD, Marsh CB, et al. Lipopolysaccharide-induced macrophage inflammatory response is regulated by SHIP. J Immunol 2004;173:360–6.
Spasojevic I, Obradovic B, Spasic S. Bench-to-bedside review: neonatal sepsis- redox processes in pathogenesis. Crit Care 2012;16:221.
Slimani H, Zhai Y, Yousif NG, Ao L, Zeng Q, Fullerton DA, et al. Enhanced monocyte chemoattractant protein-1 production in aging mice exaggerates cardiac depression during endotoxemia. Crit Care 2014;18:527.
Schall TJ, Bacon KB. Chemokines, leukocyte trafficking, and inflammation. Curr Opinion Immunol 1994;6:865–73.
Li YS, Shyy YJ, Wright JG, Valente AJ, Cornhill JF, Kolattukudy PE. The expression of monocyte chemotactic protein (MCP-1) in human vascular endothelium in vitro and in vivo. Mol Cell Biochem 1993;126:61–8.
Hunter JD, Doddi M. Sepsis and the heart. Br J Anaesth 2010;104:3–11.
Beesley SJ, Weber G, Sarge T, Nikravan S, Grissom CK, Lanspa MJ, et al. Septic cardiomyopathy. Crit Care Med 2018;46:625–34.
Jeong HS, Lee TH, Bang CH, Kim JH, Hong SJ. Risk factors and outcomes of sepsis-induced myocardial dysfunction and stress-induced cardiomyopathy in sepsis or septic shock. Medicine (Baltimore) 2018;97:e0263.
Palmieri V, Innocenti F, Guzzo A, Guerrini E, Vignaroli D, Pini R. Left ventricular systolic longitudinal function as predictor of outcome in patients with sepsis. Circ Cardiovasc Imaging 2015;8:e003865.
Wink DA, Hines HB, Cheng RY, Switzer CH, Flores-Santana W, Vitek MP, et al. Nitric oxide and redox mechanisms in the immune response. J Leukoc Biol Suppl 2011;89:873–91.
Ward PA. The sepsis seesaw: seeking a heart salve. Nat Med 2009;15:497–8.
Morimoto H, Takahashi M. Role of monocyte chemoattractant protein-1 in myocardial infarction. Int J Biomed Sci 2007;3:159–67.
Kolattukudy PE, Quach T, Bergese S, Breckenridge S, Hensley J, Altschuld R, et al. Myocarditis induced by targeted expression of the MCP-1 gene in murine cardiac muscle. Am J Pathol 1998;152:101–11.
Grimaldi D, Goicoechea Turcott EW, Taccone FS. IL-1 receptor antagonist in sepsis: new findings with old data? J Thorac Dis 2016;8:2379–82.
Beutler B, Milsark IW, Cerami AC. Passive immunization against cachectin/ tumor necrosis factor protects mice from lethal effect of endotoxin. Science 1985;229(4716)869-71 Classical article. J Immunol. 2008;181:7–9.
Panacek EA, Marshall JC, Albertson TE, Johnson DH, Johnson S, MacArthur RD, et al. Efficacy and safety of the monoclonal anti-tumor necrosis factor antibody F(ab’)2 fragment afelimomab in patients with severe sepsis and elevated interleukin-6 levels. Crit Care Med 2004;32:2173–82.
Ping Q, Xizhong C, Junfeng S, Judith W, Charles N, Peter QE. Antitumor necrosis factor therapy is associated with improved survival in clinical sepsis trials: a meta-analysis. Crit Care Med 2013;41:.
Yang J, Zhang R, Jiang X, Lv J, Li Y, Ye H, et al. Toll-like receptor 4-induced ryanodine receptor 2 oxidation and sarcoplasmic reticulum Ca(2+) leakage promote cardiac contractile dysfunction in sepsis. J Biol Chem 2018;293:794–807.
Rice TW, Wheeler AP, Bernard GR, Vincent JL, Angus DC, Aikawa N, et al. A randomized, double-blind, placebo-controlled trial of TAK-242 for the treatment of severe sepsis. Crit Care Med 2010;38:1685–94.
Alexander HR, Doherty GM, Buresh CM, Venzon DJ, Norton JA. A recombinant human receptor antagonist to interleukin 1 improves survival after lethal endotoxemia in mice. J Exp Med 1991;173:1029–32.
Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med 2017;45:486–552.
Tisoncik JR, Korth MJ, Simmons CP, Farrar J, Martin TR, Katze MG. Into the eye of the cytokine storm. Microbiol Mol Biol Rev 2012;76:16–32.
Serio KJ, Reddy KV, Bigby TD. Lipopolysaccharide induces 5-lipoxygenase-activating protein gene expression in THP-1 cells via a NF-kappaB and C/EBP-mediated mechanism. Am J Physiol Cell Physiol 2005;288:C1125–33.
Heusch G, Schulz R. A radical view on the contractile machinery in human heart failure. J Am Coll Cardiol 2011;57:310–2.
Bishayi B, Bandyopadhyay D, Majhi A, Adhikary R. Effect of exogenous MCP-1 onTLR-2 neutralized murine macrophages and possible mechanisms of CCR-2/ TLR-2 and MCP-1 signalling during Staphylococcus aureus infection. Immunobiology 2015;220:350–62.
Abraham E. Alterations in cell signaling in sepsis. Clin Infect Dis 2005;41 (Suppl. 7):S459–64.
Sha T, Sunamoto M, Kitazaki T, Sato J, Ii M, Iizawa Y. Therapeutic effects of TAK-242, a novel selective Toll-like receptor 4 signal transduction inhibitor, in mouse endotoxin shock model. Euro J Pharmacol 2007;571:231–9.
Takashima K, Matsunaga N, Yoshimatsu M, Hazeki K, Kaisho T, Uekata M, et al. Analysis of binding site for the novel small-molecule TLR4 signal transduction inhibitor TAK-242 and its therapeutic effect on mouse sepsis model. Br J Pharmacol 2009;157:1250–62.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, SY., Hsin, LW., Su, MJ. et al. A novel isoquinoline derivative exhibits anti-inflammatory properties and improves the outcomes of endotoxemia. Pharmacol. Rep 71, 1281–1288 (2019). https://doi.org/10.1016/j.pharep.2019.06.015
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2019.06.015