Abstract
Introduction
Osteoporosis is a skeletal disease characterized by loss of bone mass, reduced bone strength and increased bone fragility predisposing to fractures. This study was planned to evaluate the efficacy, safety and relative bioavailability of Microcore NESC® (Natural Egg Shell Calcium) in osteopenia and osteoporotic patients.
Methods
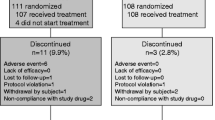
This was a randomized, open label, parallel group interventional clinical trial which included 60 study participants with osteopenia and osteoporosis who were randomized into three groups (20 each). Group 1—Microcore NESC®, Group 2—Shelcal and Group 3—CCM with 12 weeks treatment period. The participants were evaluated for relative oral bioavailability, bone mineral density (BMD), serum osteocalcin, change in VAS pain scale and quality of life—Questionnaires.
Results
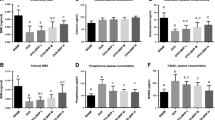
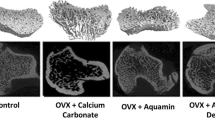
There was significant improvement in the BMD T scores—post-treatment with MICROCORE NESC® and Shelcal. Higher percentage of improvement in calcium absorption as depicted by an increase in serum calcium levels (10.23%) in the MICROCORE NESC®-treated group when compared to Shelcal (7.7%) and CCM (7.2%). The relative bioavailability of MICROCORE NESC® with respect to Shelcal was 93%.
Discussion
MICROCORE NESC®, has shown a better oral relative bio availability of calcium (93%), better improvement of BMD T score compared to Shelcal and CCM. The general health status has improved to very good/excellent in 83% of patients in MICROCORE NESC®-treated group. Thus, MICROCORE NESC® can be considered a better and safe calcium supplement, as there are very few side effects observed without any clinically significant abnormalities in lab parameters.



Similar content being viewed by others
Data availability
The data that supports the findings of this study will be available on request to the corresponding author.
References
NIH consensus development panel on osteoporosis prevention. (2001). diagnosis, and therapy, march 7–29 2000: Highlights of the conference. Southern Medical Journal, 94, 569–573.
Charles, P. (1992). Calcium absorption and calcium bioavailability. Journal of Internal Medicine, 231, 161–168.
Reginster, J. Y., Denis, D., Bartsch, V., Deroisy, R., Zegels, B., Franchimont, P., et al. (1993). Acute biochemical variations induced by four different calcium salts in healthy male participants. Osteoporosis International, 3, 271–275.
Wood, R. J., & Martini, L. (2003). Reply to R. P. Heaney. The American Journal of Clinical Nutrition, 78, 493–494.
Levenson, D. I., & Bockman, R. S. (1994). A review of calcium preparations. Nutrition Reviews, 52, 221–232.
Porter, C. (2003). Vitamin and mineral supplements: Part 1. Rx Consultant., 12, 1–8.
Sakhaee, K., Bhuket, T., Adams-Huet, B., & Rao, D. S. (1999). Meta-analysis of calcium bioavailability: A comparison of calcium citrate with calcium carbonate. American Journal of Therapeutics, 6, 313–321.
Heller, H. J., Stewart, A., Haynes, S., & Pak, C. Y. (1999). Pharmacokinetics of calcium absorption from two commercial calcium supplements. Journal of Clinical Pharmacology, 39, 1151–1154.
Heaney, R. P. (2001). Factors influencing the measurement of bioavailability, taking calcium as a model. Journal of Nutrition, 131, 1334–1348.
Abrams, S.A., Copeland, K.C., Gunn, S.K., Gundberg, C.M., Klein, K.O., Ellis, K.J. (2000). Calcium absorption, bone mass accumulation, and kinetics increase during early pubertal development in girls. The Journal of Clinical Endocrinology & Metabolism, 85(5):1805
Shankar, K., M, S., Raizada, P., & Jain, R. (2018). A Randomized open-label clinical study comparing the efficacy, safety, and bioavailability of calcium lysinate with calcium carbonate and calcium citrate malate in osteopenia participants. Journal of Orthopaedic Case Reports, 8(4), 15–19.
Rovenský, J., Stancíková, M., Masaryk, P., Svík, K., & Istok, R. (2003). Eggshell calcium in the prevention and treatment of osteoporosis. International Journal of Clinical Pharmacology Research, 23(2–3), 83–92.
Ruff, K. J., Winkler, A., Jackson, R. W., DeVore, D. P., & Ritz, B. W. (2009). Eggshell membrane in the treatment of pain and stiffness from osteoarthritis of the knee: A randomized, multicenter, double-blind, placebo-controlled clinical study. Clinical Rheumatology, 28(8), 907–914.
Benson, K. F., Ruff, K. J., & Jensen, G. S. (2012). Effects of natural eggshell membrane (NEM) on cytokine production in cultures of peripheral blood mononuclear cells: Increased suppression of tumor necrosis factor-α levels after in vitro digestion. Journal of Medicinal Food, 15(4), 360–368.
Laird, E., Ward, M., McSorley, E., Strain, J. J., & Wallace, J. (2010). Vitamin D and bone health: Potential mechanisms. Nutrients, 2(7), 693–724.
Kołodziejska, B., Stępień, N., & Kolmas, J. (2021). The influence of strontium on bone tissue metabolism and its application in osteoporosis treatment. International Journal of Molecular Sciences, 22(12), 6564.
Castiglioni, S., Cazzaniga, A., Albisetti, W., & Maier, J. A. (2013). Magnesium and osteoporosis: Current state of knowledge and future research directions. Nutrients, 5(8), 3022–3033.
O’Connor, J. P., Kanjilal, D., Teitelbaum, M., Lin, S. S., & Cottrell, J. A. (2020). Zinc as a therapeutic agent in bone regeneration. Materials (Basel), 13(10), 2211.
Fung, E. B., Kwiatkowski, J. L., Huang, J. N., Gildengorin, G., King, J. C., & Vichinsky, E. P. (2013). Zinc supplementation improves bone density in participants with thalassemia: A double-blind, randomized, placebo-controlled trial. American Journal of Clinical Nutrition, 98(4), 960–971.
Acknowledgements
We acknowledge the sponsor (Microcore) and the CRO (ki3 Pvt. Ltd).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The study was sponsored by Microcore.
Ethical Standard Statement
This article does not contain any studies with human or animal subjects performed by the any of the authors.
Informed Consent
Informed consent was obtained from the study participants.
Limitations
Sample size was small and only relative bio availability was calculated in our study. Baseline Vitamin D 3 and PTH levels could have been monitored to rule out other factors affecting calcium metabolism.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gopal, K., Deb, B., Uma, N. et al. Comparative Evaluation of Bioavailability, Efficacy and Safety of MICROCORE NESC® with Calcium Carbonate and Calcium Citrate Malate in Osteopenic and Osteoporotic Patients: A Randomized Clinical Trial. JOIO 58, 535–541 (2024). https://doi.org/10.1007/s43465-024-01117-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43465-024-01117-3