Abstract
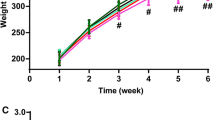
Anoectochilus roxburghii (Wall.) Lindl., Orchidaceae, is a Chinese medicinal plant which can be effective for some diseases such as hepatitis, nephritis, pneumonia. Its active ingredient is kinsenoside. The mechanisms of kinsenoside on the liver-protective effect have not been fully explored until today. The present study was aimed to investigate the protective effect and mechanism of kinsenoside on acute alcoholic liver injury. The protected activity of kinsenoside (10, 20 and 40 mg/kg) were investigated on acute alcoholic liver injury in mice. Male C57BL/6 J mice were fed with non-fat feed for 30 days and oral administrated 14 ml/kg bw of ethanol (50%) on the 31st day. The activities of serum aspartate aminotransferase, serum alanine aminotransferase, triacylglyceride and very low density lipoprotein were determined in serum. The hepatic levels of oxidative stress as glutathione, malondialdehyde were measured in liver homogenates. The levels of cytochrome P450 2E1 (CYP2E1) were measured by immunohistochemistry. Furthermore, histopathological observations were carried out on the separated livers of mice. It was suggested that the trends of acute hepatic injury and fatty degeneration induced by alcohol were reduced in the ethanol group after kinsenoside treatment. Compared to ethanol groups, triacylglyceride, malondialdehyde, very low density lipoprotein, reduced glutathione, serum alanine aminotransferase and serum aspartate aminotransferase levels of kinsenoside (20, 40 mg/kg) groups were decreased (p < 0.05). Meanwhile kinsenoside significantly decreased the level of protein CYP2E1. In conclusion, kinsenoside enhances antioxidant capacity of mice and antagonizes alcohol-induced lipid metabolism disorders. Besides, kinsenoside inhibits alcohol-caused hepatocyte apoptosis, reduces oxidative stress, and relieves hepatocyte death, which may be a mechanism of kinsenoside in the treatment of alcoholic liver.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Brodsky, J.L., Fisher, E.A., 2008. The many intersecting pathways underlying apolipoprotein B secretion and degradation. Trends Endocrinol. Metab. 19, 254–259, https://doi.org/10.1016/j.tem.2008.07.002.
Browning, J.D., Szczepaniak, L.S., Dobbins, R., Nuremberg, P., Horton, J.D., Cohen, J.C., Grundy, S.M., Hobbs, H.H., 2004. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology 40, 1387–1395, https://doi.org/10.1002/hep.20466.
Cao, Y.W., Jiang, Y., Zhang, D.Y., Wang, M., Chen, W.S., Su, H., Wang, Y.T., Wan, J.B., 2015. Protective effects of Penthorum chinense Pursh against chronic ethanol-induced liver injury inmice. J. Ethnopharmacol. 161, 92–98, https://doi.org/10.1016/jjep.2014.12.013.
Cederbaum, A.I., Kessova, I., 2003. CYP2E1: biochemistry, toxicology, regulation and function in ethanol-induced liver injury. Curr. Mol. Med. 3, 509–518, https://doi.org/10.1111/j.1939-0025.1956.tb06158.x.
Chen, P., Miyamoto, Y., Mazagova, M., Lee, K.C., Eckmann, L. Schnabl, B., 2015. Micro-biota protects mice against acute alcohol-induced liver injury. Alcohol. Clin. Exp. Res. 39, 2313–2323, https://doi.org/10.1111/acer.12900.
Chen, Y.Y., Zhang, C.L., Zhao, X.L., Xie, K.Q., Zeng, T., 2014. Inhibition of cytochrome P4502E1 by chlormethiazole attenuated acute ethanol-induced fatty liver. Chem. Biol. Interact. 222, 18–26, https://doi.org/10.1016/j.cbi.2014.08.009.
Das, S.K., Vasudevan, D.M., 2007. Alcohol-induced oxidative stress. Life Sci. 81, 177–187, https://doi.org/10.1016/j.lfs.2007.05.005.
Ding, R.B., Tian, K., Cao, Y.W., Bao, J.L., Wang, M., He, C., Hu, Y., Su, H., Wan, J.B., 2015. Protective effect of Panax notoginseng saponins on acute ethanol-induced liver injury is associated with ameliorating hepatic lipid accumulation and reducing ethanol-mediated oxidativestress. J. Agric. Food Chem. 63, 2413–2422, https://doi.org/10.1021/jf502990n.
Ding, R.B., Tian, K., Huang, L.L., He, C.W., Jiang, Y., Wang, Y.T., Wan, J.B., 2012. Herbal medicines for the prevention of alcoholic liver disease: areview. J. Ethnopharmacol. 144, 457–465, https://doi.org/10.1016/jjep.2012.09.044.
Gan, D., Ma, L., Jiang, C., Wang, M., Zeng, X., 2012. Medium optimization and potential hepatoprotective effect of mycelial polysaccharides from Pholiota dinghuensis Bi against carbon tetrachloride-induced acute liver injury in mice. Food Chem. Toxicol. 50, 2681–2688, https://doi.org/10.1016/j.fct.2012.05.003.
Gerbal-Chaloin, S., Dume, A.S., Briolotti, P., Klieber, S., Raulet, E., Duret, C., Fabre, J.M., Ramos, J., Maurel, P., Daujat-Chavanieu, M., 2014. The WNT/Catenin pathway is a transcriptional regulator of CYP2E1, CYP1A2, and aryl hydrocarbon receptor gene expression in primary human hepatocytes. Mol. Pharmacol. 86, 624–634, https://doi.org/10.1124/mol.114.094797.
Groll, N., Petrikat, T., Vetter, S., Colnot, S., Weiss, F., Poetz, O., Joos, T.O., Rothbauer, U., Schwarz, M., Braeuning, A., 2016. Coordinate regulation of Cyp2e1 by ß-catenin-and hepatocyte nuclear factor 1a-dependent signaling. Toxicology 350–352, 40–48, https://doi.org/10.1016/j.tox.2016.05.004.
Guo, Y., Xiao, L., Sun, L., Liu, F., 2012. Wnt/ß-catenin signaling: A promising new target for fibrosis diseases. Physiol. Res. 61, 337–346.
Huang, CK., Yu, T., De La Monte, S.M., Wands, J.R., Derdak, Z., Kim, M., 2015. Restoration of Wnt/ß-catenin signaling attenuates alcoholic liver disease progression in a ratmodel. J. Hepatol. 63, 191–198, https://doi.org/10.1016/jjhep.2015.02.030.
Jiang, Z., Wang, J., Xue, H., Wang, M., Jiang, H., Liang, Y., Dias, A.C., Gregory, M., Chen, C., Zhang, X., 2017. Protective effect of wild Corni fructus methanolic extract against acute alcoholic liver injury in mice. Redox Rep. 22, 338–345, https://doi.org/10.1080/13510002.2016.1239867.
Lieber, CS., 2004. The discovery of the microsomal ethanol oxidizing system and its physiologic and pathologic role. Drug Metab. Rev. 36, 511–529, https://doi.org/10.1081/DMR-200033441.
Liu, Q., Qiao, A.M., Yi, L.T., Liu, Z.L., Sheng, S.M., 2016. Protection of kinsenoside against AGEs-induced endothelial dysfunction in human umbilical vein endothelial cells. Life Sci. 162, 102–107, https://doi.org/10.1016/j.lfs.2016.08.022.
Loomba, R., Sanyal, A.J., 2013. The global NAFLD epidemic. Nat. Rev. Gastroenterol. Hepatol. 10, 686–690, https://doi.org/10.1038/nrgastro.2013.171.
Ma, L., Gan, D., Wang, M., Zhang, Z., Jiang, C., Zeng, X., 2012. Optimization of extraction, preliminary characterization and hepatoprotective effects of polysaccharides fromStachys floridana Schuttl. ex Benth. Carbohydr. Polym. 87, 1390–1398, https://doi.org/10.1016/j.carbpol.2011.09.032.
Mathurin, P., Bataller, R., 2015. Trends in the management and burden of alcoholic liverdisease. J. Hepatol. 62, S38–S46, https://doi.org/10.1016/jjhep.2015.03.006.
Nada, S.A., Omara, E.A., Abdel-Salam, O.M.E., Zahran, H.G., 2010. Mushroom insoluble polysaccharides prevent carbon tetrachloride-induced hepatotoxicity in rat. Food Chem. Toxicol. 48, 3184–3188, https://doi.org/10.1016/j.fct.2010.08.019.
Nieto, N., Friedman, S.L., Cederbaum, A.I., 2002. Cytochrome P450 2E1-derived reactive oxygen species mediate paracrine stimulation of collagen I protein synthesis by hepatic stellate cells. J. Biol. Chem. 277, 9853–9864, https://doi.org/10.1074/jbc.M110506200.
Qi, Cxing, Zhou, Q., Yuan, Z., Luo, Z.wei, Dai, C., Zhu, H.cheng, Chen, C.mei, Xue, Y.bo, Wang, Jping, Wang, Yfen, Liu, Yping, Xiang, M., Sun, W.guang, Zhang, Jwen, Zhang, Y.hui, 2018. Kinsenoside: a promising bioactive compound from Anoectochilus species. Curr. Issues Pharm. Med. Sci. Pract. 38, 11–18, https://doi.org/10.1007/s11596-018-1841-1.
Shih, CC., Wu, Y.W., Lin, W.C., 2005. Aqueous extract of Anoectochilus for-mosanus attenuate hepatic fibrosis induced by carbon tetrachloride in rats. Phytomedicine 12, 453–460, https://doi.org/10.1016/j.phymed.2004.02.008.
Stewart, S., Jones, D., Day, C.P., 2001. Alcoholic liver disease: new insights into mechanisms and preventative strategies. Trends Mol. Med. 7, 408–413, https://doi.org/10.1016/S1471-4914(01)02096-2.
Wang, Y., Millonig, G., Nair, J., Patsenker, E., Stickel, F., Mueller, S., Bartsch, H., Seitz, H.K., 2009. Ethanol-induced cytochrome P4502E1 causes carcinogenic etheno-DNA lesions in alcoholic liver disease. Hepatology 50, 453–461, https://doi.org/10.1002/hep.22978.
Wang, Z., Su, B., Fan, S., Fei, H., Zhao, W., 2015. Protective effect of oligomeric proanthocyanidins against alcohol-induced liver steatosis and injury in mice. Biochem. Biophys. Res. Commun. 458, 757–762, https://doi.org/10.1016/j.bbrc.2015.01.153.
Wu, D., Cederbaum, A.I., 2013. Inhibition of autophagy promotes CYP2E1-dependent toxicity in HepG2 cells via elevated oxidative stress, mitochondria dysfunction and activation of p38 and JNK MAPK. Redox Biol. 1, 552–565, https://doi.org/10.1016/j.redox.2013.10.008.
Wu, H.T., He, X.J., Hong, Y.K., Ma, T., Xu, Y.P., Li, H.H., 2010. Chemical characterization of Lycium barbarum polysaccharides and its inhibition against liver oxidative injury of high-fat mice. Int. J. Biol. Macromol. 46, 540–543, https://doi.org/10.1016/j.ijbiomac.2010.02.010.
Yang, L., Rozenfeld, R., Wu, D., Devi, L.A., Zhang, Z., Cederbaum, A., 2014. Cannabidiol protects liver from binge alcohol-induced steatosis by mechanisms including inhibition of oxidative stress and increase in autophagy. Free Radic. Biol. Med. 68, 260–267, https://doi.org/10.1016/j.freeradbiomed.2013.12.026.
Yang, M.X., Cederbaum, A.I., 1997. Glycerol increases content and activity of human cytochrome P-4502E1 in a transduced HepG2 cell line by protein stabilization. Alcohol. Clin. Exp. Res. 21, 340–347, https://doi.org/10.1111/j.1530-0277.1997.tb03770.x.
Zeng, B., Su, M., Chen, Q., Chang, Q., Wang, W., Li, H., 2017. Protective effect of a polysaccharide from Anoectochilus roxburghii against carbon tetrachloride-induced acute liver injury inmice. J. Ethnopharmacol. 200, 124–135, https://doi.org/10.1016/jjep.2017.02.018.
Zeng, T., Guo, F.F., Zhang, C.L., Song, F.Y., Zhao, X.L., Xie, K.Q., 2013. Roles of cytochrome P4502E1 gene polymorphisms and the risks of alcoholic liver disease: a meta-analysis. PLoS One 8, https://doi.org/10.1371/journal.pone.0054188.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
SZ and JZ contributed to the conception of the study. SZ and YW contributed significantly to analysis, and manuscript preparation. QZ performed the data analyses and wrote the manuscript. LY helped perform the analysis with constructive discussions. YZ and JZ contributed to project administration and responses.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zou, S., Wang, Y., Zhou, Q. et al. Protective effect of kinsenoside on acute alcohol-induced liver injury in mice. Rev. Bras. Farmacogn. 29, 637–643 (2019). https://doi.org/10.1016/j.bjp.2019.06.006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2019.06.006