Abstract
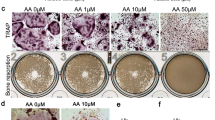
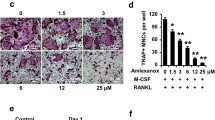
Bone homeostasis is dynamically balanced by osteoclastic bone resorption and osteoblastic bone formation. Increased numbers and enhanced activities of osteoclasts can lead to diseases, such as osteoporosis, aseptic prosthetic loosening, rheumatoid arthritis, and tumor bone metastasis. In postmenopausal women, enhanced osteoclast formation causes excessive bone matrix resorption, less bone mass, and fragile bone mechanical property. Given the importance of osteoclast in osteolytic diseases, inhibiting osteoclast is considered as an effective method for the prevention or treatment of these diseases. In this study, we initially assessed the effects of artemether on osteoclast formation, osteoblast differentiation, and bone mineralization in vitro, further proved by ovariectomy-induced bone loss model in vivo. Here, we found that artemether effectively inhibited osteoclastogenesis in vitro and suppressed ovariectomy-induced bone loss, yet had no effect on osteoblast differentiation and osteoblast-related gene expression.
Graphical Abstract





Similar content being viewed by others
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Boyce BF (2013) Advances in the regulation of osteoclasts and osteoclast functions. J Dent Res 92:860–867. https://doi.org/10.1177/0022034513500306
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342. https://doi.org/10.1038/nature01658
Cappariello A (2014) The great beauty of the osteoclast. Arch Biochem Biophys 15:70–78. https://doi.org/10.1016/j.abb.2014.06.017
Chen S, Jin G, Huang KM, Ma JJ, Wang Q, Ma Y, Tang XZ, Zhou ZJ, Hu ZJ, Wang JY, Qin A, Fan SW (2015) Lycorine suppresses RANKL-induced osteoclastogenesis in vitro and prevents ovariectomy-induced osteoporosis and titanium particle-induced osteolysis in vivo. Sci Rep 5:12853. https://doi.org/10.1038/srep12853
Commons RJ, Simpson JA, Thriemer K, Abreha T, Adam I, Anstey NM, Assefa A, Awab GR, Baird JK, Barber BE, Chu CS, Dahal P, Daher A, Davis TME, Dondorp AM, Grigg MJ, Humphreys GS, Hwang J, Karunajeewa H, Laman M, Lidia K, Moore BR, Mueller I, Nosten F, Pasaribu AP, Pereira DB, Phyo AP, Poespoprodjo JR, Sibley CH, Stepniewska K, Sutanto I, Thwaites G, Hien TT, White NJ, William T, Woodrow CJ, Guerin PJ, Price RN (2019) The efficacy of dihydroartemisinin-piperaquine and artemether-lumefantrine with and without primaquine on Plasmodium vivax recurrence: a systematic review and individual patient data meta-analysis. PLoS Med 16:e1002928. https://doi.org/10.1371/journal.pmed.1002928
Cuzzocrea S, Saadat F, Di Paola R, Mirshafiey A (2005) Artemether: a new therapeutic strategy in experimental rheumatoid arthritis. Immunopharmacol Immunotoxicol 27:615–630. https://doi.org/10.1080/08923970500418786
Deliloglu-Gurhan SI, Vatansever HS, Ozdal-Kurt F, Tuglu I (2006) Characterization of osteoblasts derived from bone marrow stromal cells in a modified cell culture system. Acta Histochem 108:49–57. https://doi.org/10.1016/j.acthis.2005.11.001
Drake MT, Cremers SC (2010) Bisphosphonate therapeutics in bone disease: the hard and soft data on osteoclast inhibition. Mol Interv 10:141–152. https://doi.org/10.1124/mi.10.3.5
Ebong C, Sserwanga A, Namuganga JF, Kapisi J, Mpimbaza A, Gonahasa S, Asua V, Gudoi S, Kigozi R, Tibenderana J, Bwanika JB, Bosco A, Rubahika D, Kyabayinze D, Opigo J, Rutazana D, Sebikaari G, Belay K, Niang M, Halsey ES, Moriarty LF, Lucchi NW, Souza SSS, Nsobya SL, Kamya MR, Yeka A (2021) Efficacy and safety of artemether-lumefantrine and dihydroartemisinin-piperaquine for the treatment of uncomplicated Plasmodium falciparum malaria and prevalence of molecular markers associated with artemisinin and partner drug resistance in Uganda. Malar J 20:484. https://doi.org/10.1186/s12936-021-04021-5
Feng MX, Hong JX, Wang Q, Fan YY, Yuan CT, Lei XH, Zhu M, Qin A, Chen HX, Hong D (2016) Dihydroartemisinin prevents breast cancer-induced osteolysis via inhibiting both breast cancer cells and osteoclasts. Sci Rep 6:19074. https://doi.org/10.1038/srep19074
Fu W, Ma Y, Li L, Liu J, Fu L, Guo Y, Zhang Z, Li J, Jiang H (2020) Artemether regulates metaflammation to improve glycolipid metabolism in db/db mice. Diabetes Metab Syndr Obes 13:1703–1713. https://doi.org/10.2147/DMSO.S240786
Geng Q, Gao H, Yang R, Guo K, Miao D (2019) Pyrroloquinoline quinone prevents estrogen deficiency-induced osteoporosis by inhibiting oxidative stress and osteocyte senescence. Int J Biol Sci 15:58–68. https://doi.org/10.7150/ijbs.25783
Gossiel F, Hoyle C, McCloskey EV, Naylor KE, Walsh J, Peel N, Eastell R (2016) The effect of bisphosphonate treatment on osteoclast precursor cells in postmenopausal osteoporosis: the TRIO study. Bone 92:94–99. https://doi.org/10.1016/j.bone.2016.08.010
Honma M, Ikebuchi Y, Kariya Y, Suzuki H (2014) Regulatory mechanisms of RANKL presentation to osteoclast precursors. Curr Osteoporos Rep 12:115–120. https://doi.org/10.1007/s11914-014-0189-0
Hu JP, Nishishita K, Sakai E, Yoshida H, Kato Y, Tsukuba T, Okamoto K (2008) Berberine inhibits RANKL-induced osteoclast formation and survival through suppressing the NF-kappaB and Akt pathways. Eur J Pharmacol 580:70–79. https://doi.org/10.1016/j.ejphar.2007.11.013
Ikeda K, Takeshita S (2014) Factors and mechanisms involved in the coupling from bone resorption to formation: how osteoclasts talk to osteoblasts. J Bone Metab 21:163–167. https://doi.org/10.11005/jbm.2014.21.3.163
Koga T, Inui M, Inoue K, Kim S, Suematsu A, Kobayashi E, Iwata T, Ohnishi H, Matozaki T, Kodama T, Taniguchi T, Takayanagi H, Takai T (2004) Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428:758–763. https://doi.org/10.1038/nature02444
Kumar A, Mahendra J, Mahendra L, Abdulkarim HH, Sayed M, Mugri MH, Ahmad ZH, Bhati AK, Faqehi HH, Algregri WO, Varadarajan S, Balaji TM, Baeshen HA, Patil S (2021) Synergistic effect of biphasic calcium phosphate and platelet-rich fibrin attenuate markers for inflammation and osteoclast differentiation by suppressing NF-κB/MAPK signaling pathway in chronic periodontitis. Molecules 26:6578. https://doi.org/10.3390/molecules26216578
Lasco A, Morabito N, Basile G, Atteritano M, Gaudio A, Giorgianni GM, Morini E, Faraci B, Bellone F, Catalano A (2016) Denosumab inhibition of RANKL and insulin resistance in postmenopausal women with osteoporosis. Calcif Tissue Int 98:123–128. https://doi.org/10.1007/s00223-015-0075-5
Li QZY-P (2007) Osteoclast differentiation and gene regulation. Front Biosci 12:2519–2529. https://doi.org/10.2741/2252
Martin TJ (2013) Historically significant events in the discovery of RANK/RANKL/OPG. World J Orthop 4:186–197. https://doi.org/10.5312/wjo.v4.i4.186
Matsushita Y, Hayashida S, Morishita K, Sakamoto H, Naruse T, Sakamoto Y, Yamada SI, Yanamoto S, Fujita S, Ikeda T, Umeda M (2016) Denosumab-associated osteonecrosis of the jaw affects osteoclast formation and differentiation: pathological features of two cases. Mol Clin Oncol 4:191–194. https://doi.org/10.3892/mco.2015.696
Mirzaei-Parsa MJ, Najafabadi MRH, Haeri A, Zahmatkeshan M, Ebrahimi SA, Pazoki-Toroudi H, Adel M (2020) Preparation, characterization, and evaluation of the anticancer activity of artemether-loaded nano-niosomes against breast cancer. Breast Cancer 27:243–251. https://doi.org/10.1007/s12282-019-01014-w
Miyamoto T (2013) Role of osteoclasts in regulating hematopoietic stem and progenitor cells. World J Orthop 4:198–206. https://doi.org/10.5312/wjo.v4.i4.198
Nhama A, Nhamussua L, Macete E, Bassat Q, Salvador C, Enosse S, Candrinho B, Carvalho E, Nhacolo A, Chidimatembue A, Saifodine A, Zulliger R, Lucchi N, Svigel SS, Moriarty LF, Halsey ES, Mayor A, Aide P (2021) In vivo efficacy and safety of artemether-lumefantrine and amodiaquine-artesunate for uncomplicated Plasmodium falciparum malaria in Mozambique 2018. Malar J 20:390. https://doi.org/10.1186/s12936-021-03922-9
Plotkin LI (2014) Apoptotic osteocytes and the control of targeted bone resorption. Curr Osteoporos Rep 12:121–126. https://doi.org/10.1007/s11914-014-0194-3
Potutkin DS, Tipisova EV, Kipriyanova KE, Gorenko IN, Popkova VA, Elfimova AE, Lobanov AA, Popov AI, Andronov SV (2018) Levels of sex hormones, sex hormone binding globulin, antisperm antibodies and dopamine in postmanopausal women of nomadic and settled aboriginal and local caucasoid populations of the arctic territories. Klin Lab Diagn 63:761–767. https://doi.org/10.18821/0869-2084-2018-63-12-761-767
Purdue PE, Koulouvaris P, Potter HG, Nestor BJ, Sculco TP (2007) The cellular and molecular biology of periprosthetic osteolysis. Clin Orthop Relat Res 454:251–261. https://doi.org/10.1097/01.blo.0000238813.95035.1b
Ren ZQ, Wang YF, Ao GF, Chen HX, Huang M, Lai MX, Zhao HD, Zhao R (2020) Overall adjustment acupuncture for postmenopausal osteoporosis (PMOP): a study protocol for a randomized sham-controlled trial. Trials 21:465. https://doi.org/10.1186/s13063-020-04435-7
Sasaki T, Kawashima H, Ariizumi T, Yamagishi T, Oike N, Umezu H, Inagawa S, Hotta T, Endo N, Ogose A (2018) Denosumab as a potential therapeutic option for leiomyosarcoma with osteoclast-like giant cells: a case report. Mol Clin Oncol 8:30–33. https://doi.org/10.3892/mco.2017.1489
Sims NA, Vrahnas C (2014) Regulation of cortical and trabecular bone mass by communication between osteoblasts, osteocytes and osteoclasts. Arch Biochem Biophys 561:22–28. https://doi.org/10.1016/j.abb.2014.05.015
Souza PP, Lerner UH (2013) The role of cytokines in inflammatory bone loss. Immunol Invest 42:555–622. https://doi.org/10.3109/08820139.2013.822766
Taylor-Miller T, Sivaprakasam P, Smithson SF, Steward CG, Burren CP (2021) Challenges in long-term control of hypercalcaemia with denosumab after haematopoietic stem cell transplantation for TNFRSF11A osteoclast-poor autosomal recessive osteopetrosis. Bone Rep 14:100738. https://doi.org/10.1016/j.bonr.2020.100738
Teti A (2013) Mechanisms of osteoclast-dependent bone formation. Bonekey Rep 2:449. https://doi.org/10.1038/bonekey.2013.183
Wang J, Yao L, Wu X, Guo Q, Sun S, Li J, Shi G, Caldwell RB, Caldwell RW, Chen Y (2021) Protection against doxorubicin-induced cardiotoxicity through modulating iNOS/ARG 2 balance by electroacupuncture at PC6. Oxid Med Cell Longev 2021:6628957. https://doi.org/10.1155/2021/6628957
Wu H, Hu B, Zhou X, Zhou C, Meng J, Yang Y, Zhao X, Shi Z, Yan S (2018) Artemether attenuates LPS-induced inflammatory bone loss by inhibiting osteoclastogenesis and bone resorption via suppression of MAPK signaling pathway. Cell Death Dis 9:498. https://doi.org/10.1038/s41419-018-0540-y
Wu Z, Li C, Chen Y, Liu Q, Li N, He X, Li W, Shen R, Li L, Wei C, Shao S, Fu F, Ding J, Sun X, Wang D, Yuan G, Su Y, Zhao J, Xu J, Xu R, Xu X, Xu F (2022) Chrysin protects against titanium particle-induced osteolysis by attenuating osteoclast formation and function by inhibiting NF-κB and MAPK signaling. Front Pharmacol 13:793087. https://doi.org/10.3389/fphar.2022.793087
Yao Y, Cai X, Ren F, Ye Y, Wang F, Zheng C, Qian Y, Zhang M (2021) The macrophage-osteoclast axis in osteoimmunity and osteo-related diseases. Front Immunol 12:664871. https://doi.org/10.3389/fimmu.2021.664871
Yasuda H (2013) RANKL, a necessary chance for clinical application to osteoporosis and cancer-related bone diseases. World J Orthop 4:207–217. https://doi.org/10.5312/wjo.v4.i4.207
Funding
This work was supported by the Public Welfare Foundation Project of Zhejiang Province for Dr. Xiao Teng, China (LGF18H070004); the Public Welfare Foundation Project of Zhejiang Province for Dr. Ming-Xuan Feng, China (LGF19H070003); Public Welfare Foundation Project of Zhejiang Province for Dr. Zhao-bo Zhang, China (LGF20H070005); and Science and Technology Foundation of Taizhou City, Zhejiang Province (1901ky39).
Author information
Authors and Affiliations
Contributions
XT and DH designed most of the experiments. M-X F, XC and Z-B Z preformed animals’ surgery. M-X F, XC and J-S Z carried out the cell experiments. M-X F, XT and DH analyzed the data. M-XF wrote the paper. All authors approved the final manuscript for publication.
Corresponding authors
Ethics declarations
Ethical Approval
The animal experiments in this study were conducted in accordance with guidelines and procedures authorized by the Animal Experimental Ethical Committee of Taizhou Central Hospital, Taizhou University (SYXK 2019–0030).
Competing Interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, MX., Zhang, ZB., Cheng, X. et al. Artemether Inhibits RANKL-Induced Osteoclast Differentiation and Prevents Bone Loss in Ovariectomized Mice. Rev. Bras. Farmacogn. 33, 812–821 (2023). https://doi.org/10.1007/s43450-023-00377-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00377-8