Abstract
Background
The opioid system modulates prolactin release during late pregnancy. Its role and the participation of ovarian hormones in this modulation are explored in ether stress-induced prolactin release.
Methods/Results
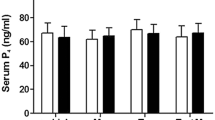
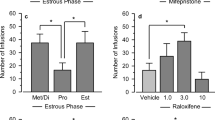
Estrous, 3-day and 19-day pregnant rats were used. We administered the antagonist mifepristone (Mp) and tamoxifen to evaluate progesterone and estradiol action in naloxone (NAL, opioid antagonist) or saline treated rats. Ether stress had no effect on serum prolactin levels in controls but increased prolactin release in NAL-treated rats. Prolactin response to stress in NAL-treated rats was blocked by l-DOPA administration. Mp treatment on day 18 of pregnancy increased prolactin levels after stress without alterations by NAL. Tamoxifen on days 14 and 15 of pregnancy completely blocked Mp and NAL effects on prolactin release at late pregnancy. In contrast, stress significantly increased prolactin levels in estrous rats and pretreatment with NAL prevented this. On day 3 of pregnancy, at 6.00 p.m., stress and NAL treatment inhibited prolactin levels in saline-treated rat. No effect of stress or NAL administration was detected on day 3 of pregnancy at 9.00 a.m. icv administration of specific opioids antagonist, B-Funaltrexamine but not Nor-Binaltorphimine or Naltrindole, caused a significant increase in stress-induced prolactin release.
Conclusions
Opioid system suppression of prolactin stress response during late pregnancy was observed only after progesterone withdrawal, involving a different opioid mechanism from its well-established stimulatory role. This mechanism acts through a mu opioid receptor and requires estrogen participation. The opioid system and progesterone may modulate stress-induced prolactin release, probably involving a putative prolactin-releasing factor.
Similar content being viewed by others
References
Arbogast LA, Voogt JL. Endogenous opioid peptides contribute to suckling-induced prolactin release by suppressing tyrosine hydroxylase activity and messenger ribonucleic acid levels in tuberoinfundibular dopaminergic neurons. Endocrinology 1998;139:2857–62.
Baumann MH, Rabii J. Inhibition of suckling-induced prolactin release by mu-and kappa-opioid antagonists. Brain Res 1991;567:224–30.
Callahan P, Baumann MH, Rabii J. Inhibition of tuberoinfundibular dopaminergic neural activity during suckling: involvement of mu and kappa opiate receptor subtypes. J Neuroendocrinol 1996;8:771–6.
Callahan P, Klosterman S, Prunty D, Tompkins J, Janik J. Immunoneutralization of endogenous opioid peptides prevents the suckling-induced prolactin increase and the inhibition of tuberoinfundibular dopaminergic neurons. Neuroendocrinology 2000;71:268–76.
Sagrillo CA, Voogt JL. Endogenous opioids mediate the nocturnal prolactin surge in the pregnant rat. Endocrinology 1991;129:925–30.
Selmanoff M, Wise PM. Decreased dopamine turnover in the median eminence in response to suckling in the lactating rat. Brain Res 1981;212:101–15.
Soaje M, Deis RP. Opioidergic regulation of prolactin secretion during pregnancy: role of ovarian hormones. J Endocrinol 1997;155:99–106.
Soaje M, Deis RP. A modulatory role of endogenous opioids on prolactin secretion at the end of pregnancy in the rat. J Endocrinol 1994;140:97–102.
Andrews ZB, Grattan DR. Opioid control of prolactin secretion in late pregnant rats is mediated by tuberoinfundibular dopamine neurons. Neurosci Lett 2002;328:60–4.
Sagrillo CA, Voogt JL. Mechanisms for the stimulatory effects of opioidergic and serotonergic input signals on prolactin in pregnant rats. Life Sci 1992;50:1479–89.
Soaje M, Valdez S, Bregonzio C, Penissi A, Deis RP. Dopaminergic mechanisms involved in prolactin release after mifepristone and naloxone treatment during late pregnancy in the rat. Neuroendocrinology 2006;84:58–67.
Ben Jonathan N, Hnasko R. Dopamine as a prolactin (PRL) inhibitor. Endocr Rev 2001;22:724–63.
Freeman ME, Kanyicska B, Lerant A, Nagy G. Prolactin: structure, function, and regulation of secretion. Physiol Rev 2000;80:1523–631.
Gabutti CV, Ezquer M, Deis R, Maldonado C, Soaje M. Pituitary changes involved in prolactin secretion induced by mifepristone and naloxone during late pregnancy. Neuroendocrinology 2009;89:200–9.
Jurcovicova J, Kvetnansky R, Dobrakovova M, Jezova D, Kiss A, Makara GB. Prolactin response to immobilization stress and hemorrhage: the effect of hypothalamic deafferentations and posterior pituitary denervation. Endocrinology 1990;126:2527–33.
Lookingland KJ, Gunnet JW, Toney TW, Moore KE. Comparison of the effects of ether and restraint stress on the activity of tuberoinfundibular dopaminergic neurons in female and male rats. Neuroendocrinology 1990;52:99–105.
Murai I, Ben Jonathan N. Prolactin secretion in posterior pituitary lobectomized rats: differential effects of 5-hydroxytryptophan and ether. Brain Res 1987;420:227–32.
Petraglia F, Vale W, Rivier C. Beta-endorphin and dynorphin participate in the stress-induced release of prolactin in the rat. Neuroendocrinology 1987;45:338–42.
Vuong C, Van Uum SH, O’Dell LE, Lutfy K, Friedman TC. The effects of opioids and opioid analogs on animal and human endocrine systems. Endocr Rev 2010;31:98–132.
Xu RK, McCann SM. Differential effects of naloxone on basal and stress-induced release of ACTH and prolactin in the male rat. Life Sci 1989;45:1591–9.
Degli Uberti EC, Petraglia F, Bondanelli M, Guo AL, Valentini A, Salvadori S, et al. Involvement of mu-opioid receptors in the modulation of pituitary–adrenal axis in normal and stressed rats. J Endocrinol Invest 1995;18:1–7.
Kiritsy-Roy JA, Appel NM, Bobbitt FG, Van Loon GR. Effects of mu-opioid receptor stimulation in the hypothalamic paraventricular nucleus on basal and stress-induced catecholamine secretion and cardiovascular responses. J Pharmacol Exp Ther 1986;239:814–22.
Jahn GA, Deis RP. Stress-induced prolactin release in female, male and androgenized rats: influence of progesterone treatment. J Endocrinol 1986;110: 423–8.
Deis RP, Leguizamon E, Jahn GA. Feedback regulation by progesterone of stress-induced prolactin release in rats. J Endocrinol 1989;120:37–43.
Poletini MO, Szawka RE, Franci CR, Anselmo-Franci JA. Ovarian steroids but not the locus coeruleus regulate stress-induced prolactin secretion in female rats. J Neuroendocrinol 2006;18:938–48.
Bregonzio C, Navarro CE, Donoso AO. NMDA receptor antagonists block stress-induced prolactin release in female rats at estrus. Eur J Pharmacol 1998;350:259–65.
Valdez SR, Bonafede M, Carreño N,Deis R, Jahn G. Lactation deficit in OFA hr/hr rats may be caused by differential sensitivity to stress compared with Wistar and Sprague Dawley rats. Stress 2012;15:361–77.
Cho BN, Suh YH, Yoon YD, Lee CC, Kim K. Progesterone inhibits the estrogen-induced prolactin gene expression in the rat pituitary. Mol Cell Endocrinol 1993;93:47–52.
Russell JA, Douglas AJ, Brunton PJ. Reduced hypothalamo–pituitary–adrenal axis stress responses in late pregnancy: central opioid inhibition and noradrenergic mechanisms. Ann N Y Acad Sci 2008;1148:428–38.
Brunton PJ, McKay AJ, Ochedalski T, Piastowska A, Rebas E, Lachowicz A, et al. Central opioid inhibition of neuroendocrine stress responses in pregnancy in the rat is induced by the neurosteroid allopregnanolone. J Neurosci 2009;29:6449–60.
Grattan DR, Steyn FJ, Kokay IC, Anderson GM, Bunn SJ. Pregnancy-induced adaptation in the neuroendocrine control of prolactin secretion. J Neuroendocrinol 2008;20:497–507.
Brunton PJ, Russell JA. The expectant brain: adapting for motherhood. Nat Rev Neurosci 2008;9:11–25.
Brunton PJ, Russell JA. Neuroendocrine control of maternal stress responses and fetal programming by stress in pregnancy. Prog Neuropsychopharmacol Biol Psychiatry 2011;35:1178–91.
Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 2nd ed. Sidney: Academic Press; 1986.
Jahn GA, Alonso N, Deis RP. Ovarian and feto-placental factors and the regulation of prolactin release during pregnancy in the rat. J Reprod Fertil 1986;77:125–33.
Severino GS, Fossati IA, Padoin MJ, Gomes CM, Trevizan L, Sanvitto GL, et al. Effects of neonatal handling on the behavior and prolactin stress response in male and female rats at various ages and estrous cycle phases of females. Physiol Behav 2004;81:489–98.
Soaje M, de Di Nasso EG, Deis RP. Regulation by endogenous opioids of suckling-induced prolactin secretion in pregnant and lactating rats: role of ovarian steroids. J Endocrinol 2002;172:255–61.
Niswender GD, Chen CL, Midgley Jr AR, Meites J, Ellis S. Radioimmunoassay for rat prolactin. Proc Soc Exp Biol Med 1969;130:793–7.
Robertson MC, Friesen HG. Two forms of rat placental lactogen revealed by radioimmunoassay. Endocrinology 1981;108:2388–90.
Vermouth NT, Deis RP. Prolactin release induced by prostaglandin F2 in pregnant rats. Nat New Biol 1972;238:248–50.
Lee Y, Voogt JL. Feedback effects of placental lactogens on prolactin levels and Fos-related antigen immunoreactivity of tuberoinfundibular dopaminergic neurons in the arcuate nucleus during pregnancy in the rat. Endocrinology 1999;126:2159–66.
Milenkovic L, Bogic L, Martinovic JV. Effects of oestradiol and progesterone on stress-induced secretion of prolactin in ovariectomized and/or adrenalectomized female rats. Acta Endocrinol (Copenh) 1986;112:79–82.
Demarest KT, Moore KE, Riegle GD. Acute restraint stress decreases dopamine synthesis and turnover in the median eminence: a model for the study of the inhibitory neuronal influences on tuberoinfundibular dopaminergic neurons. Neuroendocrinology 1985;41:437–44.
Neill DJ, Nagy GM. Prolactin secretion and its control. In: Knobil E, Neill JD, editors. The physiology of reproduction. New York: Raven Press; 1994 . p. 1833–60.
Moore KE, Demarest KT, Lookingland KJ. Stress, prolactin and hypothalamic dopaminergic neurons. Neuropharmacology 1987;26:801–8.
Campbell RE, Grove KL, Smith MS. Distribution of corticotropin releasing hormone receptor immunoreactivity in the rat hypothalamus: coexpression in neuropeptide Y and dopamine neurons in the arcuate nucleus. Brain Res 2003;973:223–32.
Gala RR. The physiology and mechanisms of the stress-induced changes in prolactin secretion in the rat. Life Sci 1990;46:1407–20.
Buckingham JC, Cooper TA. Interrelationships of opioidergic and adrenergic mechanisms controlling the secretion of corticotrophin releasing factor in the rat. Neuroendocrinology 1987;46:199–206.
Van de Kar LD, Blair ML. Forebrain pathways mediating stress-induced hormone secretion. Front Neuroendocrinol 1999;20:1–48.
Hrabovszky E, Liposits Z. Adrenergic innervation of dopamine neurons in the hypothalamic arcuate nucleus of the rat. Neurosci Lett 1994;182: 143–6.
Jahn GA, Deis RP. Involvement of the adrenergic system on the release of prolactin and lactogenesis at the end of pregnancy in the rat. J Endocrinol 1991;129:343–50.
Seltzer AM, Donoso AO, Podesta E. Restraint stress stimulation of prolactin and ACTH secretion: role of brain histamine. Physiol Behav 1986;36:251–5.
Villegas-Gabutti C, Jahn G, Soaje M. Ocitocina (OT) participa en la acción estimuladora de Mifepristone y Naloxona sobre la secreción de Prolactina (PRL). Medicina 2009;69:132.
Arbogast LA, Voogt JL. Progesterone reverses the estradiol-induced decrease in tyrosine hydroxylase mRNA levels in the arcuate nucleus. Neuroendocrinology 1993;58:501–10.
Lerant A, Herman ME, Freeman ME. Dopaminergic neurons of periventricular and arcuate nuclei of pseudopregnant rats: semicircadian rhythm in Fos-related antigens immunoreactivities and in dopamine concentration. Endocrinology 1996;137:3621–8.
McKay D, Pasieka C, Moore KE, Riegle GD, Demarest KT. Semicircadian rhythm of tuberoinfundibular dopamine neuronal activity during early pregnancy and pseudopregnancy in the rat. Neuroendocrinology 1982;34:229.
Mao J, Regelson W, Kalimi M. Molecular mechanism of RU 486 action: a review. Mol Cell Biochem 1992;109:1–8.
Kiem DT, Bartha L, Makara GB. Effect of dexamethasone implanted in different brain areas on the morphine-induced PRL, GH and ACTH/corticosterone secretion. Brain Res 1991;563:107–13.
Villegas-Gabutti C, Ortiz JM, Soaje M. Estradiol modula la acción estimuladora de Mifepristone y Naloxona sobre la secreción de Prolactina. Medicina 2007;67:223.
Kapoor R, Willoughby JO. Activation of mu opioid receptors in the pre-opticanterior hypothalamus releases prolactin in the conscious male rat. Neuropeptides 1991;19:231–6.
Tavakoli-Nezhad M, Arbogast LA. Mu and kappa opioid receptor expression in the mediobasal hypothalamus and effectiveness of selective antagonists on prolactin release during lactation. Neuroscience 2010;166:359–67.
Yamamoto M, Komori T, Matsumoto T, Zhang K, Miyahara S, Shizuya K, et al. Effects of single and repeated prolonged stress on mu-opioid receptor mRNA expression in rat gross hypothalamic and midbrain homogenates. Brain Res 2003;980:191–6.
Sanchez MD, Milanes MV, Pazos A, Diaz A, Laorden ML. Autoradiographic evidence of mu-opioid receptors down-regulation after prenatal stress in offspring rat brain.Brain Res. Dev Brain Res 1996;94:14–21.
Stuckey J, Marra S, Minor T, Insel TR. Changes in mu opiate receptors following inescapable shock. Brain Res 1989;476:167–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valdez, S.R., Pennacchio, G.E., Gamboa, D.F. et al. Opioid modulation of prolactin secretion induced by stress during late pregnancy. Role of ovarian steroids. Pharmacol. Rep 66, 386–393 (2014). https://doi.org/10.1016/j.pharep.2013.12.006
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2013.12.006