Abstract
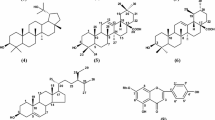
The phytochemical study of the extract leaves from Maytenus distichophylla Mart. and Salacia crassifolia (Mart. ex Schult.) G. Don, Celastraceae, resulted in the isolation of 3-oxofriedelane, 3β-hydroxyfriedelane, 3β,24-dihydroxyfriedelane, 3-oxo-28,29-dihydroxyfriedelane, two mixtures of pentacyclic triterpenes (α-amyrin with β-amyrin and 3β-stearyloxy-urs-12-ene with 3β-stearyloxy-olean-12-ene), 3β-palmityloxy-urs-12-ene, the steroid β-sitosterol and its glycosylated derivative β-glucosyl-β-sitosterol, tritriacontanoic acid and the natural polymer gutta percha. The chemical structures of these constituents were established by IR, 1H and 13C NMR spectral data. Crude extracts, the mixtures of triterpenes and the isolated constituents were subjected to in vitro acetylcholinesterase inhibitory evaluation. Acetylcholinesterase inhibitory effect was observed for crude chloroform extract leaves from M. distichophylla (100%) and S. crassifolia (97.93 ± 5.63%) and for the triterpenes 3β,24-dihydroxyfriedelane (99.05± 1.12%), 3-oxo-28,29-dihydroxyfriedelane (90.59±3.76%) and 3β-palmityloxy-urs-12-ene (97.93 ± 1.47%). The percent inhibitions induced by these natural products were very similar to those produced by physostigmine (93.94 ±2.10%) a standard acetylcholinesterase inhibitor. Therefore, these results open perspectives for the use of these species as source of compounds with similar physostigmine pharmacological effect.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alarcón, J., Astudillo, L., Gutierrez, M., 2008. Inhibition of acetylcholinesterase activity by dihydroagarofuran sesquiterpenes isolated from Chilean Celastraceae. Z. Naturforsch. 63c, 853–856.
Alarcón, J., Céspedes, C.L., Muñoz, E., Balbontin, C., Valdes, F., Gutierrez, M., Astudillo, L., Seigler, D.S., 2015. Dihydroagarofuranoid sesquiterpenes as acetylcholinesterase inhibitors from Celastraceae plants: Maytenus disticha and Euonymus japonicus. J. Agric. Food Chem. 63, 10250–10256.
Costa, P.M., Carvalho, M.G., 2003. New triterpene isolated from Eschweilera longipes (Lecythidaceae). An. Acad. Bras. Cienc. 75, 21–25.
Cramer, P.E., Cirrito, J.R., Wesson, D.W., Lee, C.Y.D., Karlo, J.C., Zinn, A.E., Casali, B.T., Restivo, J.L., Goebel, W.D., James, M.J., Brunden, K.R., Wilson, D.A., Landreth, G.E., 2012. ApoE-directed therapeutics rapidly clear β-amyloid and reverse deficits in ad mouse models. Science 335, 1503–1506.
De Andrade, S.F., Lemos, M., Comunello, E., Noldin, V.F., Chechinel-Filho, V., Niero, R., 2007. Evaluation of the antiulcerogenic activity of Maytenus robusta (Celastraceae) in different experimental ulcer models. J. Ethnoparmacol. 113, 252–257.
Ellman, G.L., Courtney, K.D., Andres, V., Featherstone, R.M., 1961. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95.
Filho, J.M.B., Medeiros, K.C.P., Diniz, M.F., Batista, L.M., Athayde-Filho, P.F., Silva, M.S., Cunha, E.V.L., Almeida, J.R.G.S., Quintans-Junior, L.J., 2006. Natural products inhibitors of the enzyme acetylcholinesterase. Rev. Bras. Farmacogn. 16, 258–285.
Gurovic, M.S.V., Castro, M.J., Richmond, V., Faraoni, M.B., Maier, M.S., Murray, A.P., 2010. Triterpenoids with acetylcholinesterase inhibition from Chuquiraga erinacea D. Don. subsp. erinacea (Asteraceae). Planta Med. 76, 607–610.
Hamdan, D., Wink, M., El-Shazly, A., 2014. Secondary metabolites isolated from dichloromethane fraction of rough lemon stem and hepatoprotective effect of limonianin. Br. J. Pharm. Res. 4, 1963–1975.
Lendl, A., Werner, I., Glasl, S., Kletter, C., Mucaji, P., Presser, A., Reznicek, G., Jurenitsch, J., Taylor, D.W., 2005. Phenolic and terpenoid compounds from Chione venosa (sw.) Urban var. venosa (Bois Bandé). Phytochemistry 66, 2381–2387.
Liu, J.-Q., Peng, X.R., Li, X.-Y., Li, T.Z., Zhang, W.-M., Shi, L., Han, J., Qiu, M.H., 2013. NorfriedelinsA-Cwith acetylcholinesterase inhibitory activity from acerola tree (Malpighiaemarginata).Org. Lett. 15, 1580–1583.
Mahato, S.B., Kundu, A.P., 1994. 13C NMR spectra of pentacyclic triterpenoids - a compilation and some salient features. Phytochemistry 37, 1517–1575.
Miranda, R.R.S., Silva, G.D.F., Duarte, L.P., Fortes, I.C.P., Vieira-Filho, S.A., 2006. Structural determination of 3β-stearyloxy-urs-12-ene from Maytenus salicifolia by 1D and 2D NMR and quantitative 13C NMR spectroscopy. Magn. Reson. Chem. 44, 127–131.
Oliveira, D.M., Silva, G.D.F., Duarte, L.P., Vieira-Filho, S.A., 2006. Chemical constituents isolated from roots of Maytenus acanthophylla Reissek (Celastraceae). Biochem. Syst. Ecol. 34, 661–665.
Omena, M.R.L.A., 2009. Ensaio etnofarmacológico de espécies vegetais com ação no sistema nervoso central, originárias do bioma caatinga. Saúde Ambient. Rev. 2, 92–95.
Park, S.Y., 2010. Potencial therapeutic agents against Alzheimer’s disease from natural sources. Arch. Pharm. Res. 33, 1589–1609.
Park, S.J., Jung, J.M., Lee, H.E., Lee, Y.W., Kim, D.H., Kim, J.M., Hong, J.G., Lee, C.H., Jung, I.H., Cho, Y.B., Jang, D.S., Ryu, J.H., 2012. The memory ameliorating effects of INM-176, an ethanolic extract of Angelica gigas, against scopolamine-or Aβ(1–42)-induced cognitive dysfunction in mice. J. Ethnopharmacol. 142, 611–620.
Rodrigues, V.G., Silva, F.C., Duarte, L.P., Takahashi, J.A., Matildes, B.L.G., Silva, G.D.F., Silva, R.R., Vieira-Filho, S.A., 2014. Pentacyclic triterpenes from Maytenus genus as acetylcholinesterase inhibitors. Int. J. Pharm. Pharm. Sci. 6, 918–920.
Santos, V.L., Costa, V.B.M., Agra, M.F., Silva, B.A., Batista, L.M., 2007. Pharmacological studies of ethanolic extracts of Maytenus rigida Mart (Celastraceae) in animal models. Rev. Bras. Farmacogn. 17, 336–342.
Silva, F.C., Oliveira, M.L.G., Rodrigues, V.G., Carvalho, S.M., Duarte, L.P., Silva, G.D.F., Miranda, R.R.S., Figueiredo, R.C., Moraes, J.C., Vieira-Filho, S.A., 2013. Triterpenes from Maytenus gonoclada and their attractive effects on Tenebrio molitor. Chem. Nat. Compd. 49, 571–574.
Sousa, G.F., Duarte, L.P., Alcântara, A.F.C., Silva, G.D.F., Vieira-Filho, S.A., Silva, R.R., Oliveira, D.M., Takahashi, J.A., 2012. New triterpenes from Maytenus robusta: structural elucidation based on NMR experimental data and theoretical calculations. Molecules 17, 13439–13456.
Sousa, G.F., Aguilar, M.G., Takahashi, J.A., Alves, T.M.A., Kohlhoff, M., Vieira Filho, S.A., Silva, G.D.F., Duarte, L.P., 2016. Flavonol triglycosides of leaves from Maytenus robusta with acetylcholinesterase inhibition. Phytochem. Lett. 19, 34–38.
Triggle, D.J., Mitchell, J.M., Filler, R., 1998. The pharmacology of physostigmine. CNS Drug Rev. 4, 87–136.
Vieira-Filho, S.A., Duarte, L.P., Silva, G.D.F., Howarth, O.W., Lula, I.S., 2003. 3β-(Stearyloxy) olean-12-ene from Austroplenckia populnea: structure elucidation by 2D-NMR and quantitative 13C-NMR spectroscopy. Helv. Chim. Acta 86, 3445–3449.
Weeratunga, G., Kumar, V., Sultanbawa, M.U.S., Balasubramaniam, S., 1982. 28,29-Dihydroxyfriedelan-3-one, a friedelane with two oxygenated methyl groups, from Elaeodendron balae (Celastraceae). J. Chem. Soc. Perkin Trans. 1, 2457–2459.
Yang, Z.D., Duan, D.Z., Xue, W.W., Yao, X.J., Li, S., 2012. Steroidal alkaloids from Holarrhena antidysenterica as acetylcholinesterase inhibitors and the investigation for structure-activity relationships. Life Sci. 90, 929–933.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
FLF, VGR, FCS, GDFS and LPD contributed to the experimental work, extraction and isolation of the metabolites from plants, the interpretation of all these data and wrote the manuscript. SAVF and DMO wrote and revised the manuscript. BLGM and JAT conducted the test in vitro AChE inhibitory activity. All authors read and approved the final manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ferreira, F.L., Rodrigues, V.G., Silva, F.C. et al. Maytenus distichophylla and Salacia crassifolia: source of products with potential acetylcholinesterase inhibition. Rev. Bras. Farmacogn. 27, 471–474 (2017). https://doi.org/10.1016/j.bjp.2016.12.006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2016.12.006