Abstract
Bone pain typically occurs immediately following skeletal damage with mechanical distortion or rupture of nociceptive fibres. The pain mechanism is also associated with chronic pain conditions where the healing process is impaired. Any load impacting on the area of the fractured bone will stimulate the nociceptive response, necessitating rapid clinical intervention to relieve pain associated with the bone damage and appropriate mitigation of any processes involved with the loss of bone mass, muscle, and mobility and to prevent death. The following review has examined the mechanisms of pain associated with trauma or cancer-related skeletal damage focusing on new approaches for the development of innovative therapeutic interventions. In particular, the review highlights tissue engineering approaches that offer considerable promise in the application of functional biomimetic fabrication of bone and nerve tissues. The strategic combination of bone and nerve tissue engineered models provides significant potential to develop a new class of in vitro platforms, capable of replacing in vivo models and testing the safety and efficacy of novel drug treatments aimed at the resolution of bone-associated pain. To date, the field of bone pain research has centred on animal models, with a paucity of data correlating to the human physiological response. This review explores the evident gap in pain drug development research and suggests a step change in approach to harness tissue engineering technologies to recapitulate the complex pathophysiological environment of the damaged bone tissue enabling evaluation of the associated pain-mimicking mechanism with significant therapeutic potential therein for improved patient quality of life.
Graphical abstract
Rationale underlying novel drug testing platform development. Pain detected by the central nervous system and following bone fracture cannot be treated or exclusively alleviated using standardised methods. The pain mechanism and specificity/efficacy of pain reduction drugs remain poorly understood. In vivo and ex vivo models are not yet able to recapitulate the various pain events associated with skeletal damage. In vitro models are currently limited by their inability to fully mimic the complex physiological mechanisms at play between nervous and skeletal tissue and any disruption in pathological states. Robust innovative tissue engineering models are needed to better understand pain events and to investigate therapeutic regimes

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The increase in life expectancy as a consequence of medical advances over the past decades has also heralded an emergence of pathologies correlated to the ageing demographic [1, 2]. Acquired skeletal diseases such as osteoporosis, cancer metastasis and multiple myeloma, and genetic disorders such as osteogenesis imperfecta and fibrous dysplasia of bone are known to cause bone fragility as a consequence of osteopenia or locally enhanced bone resorption [3, 4]. Thus, skeletal disorders often lead to bone fractures, resulting in extremely painful consequences for the patient both during movement or at rest [5]. For instance, the initial skeletal damage elicited by primary bone tumours or metastasis typically facilitates the fracture of diseased bone tissue with a consequent inflammation response and sudden pain. During the progress of the pathological state, cancer bone pain becomes more perceptible, becoming progressively constant and intense [5]. Pathological pain is generally perceived at the site of injury or often as referred pain with muscle spasms together with an extensive range of action, involving multiple sites other than the site of lesion [6, 7].
As well as pathological conditions, bone fracture/trauma is often a consequence of damage as a result of sporting activities [1]. Sports-related fractures, especially in the upper limbs, are currently the third leading cause of bone fractures, affecting a wide demographic including the young [1, 8]. Furthermore, reduced physical activity, poor nutrition (such as limited vitamin D3 intake) and sedentary lifestyles can impact on bone structure, increasing the risk of bone fracture and impaired skeletal tissue composition [9,10,11,12]. There remains an unmet need for improved pain management programmes to (i) ease the pain, (ii) improve tissue healing and (iii) increase the quality of the life of hospitalised patients [13]. To date, the complexity of the pain process has limited clinical options, as current therapies often aim to alleviate the discomfort rather than remove or resolve the pain [13].
The current review explores the pathophysiology and the processes involved in pain mechanisms affecting bone tissue, from central causes related to pathological and damage-related pain to the evaluation of current approaches and therapeutic analysis. Furthermore, the review examines currently available functional in vivo and in vitro models, highlighting the most effective platforms involved in delineating the pain processes. Ultimately, a focus on current research in the skeletal pain modelling platforms is presented and the rich vistas of opportunity therein together with a detailed evaluation of future trends and perspectives for the engineering of in vitro skeletal disruption and pain.
Pathophysiology of cancer and trauma fracture bone pain
Unlike other types of physical discomfort, the pathogenesis of bone pain is still poorly understood due to the complexity of the underlying functional mechanisms [6]. Indeed, pain affecting the musculoskeletal system does not occur simply through a mechanical distortion of the nociceptive fibres but involves a plethora of supplementary mechanisms [13] (Fig. 1).
Bone pain mechanism. Bone pain cycle between osteoclasts, cancer cells and bone fractures in osteolytic metastases. Bone-derived growth factors promote proliferation and stimulate epithelial–mesenchymal transition (EMT) of cancer cells and the production of bone-modifying cytokines, in bone-colonising cancer cells. These factors further stimulate osteoclastic bone resorption via activation of the receptor activator of the nuclear factor-kB (RANKL)/RANK pathway in osteoblasts and osteoclasts, increasing the release of bone-stored growth factors. Osteoclast activity induces a local acidosis increasing TRPV1 and ASIC3 ion channel activity of nociceptor fibres. The response is sent to the central nervous system (CNS) via the dorsal root ganglion (DRG) together with mechanical stimulation due to the growing tumour mass–induced bone fractures
Mechanisms of cancer bone pain
Cancer bone pain mechanisms are complex and heterogeneous typically involving elements of inflammatory and neuropathic pain, with neuro-chemical changes from peripheral, spinal and central sites [7]. The inflammatory process arising from the bone lesion produced by the growing tumour mass involves cancer cells which release pain mediators, contributing to attract additional immune cells to the lesion site further stimulating nociceptive fibres [7]. Osteolytic lesions are characterised by a rapid resorption of the bone resulting in bone fragility and subsequent fracture risk [14]. Osteoclastogenesis is promoted by cancer cells via receptor activator of nuclear factor kappa-B ligand (RANKL) [14] stimulating osteoclast maturation, with a consequent reduction of the pH at the local micro-environment [7, 15].
The local acidosis, produced by osteoclast activity and cancer cell metabolism (Warburg effect), contributes to the sensitisation of the primary afferent neurons present in the bone [16]. The high local concentration of protons activates the sensing ion channels TRPV1 and ASIC3 of sensory neurons, increasing the nociceptive response, which is subsequently translated into an acid-evoked cancer bone pain event [7, 16]. Tumour expansion results in mechanical distortion of the bone including the stretching of the periosteum, the outer envelope of the cortical bone. The periosteum holds a high surface density of mechanosensitive sensory nerve fibres which, once stimulated, transmit the nociceptive response to the brain [4, 13, 17].
Osteoblastic lesions are typically associated with pathologies such as prostate cancer metastasis, Hodgkin’s lymphoma and medulloblastoma [18]. Each of these pathologies is characterised by the deposition of new bone, typically brittle in nature in comparison to the healthy bone tissue [19]. Moreover, the arrangement of collagen fibres has been reported to be random, indicative of woven bone with poor load distribution capacity and consequent risk of local (micro-) fractures and generation of bone pain [15]. To date, the mechanisms around the development of osteoblastic metastasis remain poorly understood [19]. The neuropathic component of bone pain may arise from cancer-induced damage to the sensory nerves caused by infiltration, compression by tumour cells, tumour-induced hyper-innervation and stretching or denervation as the lesion expands and the bone degrades [7, 16].
Mechanisms of trauma fracture bone pain
Bone fractures resulting from trauma are influenced by (i) the quality and density of the bone, (ii) the mechanical property of the bone affected and (iii) the application of the load [20]. During the traumatic event, especially in the case of a fall, bone is subjected to a multi-axial load condition, which can result in a fracture due to the multiple excess loads in an axial-torsion [21, 22]. Following fracture, the altered orientation of the damaged bone is perceived by mechanosensitive fibres as a mechanical stimulation [13]. The distortion of the cortical bone, or an increase in intraosseous pressure, activates the nociceptive C and A-δ fibres in the periosteum, responsible for the perception of dull pain and acute pain, respectively [23]. The initial nociceptive response to injury, as in cancer bone pain, is sent to the brain, generating the sensation of pain [1, 13, 17, 23]. Unlike cancer bone pain, the inflammatory component is due to the formation of a hematoma following the rupture of blood vessels accompanying the bone damage [24]. As in cancer bone pain, even in trauma-induced fracture, inflammatory-related factors can directly activate or sensitise nociceptors [13, 25]. Following bone fracture, any movement, or applied load, determines a mechanical stimulation of the sensory nerve fibres that innervate the periosteum, the mineralised bone and the bone marrow, generating a state of neuropathic pain [13, 26].
Nerve growth factor (NGF) is involved in the sensitisation and germination of nociceptor fibres [27, 28]. Following fracture, the neurotrophic factors, released by the inflammatory and stromal cells, induce an ectopic sprouting, resulting in hyper-innervation of the marrow, the mineralised bone and the periosteum. The enhanced nervous network makes any type of normal load or movement of the bone perceived as noxious [13]. Fracture realignment promotes healing with the levels of NGF, and sprouted nerve fibres reduced, restoring normal bone innervation and as a consequence pain relief [13]. During chronic pain, the concentration of inflammatory and neurotropic mediators increases, favouring excessive nerve sprouting leading to the formation of a neuroma-like structure, highly sensitive to any type of movement (mechanical allodynia) [1]. It is clear that fracture healing must occur as soon as possible in order that bone pain does not develop into a chronic pain condition.
Therapeutics and current standard treatment approaches for cancer and fracture bone pain
Optimised bone pain treatments are essential to improve patient quality of life in pathological conditions and to aid bone healing following trauma-induced bone fracture. Current pain reduction approaches include:
Radiotherapy
Typically applied for single metastases. The use of radiation aims to resize the tumour mass to reduce the chemical mediators that mediate bone pain [4]. It has been found that 70% of patients experience partial pain relief within 2 weeks with only 25% of patients experiencing total pain relief [5].
Bisphosphonates
Bisphosphonates (BPs) are often administered in combination with radiotherapy or in cases where the pain is not localised [5]. BPs can be used in addition to analgesics and in other metabolic bone diseases to reduce or prevent disease progression and to decrease disease symptoms and complications [3, 29, 30]. However, BP administration is accompanied by a number of side effects including flu-like symptoms (fever, arthralgia, myalgia and weakness), anaemia, nausea, dyspnoea, peripheral oedema and, in rare cases, osteonecrosis of the jaw [19, 30].
Monoclonal antibodies (mAbs)
Denosumab binds to RANKL preventing the interaction with the receptor on the osteoclasts and thus preventing osteoclast maturation and function and is one of the most successful mAb treatment options available [5, 19, 31, 32]. Tanezumab treatment is a potential alternative to denosumab, binding to NGF to prevent interaction with TrkA and p75 receptors [5, 33]. However, tanezumab is still under initial clinical assessment and has not yet been cleared for safe therapeutic application [34].
Analgesics
Non-steroidal anti-inflammatory drugs (NSAIDs) are administered in patients with mild or moderate bone pain [3]. NSAIDs inhibit prostaglandin (PG) synthesis acting on cyclooxygenase, reducing local oedema and PG-induced nociceptor sensitisation and local inflammation [3, 5, 35]. Following NSAID therapies, moderate or severe pain was reduced to mild pain after 2 weeks in 51% of patients [5]. Nevertheless, long-term treatments with NSAIDs can have significant detrimental effects on skeletal health as a consequence of inhibition of osteoblast growth due to cell cycle arrest and apoptosis induction [3, 36].
Corticosteroids
Dexamethasone (Dex) has been routinely used for the management of metastatic bone pain, neuropathic pain from infiltration or compression of neural structures [5, 37]. Corticosteroids exert potent anti-inflammatory effects and can directly decrease the impaired electrical activity of damaged peripheral neurons, decreasing the intensity of pain [38]. However, there remains a paucity of data on the efficacy of corticosteroids and current therapies are typically administered over limited time frames [5].
The complexity of the pain mechanism and the plethora of unsuccessful therapeutic approaches together with varied and often patient subjective response to pain necessitate new modelling and evaluation platforms. The development of appropriate modelling platforms offers new avenues to evaluate the safety and efficacy of novel drugs that could improve patient quality of life.
Tissue engineering models
Tissue engineering (TE) seeks to harness cells, engineering and materials together with biochemical and physico-chemical cues to restore, maintain, improve or replace tissues of interest. Cells in combination with 3D scaffolds offer a physiologically relevant environment to examine cell fate, tissue maturation or in situ regeneration as well as drug screening in vitro, ex vivo and in vivo [39, 40]. Cell approaches include incorporation of primary cells (terminally differentiated or stem cells) from patients that can be expanded in vitro and encapsulated in biocompatible water-based matrices, called hydrogels. Thus, biomimetic models capable of resembling the complex pathophysiological state of tissues are an important goal for the tissue engineer to generate physiologically relevant models and tissue constructs.
Cell sources
To closely mimic and recreate the tissue micro-environment, 3D models of an appropriate cell source are pivotal. Human bone marrow stromal cells (HBMSCs) are commonly used for bone tissue engineering purposes given their ready availability. HBMSCs contain a subset of cells, skeletal stem cells, that can self-renew and differentiate into cells of the stromal lineage namely, chondrocytes, osteoblasts and adipocytes [41,42,43]. Differentiated skeletal populations present a window into cellular changes due to specific pathologies from the affected donor although cell numbers are naturally limited given limited expansion capacity of a differentiated cell population [42].
The limited availability and paucity of robust methodologies for the isolation of neurons in human brain tissue have brought stem cells, specifically pluripotent stem cells (embryonic and induced pluripotent stem cells (ES and iPSCs)), to the fore as a pathway to derive and differentiate neuronal populations and for the generation of novel and functional in vitro models [43].
iPSCs have garnered significant interest given their potential as a novel cell source, avoiding crucial ethical issues, reproducibility and challenging isolation and expansion protocols. Indeed, iPSCs can be readily derived from human skin biopsies from patients and donors, and with ex vivo induction towards an embryonic-like cell state, iPSCs can subsequently differentiate into a wide spectrum of tissues [43, 44]. iPSCs are typically differentiated into neuroectodermal and neuronal cells [44, 45], with limited proven potential to differentiate into sensory cells [44]. Terminally differentiated cells such as dorsal root ganglion (DRG) offer an alternative to iPSC-induced neurons for in vitro studies and have been used in the detection of noxious stimuli and pain mechanisms [46]. Mouse DRGs are relatively easy to dissect and culture; however, it is important to note human DRGs display different responses to analgesics compared to rodent DRGs [44] as this remains applicable to a wide variety of tissues. Indeed, animal cells will always produce a biassed response to drugs that are designed to target human-specific diseases. Thus, the optimisation and validation of human cell isolation for skeletal disease and pain simulation remain of utmost importance.
Biomaterials
The mechanical properties of scaffolds at a macroscopic and microscopic scale play a crucial role in regulating cell behaviour [47] and, typically, vary dependently on the biomaterial system of choice. Hydrogels display exceptional biocompatibility, hydrophilicity, degradability and oxygen/nutrient permeability together with structural stability [48]. A number of studies have shown that mechanical properties of biomaterials influence and guide HBMSC differentiation along the different stromal lineages depending on the particular Young’s moduli (or elastic modulus, E) of the material [49]. Osteoconductivity and osteoinductivity are additional properties given a bone biomaterial platform will ideally promote bone formation and guide autologous and skeletal-residing stem cells to differentiate into bone cells [50]. In contrast, biomaterials with a low elastic modulus are required for the regeneration of neural tissues. To guide the repair of neural tissue, the biomaterial system needs to facilitate and foster neural network formation, mimicking the same function of the neural extracellular matrix (ECM) [51]. Thus, differences in Young’s moduli impact on cell fate: a biomaterial with E < 1 kPa facilitates brain tissue development with iPSCs induced to differentiate into neurons, while E in the range of 90–230 kPa may be suitable for the spinal cord tissue development [48, 52].
TE 3D models and technologies
3D in vitro models offer novel tools for the evaluation of drug safety and efficacy. Two-dimensional culture of different cell types has allowed the generation of models able to explore diseases from a multi-tissue perspective. Nevertheless, the inability to fully recapitulate the complexity of the disease micro-environment and architectural functionality has proved detrimental and limiting. 3D models have thus in recent years attracted much interest as reliable and reproducible approaches for the fabrication of new biomimetic models for drug screening.
3D bioprinting
Additive manufacturing (AM) technologies support the layering of materials to print objects from three-dimensional model data, layer upon layer, facilitating the manufacture of structures with a predefined geometry and size [53]. 3D bioprinting is a widely used AM technology to produce functional cell-laden scaffolds using polymeric bio-inks [53, 54]. Generally, a 3D-bioprinted scaffold for in vitro culture needs to present specific properties such as biocompatibility, controlled kinetics of biodegradability and comparable structural/mechanical properties to the native tissue to permit adequate oxygenation, mass transfer, nutrient exchange and vascularisation [48, 53, 54]. Thus, vascularisation, while still a major challenge, is essential to recreate the natural metabolic functions of tissues, such as nutrient transportation and waste removal [55]. Harnessing 3D bioprinting approaches, it is possible to deposit cells, layer by layer, to produce three-dimensional complex cellular structures using computer-aided design (CAD) [53, 56]. Moreover, 3D bioprinting technologies offer the advantage of building complex and hierarchical microstructures with high resolution and reproducibility, suitable for cell survival, proliferation and maturation (Fig. 2a) [53, 57].
Tissue engineering platforms for in vitro disease modelling. Modelling platforms for the recapitulation of bone-neuro pathological conditions include (a) 3D bioprinting, (b) microfluidics and (c) organoids. These systems hold great potential in mimicking the disease conditions present in bone and neural tissue. 3D bioprinting technologies offer the ability to pattern functional architectures and design as well as the ability to 3D print scalable and complex tissues. Microfluidics lack the above-listed abilities, but can precisely control small volumes of liquid required to create compartmentalised micro-environments for the development of in vitro models. Organoids, in contrast, offer biomaterial-free approaches with application of self-assembling properties of different types of cells to build and recapitulate physiologically functional tissue substitutes/models
Microfluidics
TE has benefited from advancements in microfluidic-based technologies, in particular from the development of organ-on-a-chip platforms [58,59,60]. Such devices are designed to house 3D multi-cell culture within interconnected channels and/or chambers, where the transport of nutrients and biochemical cues is precisely controlled by laminar flows. Specifically, the geometry of the device combined with the tuning of fluidic parameters allows for fine spatiotemporal patterning of chemical, physical, and mechanical properties, resulting in the reproduction of a biomimetic tissue microenvironment. The ability to integrate micro-actuators, and the application of external stimuli (e.g., mechanical [61], acoustic [62] and electrical [63]), have enhanced the ability to recreate pathophysiological conditions, providing microfluidic devices as a superior alternative to conventional in vitro models.
High-throughput analysis and enhanced reproducibility as well as cost reduction are additional advantages of microfluidic platforms [64]. However, the use of microfluidic devices for cell culture is still not widespread due to the challenging fabrication procedures and the expensive instrumentation typically required. Moreover, microfluidic platforms suffer from poor scalability and can only partially replicate, at the micro-scale, the pathophysiological damage at the tissue macro-scale level relevant for clinical applications (Fig. 2b) [48].
Organoids
Organoids are three-dimensional in vitro cell models that consist of clusters of cells, such as iPSCs, ESCs or adult stem cells (ASC) that self-organise spatially and differentiate into functional mini-organs using a scaffold-free approach. The mature structures are able to recapitulate, to some degree, the cellular composition, architecture and functions of a native organ [48, 65,66,67], thus used for in vitro disease modelling (Fig. 2c). Organoids are particularly useful for the identification and testing of new therapeutic treatments given organoids can adapt to any in vitro genome modification or gene therapy [66]. To date, reproducibility has proved a major issue, due to significant intra- and inter-batch differences in terms of size and cell organisation and composition. Furthermore, organoids lack vascular perfusion, mechanical signals, long-term stability and circulating immune cells, essential for physiological function [66, 68].
Towards development of bone pain models
Currently, animal models are the most widely used platform for drug discovery and screening, despite significant issues associated with reproducibility, availability and ethical considerations [9]. An alternative is provided by ex vivo models: explants of human or animal tissue that contain the cellular and extra-cellular composition needed to replicate the in vivo conditions [9] and can be applied for the screening of new drugs for safety and efficacy [69]. The following section details the most effective models for simulating bone and neuro tissues, as well as their close interaction within a pathological state.
Bone models
Bone is a complex dynamic tissue, capable of regeneration following damage. However, the majority of pathologies associated with skeletal tissue remain unresolved with TE addressing some of the challenges through development of viable models for the recapitulation and study of such skeletal pathologies.
In vivo
In vivo bone models allow the examination of pathologies with a comparable degree of cell, biophysical and biochemical signalling observed in the human body [70, 71]. The selection of a specific in vivo skeletal model is not without challenges from consideration of the pathophysiology of the skeletal disease, species differences, timelines and variations in bone properties (density, hardness, architecture, porosity and bone turnover) across species [70] [72]. Small-animal models (rodent) for bone-related diseases (Fig. 3a, i) are widely used given the associated low cost and their accelerated metabolism [73]. Rodents and small animals are generally indicated for the study of bone metabolism and regeneration related to age, fractures, osteoporosis and osteoarthritis [74,75,76]. Large-animal pre-clinical models (Fig. 3a, ii), such as sheep [77], dogs [78] and pigs [79], are typically used to study size-related processes or metabolic characteristics, which can be comparable to the human pathophysiology, as in the case of long-term diseases, biomechanics or bone healing efficiency. The domestic sheep has found application as a model for orthopaedics and traumatology research given their similar weight to the human body [70]. In terms of organic, inorganic volatile fraction, water and ash content, studies indicate canine models are able to recapitulate the human physiological state [71, 80], and thus have found application in examination of joint disorders, especially osteoarthritis [70]. Nevertheless, the use of preclinical animal models is currently widely accepted as replaceable, with extensive effort in providing new alternatives to reduce and ultimately replace animal models.

© 2021. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). (a, ii) The similarities between the anatomical and biomechanical characteristics of the sheep spine and the human spine allow for the study of chronic cervical spinal cord compression. A digital remote-controlled intervertebral compression device (IVCD) allowed the application of a progressive compression for 1, 5, 10, and 20 weeks, simulating a human cervical spinal cord compression [77]. This is an open-access article distributed under the terms of the Creative Commons CC BY license. (b) In vitro models. (b, i) Harnessing nanoclay-based material, a 3D mineralising micro-environment for HBMSCs to proliferate and differentiate can studies in vitro, capable of developing a functional bone model in 21 days. ALP and Von Kossa staining for HBMSC-laden 3D-bioprinted scaffolds cultured in basal and osteogenic conditioned media. Adapted from [81]; Copyright© 2020. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). (b, ii) The ability of nanoclay to foster BMSC differentiation towards bone and the culturing in osteogenic media conditioning demonstrate the ability of Laponite-based constructs to generate a bone 3D model in a shorter time (D1) with significantly increased ALP deposition [82]. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). (b, iii) A high shape fidelity of nanoclay-based bioprinted construct can be obtained by temporarily increasing the ink viscosity with the use of methylcellulose (i–ii), ensuring a stable cell viability over the time (iii). Furthermore, the nanoclay content can be exploited for a controlled release of biological active agents able to allow the development of bone tissue. [83]
In vivo and in vitro bone models. (a) In vivo models. (a, i) A novel osteoporotic mouse model developed using an innovative movable and non-invasive unloading device (ULD). Micro-CT scan images (yellow highlighted analysis region) of the trabecular and cortical structures of mice femurs from control, tail suspension and 3D-ULD groups; adapted from [73] Copyright
In vitro
In vitro models using stem cells can generate specific tissue platforms with the appropriate conditioning, and in vitro stimulation with HBMSCs has been widely used as a building block for 3D complex constructs that aim to explore bone repair and regeneration.
3D bioprinting
The use 3D bioprinting of bioactive and biodegradable materials in combination with stem cells has garnered significant interest for the fabrication of functional skeletal models. Natural and synthetic biomaterials have been extensively explored for the printing and modelling of diseased bone tissue. However, as detailed above, these approaches still cannot recapitulate the complex skeletal micro-environment [84, 85]. An alternative is provided by the use of a combination of polymeric biomaterials with calcium phosphate cements [86], hydroxyapatite [87], β tri-calcium phosphate (β-TCP) [88] or bio-glasses [89], to produce composite scaffolds improving the binding interaction and mechanical properties of the material during the 3D printing process. Such composites offer, potentially, an initial mineralised osteoinductive, as well as osteoconductive, support for encapsulated skeletal cells [30, 54, 85, 90]. Recent studies have examined the use of bioinks with nanocomposites such as the synthetic nanosilicate clay (Laponite® (LAP)) [83]. LAP displays a capacity to promote the differentiation of HBMSCs in osteogenic cells both in vitro (Fig. 3b, i) and in vivo [81] and can be used in combination with polymeric materials such as gellan gum (GG) [82] (Fig. 3b, ii), gelatin methacryloyl (GelMA) [91] and alginate-methylcellulose [83, 92] (Fig. 3a, iii) to produce functional bioinks to model bone tissue. While a range of material approaches exists for bone scaffold manufacture, to date, the recapitulation of the natural bone micro-environment has proved elusive.
Microfluidic
Osteoclasts and osteocytes have been established in co-culture in microfluidic devices in the presence of fluid flow stimulation to explore the mechanotransduction and intercellular signalling pathways involved in osteoporosis [93]. Recently, Ma and colleagues have developed a model of rheumatoid arthritis (RAs) in a microfluidic chip to investigate fibroblast-like synoviocyte-mediated bone erosion. When cultured in the RA-mimicking chip, human synovium cells were found to exhibit higher migration potential, associated with enhanced expression of cadherin-11, towards the co-culture of RANKL-stimulated osteoclastic RAW264.7 and BMSCs, confirming the modelling ability of the microfluidic platform [94]. The microfluidic approach has been found particularly suitable for the study of bone tumour invasion [95], metastatic intravasation [96] and extravasation [97]. A novel humanised organ-on-a-chip model of the breast cancer bone metastatic niche has been recently proposed [98]. The microfluidic device housed three interconnected chambers including neuro, breast and bone cells, respectively. The synergistic interplay between neurons and osteoclasts towards bone tropic breast cancer resulted in increased levels of pro-inflammatory cytokine cells.
Despite the great versatility of the microfluidic approach, a crucial limitation remains the poor level of biomaterial mimicry. Thus, the absence of rigid matrices in microfluidic chips limiting the reproduction of the complex calcified ECM found in vivo is limiting bone TE research.
Organoids
Bone-cartilage organoids (BCORG), representing “mini-joint” models, have been obtained by co-culturing cells isolated from paediatric rib tissues containing both bone and cartilage in osteochondral medium [99]. Despite limitations, in terms of ultimate maturation, BCORG may provide a tool for osteoarthritis disease modelling and drug testing. Abraham and colleagues, reported that joint organoids were partially responsive to treatment with adenosine A2A receptor agonists, previously used in murine models of osteoarthritis to reduce skeletal tissue damage [99]. Organoids have emerged as key players in modelling pathological states caused by an imbalance of bone tissue resorption and deposition processes, such as osteoporosis and loss of bone mass due to reduced mechanical stimulation during space exploration missions. For instance, Iordachescu and co-workers [100] cultured bone organoids, generated by seeding primary osteoblasts and osteoclasts onto femoral head micro-trabeculae, in a bioreactor simulating microgravity conditions. Interestingly, compared to static controls, the simulated microgravity organoids showed altered morphology of resorption sites and reduced bone mass. Thus, organoids offer an attractive approach to functional bone tissue modelling given the ease of fabrication and relatively simple, yet long, maintenance and maturation potential. However, inconsistencies in scale up and reproduction of identical bone organoids remain a significant challenge.
Neuro models
The engineering of a functional nerve tissue in vivo or in vitro remains an unmet research challenge. Two- and three-dimensional cell cultures have offered valuable information on the study of nervous system diseases but remain limited in their ability to model human neural development [101]. To date, in vivo animal-based neuro models remain the approach of choice [102] with yet limited success in recapitulating the neural micro-environment in vitro.
In vivo
In vivo animal-based neuro models are extensively used for the study of physiological and behavioural consequences of pain-associated mechanisms [102]. Currently, animal nerve injury models offer the most promising approach to study the development of pain. Surgical nerve lesions provide information on how neuropathic pain is induced in animals, which share numerous similarities to human pain [102]. A seminal model was proposed by Bennett and Xie [103], using a chronic constriction injury of the sciatic nerve in rodents closely mimicking human neuropathic distress resulting from a trauma of the peripheral as well as mechanical allodynia [102]. Another in vivo model with lesion of the partial sciatic nerve has been developed inducing neuropathic pain and mechanical allodynia [104]. Nerve injury was created by tying the third dorsal nerve at the middle of the sciatic nerve at the level of the upper thigh of the rodent [102]. Recently, the possibility of alleviating chronic pain experienced by inhibiting glial fibrillary acidic protein (GFAP)-positive glial cells has been investigated [105] (Fig. 4a, i). Results indicate the neuropathic pain behaviour induced by spared nerve injury (SNI) significantly decreased in mice with downregulated nuclear factor κB (NFκB). However, anatomical and physiological differences between rodent and the complex human nervous system given the marked differences in morphology, number of nerve cells, laminar distribution and gene expression limit many studies [101]. Human models would represent the optimal platform to study neuropathic pain-related pathological changes; however, limitations in the availability of postmortem samples, biopsies, neuroimaging and neuropharmacological treatments remain [101]. Biopsies can be subject to a severe response involving reactive cells, proliferation, and progressive neurodegeneration, reducing their suitability to accurately model a wide range of diseases [101, 106].

Copyright © 2022. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). (b) In vitro models. (b, i) 3D-bioprinted brain-like cortical tissue with 3D compartmentalised modular and concentrical architectures. The use of a silk-fibroin/ECM scaffold with a rudimentary structure provided relevant features useful for brain neural network development. In addition, the developed brain tissue showed electrophysiological functions in response to traumatic brain injury (TBI): a change in baseline signal is shown after a weigh-drop impact, with a consequent injury-triggered Glu release that mimics observations in vivo. This modular 3D brain-like tissue is capable of real-time nondestructive assessments offering the possibility to model brain disorders such as TBI. Adapted from [107]. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). (b, ii) Printing neural tissues using a lipid-bilayer-supported droplet bioprinting (i) allows for the development of a functional stimulus-responsive neural network in some weeks (ii). Neural tissue was obtained by 3D printing aqueous droplets conjoined by lipid bilayers, with a spatial pattern which not only gives a control of cell self-organisation, but also provides a neuronal network which can be obtained only after months of organoid cultures. Furthermore, diseases could also be modelled by incorporating reprogrammed patient cells with specific genetic mutations. Adapted from [108]. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). (b, iii) 3D-printed nerve system on a chip. 3D-printed device comprising silicone microchannel for axonal guidance. Superior cervical ganglia (SCG) neurons with green-labelled tau protein aligned within the microchannel. Triple channels with self-assembled network of Schwann cells stained with PRV brainbow. Close-up images of above-mentioned detailed micrographs. Adapted from [109]. This article is licensed under a Creative Commons Attribution 3.0 Unported Licence
In vivo and in vitro neuro models. (a) In vivo models. (a, i) A model for the study of functional treatment for the crossing of the blood–brain barrier (BBB) to inhibit central reporter gene expression and study glial signalling to alleviate chronic pain. GFP-positive reported gene expression (green) is extensively observed in the spinal cord tissue (left panels), while not localised in the sciatic nerve (right panels) at different time points. Glial fibrillary acidic protein (GFAP)–positive glial cells and eGFP expression were found to co-localise in specific regions—white arrows. Adapted from [105].
In vitro
Alternatively, in vitro models comprising neuronal cells are currently in development to aid the reduction, replacement and refinement (3Rs) of in vivo animal models for the simulation of pathophysiological pain mechanisms [44]. 3D neuro cultures have been shown to support cell differentiation, increased neuritis outgrowth, and myelination [65, 110], harnessing 3D bioprinting, microfluidics and organoid systems technologies.
3D bioprinting
3D-bioprinted in vitro neuro models, as detailed for bone tissue models, requires the choice of a suitable biomaterial able to support cell survival, proliferation, and functionality of the encapsulated cells. Functional biomaterials used for neuro tissue 3D bioprinting are typically natural polymers such as alginate [111, 112], collagen type I, silk fibroin [107], matrigel [113], chitosan [114], hyaluronic acid (HA) [113, 115], methylcellulose [116] and blends such as alginate/carboxymethyl-chitosan/agarose [114] as well as synthetic polymers poly(ethylene–glycol) (PEG) [117, 118] and functionalised materials, such as GelMA [119]. To improve cell viability, functionality and cell adhesion, these biomaterials are often functionalised with peptides such as -RGD, -IKVAV and -YIGSR, or proteins such as laminin and fibronectin [48]. Lozano and colleagues [51] proposed the printing of a brain-like layered structure model using an RGD-gellan gum-based bioink (RGD-GG) encapsulating primary cortical neural cells. The authors reported a porous model structure that supported cell proliferation and network formation [48, 51, 120]. A novel neural mini-tissue construct has been fabricated using human cortical neuronal cells (NSCs) embedded in a polysaccharide hydrogel comprising carboxymethyl-chitosan, agarose and alginate [86]. The system contained a homogeneous cell distribution that displayed good cell viability as well as in situ differentiation of the NSCs [114, 120]. As detailed by studies reported above, 3D bioprinting technologies are appealing for neural tissue model fabrication and have been used to fabricate complex models (Fig. 4b, i) for the study of 3D neurite outgrowth and elongation [121], human cortex design and engineering [108] (Fig. 4b, ii) as well as glioblastoma modelling [122]. However, 3D-printed models are far from recapitulating the complex neural micro-environment yet failing to support axonal growth and functional physiological activity.
Microfluidics
Microfluidic devices have been employed for in vitro studies in the development of multi tissue/interface structures to examine cortical brain structures providing a platform for drug development [123] with a focus on fabrication of microfluidic devices able to provide adequate solutions for the modelling of complex neural micro-environment such as the blood–brain barrier (BBB) facilitating screening of BBB-targeting drugs in neurological diseases [48, 124]. Crucially, the parallel microgrooves commonly used in microfluidics to connect different tissue compartments offer ideal structures for neuron functionality and activity enabling enclosures for axon extension and pathfinding towards a target situated in adjacent chambers (Fig. 4b, iii) [109]. Recent studies have focused in generating models that could facilitate communication between neural cells and other tissue, for the development of models for cancer metastasis and neuromuscular junction [125, 126]. As a result, information gained from studies on CNS axonal injury and regeneration on-a-chip [127] indicate microfluidics provides an unparalleled tool for human pain modelling, delivering unique in vitro platforms that enable the separation of axons from cell bodies and their localised treatment (e.g., drug testing and injury) through chamber compartmentalisation. In support of such an approach, Vysokov and colleagues reported a microfluidic system to model the pain synapse and investigate the role of voltage gated sodium channels (NaVs) in synaptic transmission [128]. Nevertheless, in vitro pain modelling remains poorly explored. It will undoubtedly fall on researchers to harness microfluidics in combination with other biotechnological tools such as optogenetic and multi electrode array (MEA) devices to advance neuro-pathophysiology research.
Organoids
Organoid models offer an attractive approach for the study of neural development and the testing of new treatments for neurological diseases [67]. The seminal work of Lancaster and co-workers provided a method to produce 3D cerebral human iPSCs-derived organoids [129] with the potential to apply organoids to model aspects of human neurodevelopment and neurological diseases. Subsequent studies developed protocols to reproduce organoids derived from specific brain regions. Thus, recently, the differentiation of human pluripotent stem cells into a large multicellular organoid-like structure has been reported, producing organoids with distinct layers of neuronal cells expressing genetic markers of the human midbrain [130].
In the nervous system, pain stimuli trigger sensory neurons, which in turn transmit the signal to neurons in the dorsal horn of the spinal cord. An organoid model to recapitulate in vitro such an initial step of the nociceptive circuitry has been recently reported [131]. Ao and colleagues used an organoid-on-chip device made of a 3D-printed holder with a porous polycarbonate membrane, in which a dorsal spinal cord organoid with both sensory neurons and dorsal spinal cord interneurons was maintained at the air–liquid interface. The ability to produce nociceptive responses was validated upon stimulation with known pain-evoking substances, including mustard oil, capsaicin and velvet ant venom as well as temperature increase to trigger the thermosensitive nociceptive pathway. Thus, the effect of pain relievers (e.g., cannabinoids) could be observed, confirming a platform for functional pain modelling in vitro [131].
3D models for mimicking bone pain
Bone pain models have preferentially involved the use of in vivo platforms for the monitoring of disease progression and therapeutic efficacy analysis for bone pain treatment and despite obvious limitations arising from species differences, offer a translational route to clinical application (Table 1). Osteoporotic, osteoarthritic and cancer-related bone fractures are examined in pre-clinical models for the study of bone pain with a focus on the generation of functional in vivo or in vitro models to recapitulate the mechanism underlying the disease conditions, to develop effective treatment strategies [132].
Fracture models
Bone fracture in rodent models is performed using tailored protocols where the selected bone segment is broken using a defined force [132, 144, 145]. The most common fracture model, developed by Bonnarens and Einhorn [146] provides a closed mid-shaft femoral fracture in a rat in which the femur is stabilised with an intramedullary Steinman pin or Kirschner (K) wire and a diaphyseal fracture created using the Einhorn drop-weight apparatus [144, 146] The model has proved highly reproducible, although is not suitable for the analysis of healing in delayed unions given the high healing rate typically observed in rodents. An alternative method used a femoral osteotomy, with the periosteum removed around the fracture site, creating an open fracture model [144, 147]. This technique allows for a significant impairment of bone healing and favours the observation of the healing phenomenon. Osteoarthritis and osteoporosis animal models have been engineered to fully recapitulate the skeletal discomfort and thus enable study of the underlying mechanisms and evaluation of treatment approaches for clinical translation. Osteoporotic models can be induced following ovariectomy in female mice, recreating a postmenopausal osteoporotic model with related bone fractures. However, the model is not representative of the human osteoporotic fracture as the mineral density of mice bone is lower than that found in human bone [145]. Osteoarthritic pain induction, surgical interruption of the anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL) in mouse have all been shown to be efficacious in the reproduction of osteoarthritic pain models. Such techniques allow the development of the pathology in an animal model with comparable developmental features observed in the human condition [132, 148].
In vivo bone pain models have been extensively used for decades to study skeletal pain. However, these models are involving severe discomfort for the animals, with disruptive consequences for prolonged studies and results. However, in vitro models for bone pain are missing. To date, in vitro fracture models capable of replicating skeletal-specific pain mechanisms have not been identified. Morgan and co-workers [143] reported the study of the activation of transient receptor potential cation channel subfamily V member 1 (TRPV1) expressed in bone afferent neurons (Fig. 5a) although could not recapitulate the complexity of bone fracture following the exclusion of crucial components such as the vascular network in damaged skeletal tissue.

Copyright 2019. (b) Bone cancer pain model. Destruction of bone tissue (b, i) confirmed by X-ray imaging and histological analysis, confirming the presence of tumour cells within the marrow cavity. (b, ii) Paw mechanical withdrawal threshold (PMWT) and paw withdrawal thermal latency (PWTL) with cold and hot surfaces investigated in a bone cancer rat model to evaluate the pain response in vivo. Relative expression of mRNA and Transient receptor potential ankyrin 1 (TRPA1) was enhanced in the bone cancer pain animal model. Targeting via TRPA1 antisense oligodeoxynucleotide (AS-ODN) relieved PMWT and PWTL. Adapted from [136]. Copyright© 2021 under the terms of the Creative Commons Attribution License (CC BY). (c) Human spinal organoids in a chip (c, i) positive staining for CB1 expression, for sensory, inhibitory, and excitatory neurons (CGRP + , GAD1 + and vGlut1 + , respectively). (c, ii) Response to capsaicin and electrical stimulation of spinal organoids towards nociceptive modulation, with enhanced mean firing rate and average burst frequency for BDNF and capsaicin-stimulated group. Adapted with permission from [131]. Copyright 2022 American Chemical Society. (d) Human sensorimotor organoids based on (d, i) TUJ1 + neurons and sarcomeric α-actinin (SAA) + myocytes can include both cell types (d-ii) and observed to be functional over 4 weeks. Adapted from [149]. Creative Commons CC BY
Bone pain models. (a) Bone pain in vitro model—TRPV1 expression. (a, i) L3 DRG section highlighting NF200 + neurons, TRPV1 + bone afferent neurons. (a, ii) The stimulation with 0.1 μM and 1 μM capsaicin increased the discharge frequency of both small (C fibre) and medium (Aδ fibre) amplitude. Adapted with permission from [143].
Cancer bone pain models
Cancer bone pain models have been extensively produced in vivo. The majority of the models involve the use of rodents in which bone metastases have been induced by localised cancerous cell injection. One of the first mouse models was proposed by Arguello and collaborators [150] in which intracardiac injections of myeloma cells were given to study the mechanism of bone localisation of blood-borne cancer cells. However, the pathological state was so severe the model precluded analysis of the pain experienced by the animal [132]. Schwei and colleagues [151] proposed an alternative model using a femur intramedullary injection of tumour cells to obtain a reproducible and localised tumour mass [28, 132]. The model was more effective in simulating the situation of cancer bone pain, recreating spontaneous pain and mechanical allodynia, enabling a comparison with the untreated limb [132]. Other studies have focused on ex vivo or in vitro reproduction of bone cancer using bone tissue explants or through biofabrication technologies. Nordstrand and colleagues [152] developed a co-culture system with physically separated murine calvaria explants and prostate cancer cells to study bone remodelling activity in the presence of cancer cells without being in direct contact with bone tissue [152, 153]. Established cancer bone pain models have found significant traction for the screening of novel drugs and therapies often targeting specific proteins and disrupting pathways involved in pain mechanisms (Fig. 5b). Approaches include the Transient receptor potential ankyrin 1 (TRPA1), recently targeted by Liu and co-workers [136] using TRPA1 antagonist and antisense oligodeoxynucleotide. Nevertheless, poor translation to human pathophysiology remains impairing pre-clinical drug screening and, crucially, validation.
In vitro bone pain models
There remains an evident absence of in vitro bone pain models with only a few reports detailing either fracture healing approaches without the involvement of neural fibres, or the exclusive use of functional nociceptive models. A recent approach has harnessed spinal organoids, 3D printing and microfluidic technologies to demonstrate the stable generation of an in vitro pain model (Fig. 5c) [131]. However, the use of hESCs, the inability of include multiple cell types (e.g., BMSCs, myocytes) and the lack of further investigation are still limitations in the translational potential of such a drug screen platform. In essence, incorporation of multiple cell types, recapitulating a functional tissue interface, is a prerequisite to study bone pain mechanisms. This was illustrated by Pereira and colleagues [149] with the generation of a sensorimotor organoid model comprising both neurons (TUJ1 +) and myocytes (SAA +) derived from iPSCs culture (Fig. 5d). The presence of sensory neurons and muscle cells offers a stable 3D platform approach, in vitro, for the investigation of pain. An obvious next step would be the inclusion of skeletal cell populations to provide additional perspective in the musculoskeletal pain paradigm.
Summary, challenges and future perspectives
Bone pain discomfort arising as a consequence of trauma or skeletal diseases affects patient quality of life. Despite decades of research, the underlying mechanisms of pain affecting diseased or damaged bone tissue is to be fully elucidated. There remains an unmet need to generate functional models able to recapitulate the complex temporal cascade of biological events involved in bone pain development that could aid to new therapeutic modalities for clinical pain management and drug screen research.
To date, animals have been routinely used as the perceived most reliable model for the study of bone pain mechanisms and therapy development. However, (i) ethical issues, (ii) species variability and (iii) intrinsic differences to human physiology limit routinely the translation of novel drug treatments. Animal in vivo models provide a rapid approach to gather preliminary results on the safety and efficacy of drugs. However, the conflicting pathophysiological differences between human and animal are now making the in vivo model obsolete. Today, it is clear that there is a need to resort to reliable in vitro bone pain models to improve current therapies. To date, commercially available, functional, reproducible and reliable in vitro models capable of resembling painful skeletal pathologies are still not available.
TE offers a unique way to address some of these issues, providing specific tools and models that harness relevant cell types, scaffolds together with biofabrication/microfluidic and biotechnology strategies to create new approaches to model human bone pain. In vitro models hold significant potential and new vistas in research in simulating both pathological and bone pain conditions, helping to reduce, refine and, ultimately, replace animal models for the testing of bone pain treatments. Current research in bone pain modelling seeks to reproduce, in vivo, the pathological state and to subsequently induce pain with a platform comparable to the human bone lesion. A central and rather obvious limitation in the field remains the inability to recapitulate physiological bone innervation as a consequence of often oversimplified models, lacking the essential requisite of different cell types necessary to recapitulate the bone-related pain cascade. Of course, these are not trivial issues, and the co-culture of bone and nerve cells is challenging. The possibility to engineer a micro-environment that can support chemical stimuli for cell survival, proliferation and signalling of pain, as well as mechanical stimuli due to external forces and matrix constraints, is hypothesised and yet remains, by us and others in the field, to be demonstrated. Therein lies the challenge and need for collaborative synthesis of methodologies across traditional boundaries of skeletal, nerve and indeed pain research.
The future is bright, interdisciplinary research harnessing life sciences, tissue engineers and clinicians and the raft of new methodologies for imaging to “omics” and biotechnology platforms auger well in the development of a third way, new avenues in the exploration of pain relief therapies, clearly needed for an ageing demographic. Thus, new in vitro models are anticipated, harnessing TE approaches that will lead to the prevention of bone pain with widespread benefit across the global healthcare systems and, ultimately, patients.
References
Alves CJ, Neto E, Sousa DM, et al. Fracture pain-traveling unknown pathways. Bone. 2016;85:107–14. https://doi.org/10.1016/j.bone.2016.01.026.
Lortet-Tieulent J, Georges D, Bray F, Vaccarella S. Profiling global cancer incidence and mortality by socioeconomic development. Int J Cancer. 2020;147:3029–36. https://doi.org/10.1002/ijc.33114.
Frost CØ, Hansen RR, Heegaard AM. Bone pain: current and future treatments. Curr Opin Pharmacol. 2016;28:31–7. https://doi.org/10.1016/j.coph.2016.02.007.
Ripamonti C, Fulfaro F. Malignant bone pain: pathophysiology and treatments. Curr Rev Pain. 2000;4:187–96. https://doi.org/10.1007/s11916-000-0078-3.
Zajączkowska R, Kocot-Kępska M, Leppert W, Wordliczek J. Bone pain in cancer patients: mechanisms and current treatment. Int J Mol Sci. 2019;20:65–75. https://doi.org/10.3390/ijms20236047.
Haegerstam GAT. Pathophysiology of bone pain: a review. Acta Orthop Scand. 2001;72:308–17. https://doi.org/10.1080/00016470152846682.
Falk S, Dickenson AH. Pain and nociception: mechanisms of cancer-induced bone pain. J Clin Oncol. 2014;32:1647–54. https://doi.org/10.1200/JCO.2013.51.7219.
Court-Brown CM, Wood AM, Aitken S. The epidemiology of acute sports-related fractures in adults. Injury. 2008;39:1365–72. https://doi.org/10.1016/j.injury.2008.02.004.
Ehnert S, Rinderknecht H, Aspera-Werz RH, et al. Use of in vitro bone models to screen for altered bone metabolism, osteopathies, and fracture healing: challenges of complex models. Arch Toxicol. 2020;94:3937–58. https://doi.org/10.1007/s00204-020-02906-z.
Daly RM. Exercise and nutritional approaches to prevent frail bones, falls and fractures: an update. Climacteric. 2017;20:119–24. https://doi.org/10.1080/13697137.2017.1286890.
Drenjančević I, Davidović Cvetko E. Influence of physical activity to bone metabolism. Med Glas Off Publ Med Assoc Zenica-Doboj Canton Bosnia Herzegovina. 2013;10:12–9.
Borg SA, Buckley H, Owen R, et al. Early life vitamin D depletion alters the postnatal response to skeletal loading in growing and mature bone. PLoS One. 2018;13:1–17. https://doi.org/10.1371/journal.pone.0190675.
Mitchell SAT, Majuta LA, Mantyh PW. New insights in understanding and treating bone fracture pain. Curr Osteoporos Rep. 2018;16:325–32. https://doi.org/10.1007/s11914-018-0446-8.
Cecchini MG, Wetterwald A, van der Pluijm G, Thalmann GN. Molecular and biological mechanisms of bone metastasis. EAU Updat Ser. 2005;3:214–26. https://doi.org/10.1016/j.euus.2005.09.006.
Park SH, Eber MR, Widner DB, Shiozawa Y. Role of the bone microenvironment in the development of painful complications of skeletal metastases. Cancers (Basel). 2018;10:. https://doi.org/10.3390/cancers10050141
Yoneda T, Hiasa M, Nagata Y, et al. Acidic microenvironment and bone pain in cancer-colonized bone. Bonekey Rep. 2015;4:1–9. https://doi.org/10.1038/bonekey.2015.58.
Mach DB, Rogers SD, Sabino MC, et al. Origins of skeletal pain: sensory and sympathetic innervation of the mouse femur. Neuroscience. 2002;113:155–66. https://doi.org/10.1016/S0306-4522(02)00165-3.
Migliorini F, Maffulli N, Trivellas A, et al. Bone metastases: a comprehensive review of the literature. Mol Biol Rep. 2020;47:6337–45. https://doi.org/10.1007/s11033-020-05684-0.
Macedo F, Ladeira K, Pinho F, et al. Bone metastases: an overview. Oncol Rev. 2017;11:. https://doi.org/10.4081/oncol.2017.321
Ural A. Advanced modeling methods—applications to bone fracture mechanics. Curr Osteoporos Rep. 2020;18:568–76. https://doi.org/10.1007/s11914-020-00615-1.
George WT, Vashishth D. Susceptibility of aging human bone to mixed-mode fracture increases bone fragility. Bone. 2006;38:105–11. https://doi.org/10.1016/j.bone.2005.08.002.
Danielle X. Morales, Sara E. Grineski and TWC. 乳鼠心肌提取 HHS Public Access. Physiol Behav. 2016;176:139–148. https://doi.org/10.1097/j.pain.0000000000001838.Peripheral
Messlinger K. What is nociceptor. Schmerz. 1997;11:353–35366. https://doi.org/10.1007/s004829700058.
Schmidt-Bleek K, Schell H, Kolar P, et al. Cellular composition of the initial fracture hematoma compared to a muscle hematoma: A study in sheep. J Orthop Res. 2009;27:1147–51. https://doi.org/10.1002/jor.20901.
Sawa-Wejksza K, Kandefer-Szerszeń M. Tumor-associated macrophages as target for antitumor therapy. Arch Immunol Ther Exp (Warsz). 2018;66:97–111. https://doi.org/10.1007/s00005-017-0480-8.
Seike W. Electrophysiological and histological studies on the sensibility of the bone marrow nerve terminal. Yonago Acta Med. 1976;20:192–211.
Chapurlat RD, Gensburger D, Jimenez-Andrade JM, et al. Pathophysiology and medical treatment of pain in fibrous dysplasia of bone. Orphanet J Rare Dis. 2012;7:1–9. https://doi.org/10.1186/1750-1172-7-S1-S3.
Wakabayashi H, Hiraga T, Yoneda T. Mechanism of cancer-induced bone pain. Clin Calcium. 2006;16:73–79. CliCa0604605611
Coleman R. The role of zoledronic acid in cancer: clinical studies in the treatment and prevention of bone metastases. Semin Oncol. 2001;28:11–6. https://doi.org/10.1016/s0093-7754(01)90260-x.
Costa L, Major PP. Effect of bisphosphonates on pain and quality of life in patients with bone metastases. Nat Clin Pract Oncol. 2009;6:163–74. https://doi.org/10.1038/ncponc1323.
Clohisy DR, Mantyh PW. Bone cancer pain and the role of RANKL/OPG. J Musculoskelet Neuronal Interact. 2004;4:293–300.
Lipton A, Jun S. RANKL inhibition in the treatment of bone metastases. Curr Opin Support Palliat Care. 2008;2:197–203. https://doi.org/10.1097/SPC.0b013e32830baac2.
Mantyh PW. Mechanisms that drive bone pain across the lifespan. Br J Clin Pharmacol. 2019;85:1103–13. https://doi.org/10.1111/bcp.13801.
Upadhya RK, Shenoy L, Venkateswaran R. Effect of intravenous dexmedetomidine administered as bolus or as bolus-plus-infusion on subarachnoid anesthesia with hyperbaric bupivacaine. J Anaesthesiol Clin Pharmacol. 2018;34:46–50. https://doi.org/10.4103/joacp.JOACP.
Pountos I, Georgouli T, Calori GM, Giannoudis P V. Do nonsteroidal anti-inflammatory drugs affect bone healing? A critical analysis. ScientificWorldJournal. 2012;(2012):. https://doi.org/10.1100/2012/606404
Al Farii H, Farahdel L, Frazer A, et al. The effect of NSAIDs on postfracture bone healing: a meta-analysis of randomized controlled trials. OTA Int Open Access J Orthop Trauma. 2021;4:e092. https://doi.org/10.1097/oi9.0000000000000092.
Watanabe S, Bruera E. Corticosteroids as adjuvant analgesics. J Pain Symptom Manage. 1994;9:442–5. https://doi.org/10.1016/0885-3924(94)90200-3.
Mensah-Nyagan AG, Meyer L, Schaeffer V, et al. Evidence for a key role of steroids in the modulation of pain. Psychoneuroendocrinology. 2009;34(Suppl 1):S169–77. https://doi.org/10.1016/j.psyneuen.2009.06.004.
Sengupta D, Waldman SD, Li S. From in vitro to in situ tissue engineering. Ann Biomed Eng. 2014;42:1537–45. https://doi.org/10.1007/s10439-014-1022-8.
Zhu W, Ma X, Gou M, et al. 3D printing of functional biomaterials for tissue engineering. Curr Opin Biotechnol. 2016;40:103–12. https://doi.org/10.1016/j.copbio.2016.03.014.
Alvarez-Viejo M, Menendez-Menendez Y, Blanco-Gelaz MA, et al. Quantifying mesenchymal stem cells in the mononuclear cell fraction of bone marrow samples obtained for cell therapy. Transplant Proc. 2013;45:434–9. https://doi.org/10.1016/j.transproceed.2012.05.091.
Borciani G, Montalbano G, Baldini N, et al. Co–culture systems of osteoblasts and osteoclasts: simulating in vitro bone remodeling in regenerative approaches. Acta Biomater. 2020;108:22–45. https://doi.org/10.1016/j.actbio.2020.03.043.
Bianco P, Robey PG. Stem cells in tissue engineering. Nature. 2001;414:118–21. https://doi.org/10.1038/35102181.
Chrysostomidou L, Cooper AH, Weir GA. Cellular models of pain: new technologies and their potential to progress preclinical research. Neurobiol Pain. 2021;10:100063. https://doi.org/10.1016/j.ynpai.2021.100063.
Lee G, Chambers SM, Tomishima MJ, Studer L. Derivation of neural crest cells from human pluripotent stem cells. Nat Protoc. 2010;5:688–701. https://doi.org/10.1038/nprot.2010.35.
Chakrabarti S, Hore Z, Pattison LA, et al. Sensitization of knee-innervating sensory neurons by tumor necrosis factor-α-activated fibroblast-like synoviocytes: an in vitro, coculture model of inflammatory pain. Pain. 2020;161:2129–41. https://doi.org/10.1097/j.pain.0000000000001890.
Vedadghavami A, Minooei F, Mohammadi MH, et al. Manufacturing of hydrogel biomaterials with controlled mechanical properties for tissue engineering applications. Acta Biomater. 2017;62:42–63. https://doi.org/10.1016/j.actbio.2017.07.028.
Zhuang P, Xuyang A, An J, et al. Biomaterials 3D neural tissue models : from spheroids to bioprinting. Biomaterials. 2018;154:113–33. https://doi.org/10.1016/j.biomaterials.2017.10.002.
Mao AS, Shin J-W, Mooney DJ. Effects of substrate stiffness and cell-cell contact on mesenchymal stem cell differentiation. Biomaterials. 2016;98:184–91. https://doi.org/10.1016/j.biomaterials.2016.05.004.
García-Gareta E, Coathup MJ, Blunn GW. Osteoinduction of bone grafting materials for bone repair and regeneration. Bone. 2015;81:112–21. https://doi.org/10.1016/j.bone.2015.07.007.
Lozano R, Stevens L, Thompson BC, et al. 3D printing of layered brain-like structures using peptide modified gellan gum substrates. Biomaterials. 2015;67:264–73. https://doi.org/10.1016/j.biomaterials.2015.07.022.
Saha K, Keung AJ, Irwin EF, et al. Substrate modulus directs neural stem cell behavior. Biophys J. 2008;95:4426–38. https://doi.org/10.1529/biophysj.108.132217.
Mota C, Puppi D, Chiellini F, Chiellini E. Additive manufacturing techniques for the production of tissue engineering constructs. J Tissue Eng Regen Med. 2015;9:174–90. https://doi.org/10.1002/term.1635.
Pirosa A, Gottardi R, Alexander PG, Tuan RS. Engineering in-vitro stem cell-based vascularized bone models for drug screening and predictive toxicology. Stem Cell Res Ther. 2018;9:. https://doi.org/10.1186/s13287-018-0847-8
Hann SY, Cui H, Esworthy T, et al. Dual 3D printing for vascularized bone tissue regeneration. Acta Biomater. 2021;123:263–74. https://doi.org/10.1016/j.actbio.2021.01.012.
Hutmacher DW, Sittinger M, Risbud MV. Scaffold-based tissue engineering: rationale for computer-aided design and solid free-form fabrication systems. Trends Biotechnol. 2004;22:354–62. https://doi.org/10.1016/j.tibtech.2004.05.005.
Ding S, Feng L, Wu J, et al. Bioprinting of stem cells: interplay of bioprinting process, bioinks, and stem cell properties. ACS Biomater Sci Eng. 2018;4:3108–24. https://doi.org/10.1021/acsbiomaterials.8b00399.
Whitesides GM. The origins and the future of microfluidics. Nature. 2006;442:368–73. https://doi.org/10.1038/nature05058.
Torisawa YS, Spina CS, Mammoto T, et al. Bone marrow-on-a-chip replicates hematopoietic niche physiology in vitro. Nat Methods. 2014;11:663–9. https://doi.org/10.1038/nmeth.2938.
Benam KH, Villenave R, Lucchesi C, et al. Small airway-on-a-chip enables analysis of human lung inflammation and drug responses in vitro. Nat Methods. 2016;13:151–7. https://doi.org/10.1038/nmeth.3697.
Huh D, Kim HJ, Fraser JP, et al. Microfabrication of human organs-on-chips. Nat Protoc. 2013;8:2135–57. https://doi.org/10.1038/nprot.2013.137.
Silvani G, Scognamiglio C, Caprini D, et al. Reversible cavitation-induced junctional opening in an artificial endothelial layer. Small. 2019;15:1905375. https://doi.org/10.1002/smll.201905375.
Hallfors N, Khan A, Dickey MD, Taylor AM. Integration of pre-aligned liquid metal electrodes for neural stimulation within a user-friendly microfluidic platform. Lab Chip. 2013;13:522–6. https://doi.org/10.1039/C2LC40954B.
Mehling M, Tay S. Microfluidic cell culture. Curr Opin Biotechnol. 2014;25:95–102. https://doi.org/10.1016/j.copbio.2013.10.005.
Gong L, Cao L, Shen Z, et al. Materials for neural differentiation, trans-differentiation, and modeling of neurological disease. Adv Mater. 2018;30:. https://doi.org/10.1002/adma.201705684
Nikolakopoulou P, Rauti R, Voulgaris D, et al. Recent progress in translational engineered in vitro models of the central nervous system. Brain. 2021;143:3181–213. https://doi.org/10.1093/BRAIN/AWAA268.
Corrò C, Novellasdemunt L, Li VSW. A brief history of organoids. Am J Physiol - Cell Physiol. 2020;319:C151–65. https://doi.org/10.1152/ajpcell.00120.2020.
Ingber DE. Reverse engineering human pathophysiology with organs-on-chips. Cell. 2016;164:1105–9. https://doi.org/10.1016/j.cell.2016.02.049.
Tortelli F, Pujic N, Liu Y, et al. Osteoblast and osteoclast differentiation in an in vitro three-dimensional model of bone. Tissue Eng Part A. 2009;15:2373–83. https://doi.org/10.1089/ten.tea.2008.0501.
Ribitsch I, Baptista PM, Lange-Consiglio A, et al. Large animal models in regenerative medicine and tissue engineering: to do or not to do. Front Bioeng Biotechnol. 2020;8:1–28. https://doi.org/10.3389/fbioe.2020.00972.
Nicole Gabriele Sommer, David Hahn, Begüm Okutan RM and A-MW. We are IntechOpen, the world’s leading publisher of open access books built by scientists, for scientists TOP 1 % animal models in orthopedic research : the proper animal model to answer fundamental questions on bone healing depending on pathology and implant material. 2019;0–19
Martine LC, Holzapfel BM, McGovern JA, et al. Engineering a humanized bone organ model in mice to study bone metastases. Nat Protoc. 2017;12:639–63. https://doi.org/10.1038/nprot.2017.002.
Li J, Geng J, Lin T, et al. A mouse model of disuse osteoporosis based on a movable noninvasive 3D-printed unloading device. J Orthop Transl. 2022;33:1–12. https://doi.org/10.1016/j.jot.2021.11.009.
Santos LF, Singulani MP, Stringhetta-Garcia CT, et al. Oxytocin effects on osteoblastic differentiation of bone marrow mesenchymal stem cells from adult and aging female Wistar rats. Exp Gerontol. 2018;113:58–63. https://doi.org/10.1016/j.exger.2018.09.023.
Manigrasso MB, O’Connor JP. Comparison of fracture healing among different inbred mouse strains. Calcif Tissue Int. 2008;82:465–74. https://doi.org/10.1007/s00223-008-9144-3.
Herath TDK, Saigo L, Schaller B, et al. In vivo efficacy of neutrophil-mediated bone regeneration using a rabbit calvarial defect model. Int J Mol Sci. 2021;22:. https://doi.org/10.3390/ijms222313016
Li Z, Zhai S, Liu S, et al. A sheep model of chronic cervical compressive myelopathy via an implantable wireless compression device. Eur Spine J. 2022. https://doi.org/10.1007/s00586-022-07138-6.
Sato R, Matsuura T, Akizuki T, et al. Influence of the bone graft materials used for guided bone regeneration on subsequent peri-implant inflammation: an experimental ligature-induced peri-implantitis model in Beagle dogs. Int J Implant Dent. 2022;8:. https://doi.org/10.1186/s40729-022-00403-9
Dienel K, Abu-Shahba A, Kornilov R, et al. Patient-specific bioimplants and reconstruction plates for mandibular defects: production workflow and in vivo large animal model study. Macromol Biosci. 2022;2100398:1–15. https://doi.org/10.1002/mabi.202100398.
Blanc-Sylvestre N, Bouchard P, Chaussain C, Bardet C. Pre-clinical models in implant dentistry: past, present, future. Biomedicines. 2021;9:. https://doi.org/10.3390/biomedicines9111538
Cidonio G, Glinka M, Kim Y-H, et al. Nanoclay-based 3D printed scaffolds promote vascular ingrowth ex vivo and generate bone mineral tissue in vitro and in vivo. Biofabrication. 2020;12:35010. https://doi.org/10.1088/1758-5090/ab8753.
Cidonio G, Cooke M, Glinka M, et al. Printing bone in a gel: using nanocomposite bioink to print functionalised bone scaffolds. Mater Today Bio. 2019;4:. https://doi.org/10.1016/j.mtbio.2019.100028
Ahlfeld T, Cidonio G, Kilian D, et al. Development of a clay based bioink for 3D cell printing for skeletal application. Biofabrication. 2017;9:034103. https://doi.org/10.1088/1758-5090/aa7e96.
Ma PX, Choi JW. Biodegradable polymer scaffolds with well-defined interconnected spherical pore network. Tissue Eng. 2001;7:23–33. https://doi.org/10.1089/107632701300003269.
Zhang L, Yang G, Johnson BN, Jia X. Three-dimensional (3D) printed scaffold and material selection for bone repair. Acta Biomater. 2019;84:16–33. https://doi.org/10.1016/j.actbio.2018.11.039.
Ahlfeld T, Doberenz F, Kilian D, et al. Bioprinting of mineralized constructs utilizing multichannel plotting of a self-setting calcium phosphate cement and a cell-laden bioink. Biofabrication. 2018;10:045002. https://doi.org/10.1088/1758-5090/aad36d.
Chang PC, Lin ZJ, Luo HT, et al. Degradable RGD-functionalized 3D-printed scaffold promotes osteogenesis. J Dent Res. 2021;100:1109–17. https://doi.org/10.1177/00220345211024634.
Liao H-T, Lee M-Y, Tsai W-W, et al. Osteogenesis of adipose-derived stem cells on polycaprolactone- β -tricalcium phosphate scaffold fabricated via selective laser sintering and surface coating with collagen type I. J Tissue Eng Regen Med. 2016;10:E337–53. https://doi.org/10.1002/term.1811.
Wang M, Li H, Yang Y, et al. A 3D-bioprinted scaffold with doxycycline-controlled BMP2-expressing cells for inducing bone regeneration and inhibiting bacterial infection. Bioact Mater. 2021;6:1318–29. https://doi.org/10.1016/j.bioactmat.2020.10.022.
Stanco D, Urbán P, Tirendi S, et al. 3D bioprinting for orthopaedic applications: current advances, challenges and regulatory considerations. Bioprinting (Amsterdam, Netherlands) 2020;20:None. https://doi.org/10.1016/j.bprint.2020.e00103
Cidonio G, Alcala-Orozco CR, Lim KS, et al. Osteogenic and angiogenic tissue formation in high fidelity nanocomposite Laponite-gelatin bioinks. Biofabrication. 2019;11:035027. https://doi.org/10.1088/1758-5090/ab19fd.
Cidonio G, Glinka M, Kim YH, et al. Nanoclay-based 3D printed scaffolds promote vascular ingrowth ex vivo and generate bone mineral tissue in vitro and in vivo. Biofabrication. 2020;12:035010. https://doi.org/10.1088/1758-5090/ab8753.
Middleton K, Al-Dujaili S, Mei X, et al. Microfluidic co-culture platform for investigating osteocyte-osteoclast signalling during fluid shear stress mechanostimulation. J Biomech. 2017;59:35–42. https://doi.org/10.1016/j.jbiomech.2017.05.012.
Ma H-P, Deng X, Chen D-Y, et al. A microfluidic chip-based co-culture of fibroblast-like synoviocytes with osteoblasts and osteoclasts to test bone erosion and drug evaluation. R Soc Open Sci. 2022;5:180528. https://doi.org/10.1098/rsos.180528.
Hao S, Ha L, Cheng G, et al. A spontaneous 3D bone-on-a-chip for bone metastasis study of breast cancer cells. Small. 2018;14:1702787. https://doi.org/10.1002/smll.201702787.
Bersini S, Jeon JS, Dubini G, et al. A microfluidic 3D invitro model for specificity of breast cancer metastasis to bone. Biomaterials. 2014;35:2454–61. https://doi.org/10.1016/j.biomaterials.2013.11.050.
Jj S, Simone B, Mara G, et al. Human 3D vascularized organotypic microfluidic assays to study breast cancer cell extravasation. Proc Natl Acad Sci. 2015;112:214–9. https://doi.org/10.1073/pnas.1417115112.
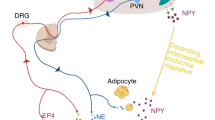
Conceição F, Sousa DM, Loessberg-Zahl J, et al. A metastasis-on-a-chip approach to explore the sympathetic modulation of breast cancer bone metastasis. Mater Today Bio. 2022;13:. https://doi.org/10.1016/j.mtbio.2022.100219
Abraham DM, Herman C, Witek L, et al. Self-assembling human skeletal organoids for disease modeling and drug testing. J Biomed Mater Res Part B Appl Biomater. 2022;110:871–84. https://doi.org/10.1002/jbm.b.34968.
Iordachescu A, Hughes EAB, Joseph S, et al. Trabecular bone organoids: a micron-scale ‘humanised’ prototype designed to study the effects of microgravity and degeneration. npj Microgravity. 2021;7:. https://doi.org/10.1038/s41526-021-00146-8
Roth JG, Huang MS, Li TL, et al. Advancing models of neural development with biomaterials. Nat Rev Neurosci. 2021;22:593–615. https://doi.org/10.1038/s41583-021-00496-y.
Basu P, Basu A. In vitro and in vivo effects of flavonoids on peripheral neuropathic pain. Molecules. 2020;25:. https://doi.org/10.3390/molecules25051171
Bennett GJ, Xie Y-K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain. 1988;33:87–107. https://doi.org/10.1016/0304-3959(88)90209-6.
Seltzer Z, Dubner R, Shir Y. A novel behavioral model of neuropathic pain disorders produced in rats by partial sciatic nerve injury. Pain. 1990;43:205–18. https://doi.org/10.1016/0304-3959(90)91074-S.
Xie AX, Taves S, McCarthy K. Nuclear factor κB-COX2 pathway activation in non-myelinating Schwann cells is necessary for the maintenance of neuropathic pain in vivo. Front Cell Neurosci. 2022;15:. https://doi.org/10.3389/fncel.2021.782275
Schwarz N, Hedrich UBS, Schwarz H, et al. Human cerebrospinal fluid promotes long-term neuronal viability and network function in human neocortical organotypic brain slice cultures. Sci Rep. 2017;7:1–12. https://doi.org/10.1038/s41598-017-12527-9.
Tang-Schomer MD, White JD, Tien LW, et al. Bioengineered functional brain-like cortical tissue. Proc Natl Acad Sci USA. 2014;111:13811–6. https://doi.org/10.1073/pnas.1324214111.
Zhou L, Wolfes AC, Li Y, et al. Lipid-bilayer-supported 3D printing of human cerebral cortex cells reveals developmental interactions. Adv Mater. 2020;2002183:. https://doi.org/10.1002/adma.202002183
Johnson BN, Lancaster KZ, Hogue IB, et al. 3D printed nervous system on a chip. Lab Chip. 2016;16:1393–400. https://doi.org/10.1039/c5lc01270h.
Benton G, George J, Kleinman HK, Arnaoutova IP. Advancing science and technology via 3D culture on basement membrane matrix. J Cell Physiol. 2009;221:18–25. https://doi.org/10.1002/jcp.21832.
Frampton JP, Hynd MR, Shuler ML, Shain W. Fabrication and optimization of alginate hydrogel constructs for use in 3D neural cell culture. Biomed Mater. 2011;6:15002. https://doi.org/10.1088/1748-6041/6/1/015002.
Banerjee A, Arha M, Choudhary S, et al. The influence of hydrogel modulus on the proliferation and differentiation of encapsulated neural stem cells. Biomaterials. 2009;30:4695–9. https://doi.org/10.1016/j.biomaterials.2009.05.050.
Liu X, Hao M, Chen Z, et al. 3D bioprinted neural tissue constructs for spinal cord injury repair. Biomaterials. 2021;272:120771. https://doi.org/10.1016/j.biomaterials.2021.120771.
Gu Q, Tomaskovic-Crook E, Wallace GG, Crook JM. 3D bioprinting human induced pluripotent stem cell constructs for in situ cell proliferation and successive multilineage differentiation. Adv Healthc Mater. 2017;6:. https://doi.org/10.1002/adhm.201700175
Ning L, Sun H, Lelong T, et al. 3D bioprinting of scaffolds with living Schwann cells for potential nerve tissue engineering applications. Biofabrication. 2018;10:035014. https://doi.org/10.1088/1758-5090/aacd30.
Cullen DK, Stabenfeldt SE, Simon CM, et al. In vitro neural injury model for optimization of tissue-engineered constructs. J Neurosci Res. 2007;85:3642–51. https://doi.org/10.1002/jnr.21434.
Mahoney MJ, Anseth KS. Three-dimensional growth and function of neural tissue in degradable polyethylene glycol hydrogels. Biomaterials. 2006;27:2265–74. https://doi.org/10.1016/j.biomaterials.2005.11.007.
Jamal M, Kadam SS, Xiao R, et al. Bio-origami hydrogel scaffolds composed of photocrosslinked PEG bilayers. Adv Healthc Mater. 2013;2:1142–50. https://doi.org/10.1002/adhm.201200458.
Klotz BJ, Gawlitta D, Rosenberg AJWP, et al. Gelatin-methacryloyl hydrogels: towards biofabrication-based tissue repair. Trends Biotechnol. 2016;34:394–407. https://doi.org/10.1016/j.tibtech.2016.01.002.
Knowlton S, Anand S, Shah T, Tasoglu S. Bioprinting for neural tissue engineering. Trends Neurosci. 2018;41:31–46. https://doi.org/10.1016/j.tins.2017.11.001.
Chen J, Huang D, Wang L, et al. 3D bioprinted multiscale composite scaffolds based on gelatin methacryloyl (GelMA)/chitosan microspheres as a modular bioink for enhancing 3D neurite outgrowth and elongation. J Colloid Interface Sci. 2020;574:162–73. https://doi.org/10.1016/j.jcis.2020.04.040.
Smits IPM, Blaschuk OW, Willerth SM. Novel N-cadherin antagonist causes glioblastoma cell death in a 3D bioprinted co-culture model. Biochem Biophys Res Commun. 2020;529:162–8. https://doi.org/10.1016/j.bbrc.2020.06.001.
Kunze A, Giugliano M, Valero A, Renaud P. Micropatterning neural cell cultures in 3D with a multi-layered scaffold. Biomaterials. 2011;32:2088–98. https://doi.org/10.1016/j.biomaterials.2010.11.047.
Cho H, Seo JH, Wong KHK, et al. Three-dimensional blood-brain barrier model for in vitro studies of neurovascular pathology. Sci Rep. 2015;5:1–9. https://doi.org/10.1038/srep15222.
Lei Y, Li J, Wang N, et al. An on-chip model for investigating the interaction between neurons and cancer cells. Integr Biol. 2016;8:359–67. https://doi.org/10.1039/c5ib00309a.
Fan Y, Nguyen DT, Akay Y, et al. Engineering a brain cancer chip for high-throughput drug screening. Sci Rep. 2016;6:25062. https://doi.org/10.1038/srep25062.
de Jongh R, Spijkers XM, Pasteuning-Vuhman S, et al. Neuromuscular junction-on-a-chip: ALS disease modeling and read-out development in microfluidic devices. J Neurochem. 2021;157:393–412. https://doi.org/10.1111/jnc.15289.
Vysokov N, McMahon SB, Raouf R. The role of NaV channels in synaptic transmission after axotomy in a microfluidic culture platform. Sci Rep. 2019;9:12915. https://doi.org/10.1038/s41598-019-49214-w.
Lancaster MA, Renner M, Martin CA, et al. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501:373–9. https://doi.org/10.1038/nature12517.
Stem C, Author C, August PMC, et al. HHS public access neurons. 2017;19:248–257. https://doi.org/10.1016/j.stem.2016.07.005.Midbrain-like
Ao Z, Cai H, Wu Z, et al. Human spinal organoid-on-a-chip to model nociceptive circuitry for pain therapeutics discovery. Anal Chem. 2022;94:1365–72. https://doi.org/10.1021/acs.analchem.1c04641.
Thompson AL, Largent-Milnes TM, Vanderah TW. Animal models for the study of bone-derived pain. Methods Mol Biol. 2019;1914:391–407. https://doi.org/10.1007/978-1-4939-8997-3_23.
Remeniuk B, King T, Sukhtankar D, et al. Disease modifying actions of interleukin-6 blockade in a rat model of bone cancer pain. Pain. 2018;159:684–98. https://doi.org/10.1097/j.pain.0000000000001139.
Zhang Z, Deng M, Huang J, et al. Microglial annexin A3 downregulation alleviates bone cancer-induced pain through inhibiting the Hif-1α/vascular endothelial growth factor signaling pathway. Pain. 2020;161:2750–62. https://doi.org/10.1097/j.pain.0000000000001962.
Stine C, Coleman DL, Flohrschutz AT, et al. Heat shock protein 90 inhibitors block the antinociceptive effects of opioids in mouse chemotherapy-induced neuropathy and cancer bone pain models. Pain. 2020;161:1798–807. https://doi.org/10.1097/j.pain.0000000000001886.
Liu Q, Feng L, Han X, et al. The TRPA1 channel mediates mechanical allodynia and thermal hyperalgesia in a rat bone cancer pain model. Front Pain Res. 2021;2:1–10. https://doi.org/10.3389/fpain.2021.638620.
Forte BL, Slosky LM, Zhang H, et al. Angiotensin-(1–7)/Mas receptor as an antinociceptive agent in cancer-induced bone pain. Pain. 2016;157:2709–21. https://doi.org/10.1097/j.pain.0000000000000690.
Fan L-J, Kan H-M, Chen X-T, et al. Vascular endothelial growth factor-A/vascular endothelial growth factor2 signaling in spinal neurons contributes to bone cancer pain. Mol Pain. 2022;18:174480692210758. https://doi.org/10.1177/17448069221075891.
Nencini S, Thai J, Ivanusic JJ. Sequestration of artemin reduces inflammation-induced activation and sensitization of bone marrow nociceptors in a rodent model of carrageenan-induced inflammatory bone pain. Eur J Pain (United Kingdom). 2019;23:397–409. https://doi.org/10.1002/ejp.1315.
Townsend K, Imbert I, Eaton V, et al. Voluntary exercise blocks ongoing pain and diminishes bone remodeling while sparing protective mechanical pain in a rat model of advanced osteoarthritis pain. Pain. 2022;163:E476–87. https://doi.org/10.1097/j.pain.0000000000002392.
Nencini S, Morgan M, Thai J, et al. Piezo2 knockdown inhibits noxious mechanical stimulation and NGF-induced sensitization in A-delta bone afferent neurons. Front Physiol. 2021;12:1–13. https://doi.org/10.3389/fphys.2021.644929.
Thai J, Kyloh M, Travis L, et al. Identifying spinal afferent (sensory) nerve endings that innervate the marrow cavity and periosteum using anterograde tracing. J Comp Neurol. 2020;528:1903–16. https://doi.org/10.1002/cne.24862.
Morgan M, Nencini S, Thai J, Ivanusic JJ. TRPV1 activation alters the function of Aδ and C fiber sensory neurons that innervate bone. Bone. 2019;123:168–75. https://doi.org/10.1016/j.bone.2019.03.040.
Schindeler A, Mills RJ, Bobyn JD, Little DG. Preclinical models for orthopedic research and bone tissue engineering. J Orthop Res. 2018;36:832–40. https://doi.org/10.1002/jor.23824.
Haffner-Luntzer M, Hankenson KD, Ignatius A, et al. Review of animal models of comorbidities in fracture-healing research. J Orthop Res. 2019;37:2491–8. https://doi.org/10.1002/jor.24454.
Bonnarens F, Einhorn TA. Production of a standard closed fracture in laboratory animal bone. J Orthop Res Off Publ Orthop Res Soc. 1984;2:97–101. https://doi.org/10.1002/jor.1100020115.
Tägil M, McDonald MM, Morse A, et al. Intermittent PTH(1–34) does not increase union rates in open rat femoral fractures and exhibits attenuated anabolic effects compared to closed fractures. Bone. 2010;46:852–9. https://doi.org/10.1016/j.bone.2009.11.009.
Ruan MZC, Dawson B, Jiang MM, et al. Quantitative imaging of murine osteoarthritic cartilage by phase-contrast micro-computed tomography. Arthritis Rheum. 2013;65:388–96. https://doi.org/10.1002/art.37766.
Pereira JD, DuBreuil DM, Devlin AC, et al. Human sensorimotor organoids derived from healthy and amyotrophic lateral sclerosis stem cells form neuromuscular junctions. Nat Commun. 2021;12:. https://doi.org/10.1038/s41467-021-24776-4
Arguello F, Baggs RB, Frantz CN. A murine model of experimental metastasis to bone and bone marrow. Cancer Res. 1988;48:6876–81.
Schwei MJ, Honore P, Rogers SD, et al. Neurochemical and cellular reorganization of the spinal cord in a murine model of bone cancer pain. J Neurosci. 1999;19:10886–97. https://doi.org/10.1523/jneurosci.19-24-10886.1999.
Nordstrand A, Nilsson J, Tieva Å, et al. Establishment and validation of an in vitro co-culture model to study the interactions between bone and prostate cancer cells. Clin Exp Metastasis. 2009;26:945–53. https://doi.org/10.1007/s10585-009-9285-4.
Francesca S, Veronica B, Silvia B, et al. An in vitro 3D bone metastasis model by using a human bone tissue culture and human sex-related cancer cells. Oncotarget. 2016;7:76966–83. https://doi.org/10.18632/oncotarget.12763.
Acknowledgements
The authors have no conflicts of interest to declare. GC acknowledges funding from AIRC Aldi Fellowship under grant agreement No. 25412. ROCO acknowledges financial support from the Biotechnology and Biological Sciences Research Council (BB/P017711/1) and the UK Regenerative Medicine Platform “Acellular / Smart Materials – 3D Architecture” (MR/R015651/1). MR and AC acknowledge the University of Pennsylvania Orphan Disease Center in partnership with the Fibrous Dysplasia Foundation (MDBR-21-110-FD) and Sapienza University RM120172B8BF5C15, RM118164289636F0). Figures were generated using Servier Medical Art (by Servier, licensed under a Creative Commons Attribution 3.0 Unported License).
Funding
Open access funding provided by Istituto Italiano di Tecnologia within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Iafrate, L., Benedetti, M.C., Donsante, S. et al. Modelling skeletal pain harnessing tissue engineering. In vitro models 1, 289–307 (2022). https://doi.org/10.1007/s44164-022-00028-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44164-022-00028-7