Abstract
Background
The objective of this study was to evaluate creatine as an anti-nociceptive compound in an animal model of thermal and inflammatory pain. Creatine has the structural potential to interact with acid-sensing ion channels (ASIC), which have been involved in pain sensation modulation. The hypothesis evaluated in this study was that creatine will interact with ASICs leading to decreased nociception.
Methods
Male and female C57BL/6J mice were fed with either a control diet or the control diet supplemented with creatine (6.25 g/kg diet). After one week on the diet, the mice were tested for thermal hyperalgesia and inflammatory pain response.
Results
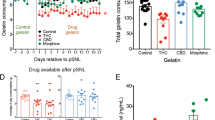
The latency to withdraw the tail during the thermal hyperalgesia test was unaffected by sex or diet. During the formalin test, males and females responded differently to the stimulus, and the female mice supplemented with creatine seemed to recover faster than the controls. To determine whether ASICs mediate the action of creatine, GMQ, an ASIC3 agonist, was injected in one paw and pain response was quantified. Females responded more strongly to GMQ injections, and all mice fed creatine had a decreased response to GMQ.
Conclusions
These preliminary data suggest a potential effect of creatine on inflammation-based nociception that may be mediated via ASIC3. While preliminary, this study warrants further research on the potential of creatine as an analgesic and can serve as a stepping stone for the development of ASIC-based therapeutics.
Similar content being viewed by others
Abbreviations
- APETx2:
-
Anthopleura Elegantissima toxin 2
- ASIC:
-
acid sensing ion channel
- CFA:
-
complete Freund’s adjuvant
- GMQ:
-
2-guanidine-4-methylquinazoline
- TRPV1:
-
transient receptor potential V1
References
Relieving pain in america. A Blueprint for Transforming Prevention, Care, Education, and Research. The National Academies Press; 2011.
Trang T, Al-Hasani R, Salvemini D, Salter MW, Gutstein H, Cahill CM. Pain and poppies the good, the bad, and the ugly of opioid analgesics. J Neurosci 2015;35(41):13879–88.
Baron A, Lingueglia E. Pharmacology of acid-sensing ion channels — Physiological and therapeutical perspectives. Neuropharmacology 2015;94:19–35.
Li WG, Xu TL. Acid-sensing ion channels: a novel therapeutic target for pain and anxiety. Curr Pharm Des 2015;21(7):885–94.
Li WG, Xu TL. ASIC3 channels in multimodal sensory perception. ACS Chem Neurosci 2011;2(1):26–37.
Kellenberger S, Schild L. International Union of Basic and Clinical Pharmacology XCI. structure, function, and pharmacology of acid-sensing ion channels and the epithelial Na+ channel. Pharmacol Rev 2015;67(1):1–35.
Xiong ZG, Zhu XM, Chu XP, Minami M, Hey J, Wei WL, et al. Neuroprotection in ischemia: blocking calcium-permeable acid-sensing ion channels. Cell 2004;118(6):687–98.
Yang ZJ, Ni X, Carter EL, Kibler K, Martin LJ, Koehler RC. Neuroprotective effect of acid-sensing ion channel inhibitor psalmotoxin-1 after hypoxia-ischemia in newborn piglet striatum. Neurobiol Dis 2011;43(2):446–54.
Wemmie JA, Taugher RJ, Kreple CJ. Acid-sensing ion channels in pain and disease. Nat Rev Neurosci 2013;14(7):461–71.
Waldmann R, Bassilana F, de Weille J, Champigny G, Heurteaux C, Lazdunski M. Molecular cloning of a non-inactivating proton-gated Na+ channel specific for sensory neurons. J Biol Chem 1997;272(34):20975–8.
Ugawa S, Ueda T, Ishida Y, Nishigaki M, Shibata Y, Shimada S. Amiloride-blockable acid-sensing ion channels are leading acid sensors expressed in human nociceptors. J Clin Invest 2002;110(8):1185–90.
Yu Y, Chen Z, Li WG, Cao H, Feng EG, Yu F, et al. A nonproton ligand sensor in the acid-sensing ion channel. Neuron 2010;68(1):61–72.
Diochot S, Baron A, Rash LD, Deval E, Escoubas P, Scarzello S, et al. A new sea anemone peptide, APETx2, inhibits ASIC3, a major acid-sensitive channel in sensory neurons. EMBO J 2004;23(7):1516–25.
Linley JE, Rose K, Ooi L, Gamper N. Understanding inflammatory pain: ion channels contributing to acute and chronic nociception. Pflugers Arch 2010;459(5):657–69.
Sluka KA, Price MP, Breese NM, Stucky CL, Wemmie JA, Welsh MJ. Chronic hyperalgesia induced by repeated acid injections in muscle is abolished by the loss of ASIC3, but not ASIC1. Pain 2003;106(3):229–39.
Jones NG, Slater R, Cadiou H, McNaughton P, McMahon SB. Acid-induced pain and its modulation in humans. J Neurosci 2004;24(48):10974–9.
Voilley N, de Weille J, Mamet J, Lazdunski M. Nonsteroid anti-inflammatory drugs inhibit both the activity and the inflammation-induced expression of acid-sensing ion channels in nociceptors. J Neurosci 2001;21(20):8026–33.
Mamet J, Baron A, Lazdunski M, Voilley N. Proinflammatory mediators, stimulators of sensory neuron excitability via the expression of acid-sensing ion channels. J Neurosci 2002;22(24):10662–70.
Karczewski J, Spencer RH, Garsky VM, Liang A, Leitl MD, Cato MJ, et al. Reversal of acid-induced and inflammatory pain by the selective ASIC3 inhibitor, APETx2. Br J Pharmacol 2010;161(4):950–60.
Deval E, Gasull X, Noel J, Salinas M, Baron A, Diochot S, et al. Acid-sensing ion channels (ASICs): pharmacology and implication in pain. Pharmacol Ther 2010;128(3):549–58.
Wyss M, Schulze A. Health implications of creatine: can oral creatine supplementation protect against neurological and atherosclerotic disease? Neuroscience 2002;112(2):243–60.
Volek JS, Duncan ND, Mazzetti SA, Staron RS, Putukian M, Gomez AL, et al. Performance and muscle fiber adaptations to creatine supplementation and heavy resistance training. Med Sci Sports Exerc 1999;31(8):1147–56.
Allen PJ. Creatine metabolism and psychiatric disorders: does creatine supplementation have therapeutic value? Neurosci Biobehav Rev 2012;36(5):1442–62.
Amital D, Vishne T, Rubinow A, Levine J. Observed effects of creatine monohydrate in a patient with depression and fibromyalgia. Am J Psychiatry 2006;163(10):1840–1.
Leader A, Amital D, Rubinow A, Amital H. An open-label study adding creatine monohydrate to ongoing medical regimens in patients with the fibromyalgia syndrome. Ann N Y Acad Sci 2009;1173:829–36.
Alves CR, Santiago BM, Lima FR, Otaduy MC, Calich AL, Tritto AC, et al. Creatine supplementation in fibromyalgia: a randomized, double-blind, placebo-controlled trial. Arthritis Care Res (Hoboken) 2013;65(9):1449–59.
Laboratory TJ. Body weight information JAX mice strain C57BL/6J. 2014.
Duarte J, Neuparth M, Soares J, Appell H. Oral creatine supplementation in mice induces hepatic protein overload. Revista Portuguesa de Ciencias do Desporto 2001;1(3):40–3.
Allah Yar R, Akbar A, Iqbal F. Creatine monohydrate supplementation for 10 weeks mediates neuroprotection and improves learning/memory following neonatal hypoxia ischemia encephalopathy in female albino mice. Brain Res 2015;1595:92–100.
Le Bars D, Gozariu M, Cadden SW. Animal models of nociception. Pharmacol Rev 2001;53(4):597–652.
Staniland AA, McMahon SB. Mice lacking acid-sensing ion channels (ASIC) 1 or 2, but not ASIC3, show increased pain behaviour in the formalin test. Eur J Pain 2009;13(6):554–63.
Chen CC, Zimmer A, Sun WH, Hall J, Brownstein MJ, Zimmer A. A role for ASIC3 in the modulation of high-intensity pain stimuli. Proc Natl Acad Sci U S A 2002;99(13):8992–7.
Deval E, Noel J, Lay N, Alloui A, Diochot S, Friend V, et al. ASIC3, a sensor of acidic and primary inflammatory pain. EMBO J 2008;27(22):3047–55.
Bartley EJ, Fillingim RB. Sex differences in pain: a brief review of clinical and experimental findings. Br J Anaesth 2013;111(1):52–8.
Mogil JS, Bailey AL. Sex and gender differences in pain and analgesia. Prog Brain Res 2010;186:141–57.
Mogil JS. Sex differences in pain and pain inhibition: multiple explanations of a controversial phenomenon. Nat Rev Neurosci 2012;13(12):859–66.
Mogil JS, Chesler EJ, Wilson SG, Juraska JM, Sternberg WF. Sex differences in thermal nociception and morphine antinociception in rodents depend on genotype. Neurosci Biobehav Rev 2000;24(3):375–89.
Sternberg WF, Smith L, Scorr L. Nociception and antinociception during the first week of life in mice: sex differences and test dependence. J Pain 2004;5(8):420–6.
Gaumond I, Arsenault P, Marchand S. The role of sex hormones on formalin-induced nociceptive responses. Brain Res 2002;958(1):139–45.
Kim SJ, Calejesan AA, Li P, Wei F, Zhuo M. Sex differences in late behavioral response to subcutaneous formalin injection in mice. Brain Res 1999;829(1–2):185–9.
Martinez-Rojas VA, Barragan-Iglesias P, Rocha-Gonzalez HI, Murbartian J, Granados-Soto V. Role of TRPV1 and ASIC3 in formalin-induced secondary allodynia and hyperalgesia. Pharmacol Rep 2014;66(6):964–71.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Izurieta Munoz, H., Gonzales, E.B. & Sumien, N. Effects of creatine supplementation on nociception in young male and female mice. Pharmacol. Rep 70, 316–321 (2018). https://doi.org/10.1016/j.pharep.2017.11.002
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2017.11.002