Abstract
Background
Ion channels have been proposed as therapeutic targets for different types of malignancies. One of the most studied ion channels in cancer is the voltage-gated potassium channel ether-à-go-go 1 or Kv10.1. Various studies have shown that Kv10.1 expression induces the proliferation of several cancer cell lines and in vivo tumor models, while blocking or silencing inhibits proliferation. Kv10.1 is a promising target for drug discovery modulators that could be used in cancer treatment. This work aimed to screen for new Kv10.1 channel modulators using a thallium influx-based assay.
Methods
Pharmacological effects of small molecules on Kv10.1 channel activity were studied using a thallium-based fluorescent assay and patch-clamp electrophysiological recordings, both performed in HEK293 stably expressing the human Kv10.1 potassium channel.
Results
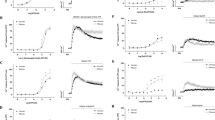
In thallium-sensitive fluorescent assays, we found that the small molecules loperamide and amitriptyline exert a potent inhibition on the activity of the oncogenic potassium channel Kv10.1. These results were confirmed by electrophysiological recordings, which showed that loperamide and amitriptyline decreased the amplitude of Kv10.1 currents in a dose-dependent manner. Both drugs could be promising tools for further studies.
Conclusions
Thallium-sensitive fluorescent assay represents a reliable methodological tool for the primary screening of different molecules with potential activity on Kv10.1 channels or other K+ channels.





Similar content being viewed by others
Abbreviations
- AMI:
-
Amitriptyline
- Eag1:
-
Human ether-à-go-go potassium channel 1
- HEK-WT:
-
HEK293 wild-type cells
- HEK-Kv10.1:
-
HEK293 cells stably expressing the human Kv10.1 potassium channel
- LP:
-
Loperamide
- SR:
-
SR 33805 oxalate
References
Alexander SPH, Striessnig J, Kelly E, Marrion NV, Peters JA, Faccenda E, et al. THE CONCISE GUIDE TO PHARMACOLOGY 2017/18: voltage-gated ion channels. Br J Pharmacol. 2017;1(174):S160–94.
Ashcroft FM. From molecule to malady. Nature. 2006; 440:440-47.
Yu FH, Catterall WA. The VGL-chanome: a protein superfamily specialized for electrical signaling and ionic homeostasis. Sci STKE. 2004;2004:1–17.
Whicher JR, MacKinnon R. Structure of the voltage-gated K+ channel Eag1 reveals an alternative voltage sensing mechanism. Science (80-). 2016;353(6300):664–9.
Wang X, Chen Y, Zhang Y, Guo S, Mo L, An H, et al. Eag1 voltage-dependent potassium channels: structure, electrophysiological characteristics, and function in cancer. J Membr Biol. 2017;250:123−132.
Occhiodoro T, Bernheim L, Liu JH, Bijlenga P, Sinnreich M, Bader CR, et al. Cloning of a human ether-a-go-go potassium channel expressed in myoblasts at the onset of fusion. FEBS Lett. 1998;434:177-82.
Bauer CK, Schwarz JR. Ether-à-go-go K + channels: effective modulators of neuronal excitability. J Physiol. 2018;596:769–83.
Martin S, Lino de Oliveira C, Mello de Queiroz F, Pardo LA, Stühmer W, Del Bel E. Eag1 potassium channel immunohistochemistry in the CNS of adult rat and selected regions of human brain. Neuroscience. 2008;155:833–44.
Mortensen LS, Schmidt H, Farsi Z, Barrantes-Freer A, Rubio ME, Ufartes R et al. Kv10.1 opposes activity-dependent increase in Ca2+ influx into the presynaptic terminal of the parallel fibre-Purkinje cell synapse. J Physiol. 2015;593:181–96.
Hemmerlein B, Weseloh RM, de Queiroz FM, Knötgen H, Sánchez A, Rubio ME, et al. Overexpression of Eag1 potassium channels in clinical tumours. Mol Cancer. 2006;5(41):1–13.
Rodríguez-Rasgado JA, Acuña-Macías I, Camacho J. Eag1 channels as potential cancer biomarkers. Sensors (Switzerland). 2012;12(5):5986–95.
Ferreira NR, Mitkovski M, Stühmer W, Pardo LA, Del Bel EA. Ether-à-go-go 1 (Eag1) potassium channel expression in dopaminergic neurons of basal ganglia is modulated by 6-hydroxydopamine lesion. Neurotox Res. 2012;21(3):317–33.
Pardo LA, Del Camino D, Sánchez A, Alves F, Brüggemann A, Beckh S, et al. Oncogenic potential of EAG K+ channels. EMBO J. 1999;18:5540–47.
Gómez-Varela D, Zwick-Wallasch E, Knötgen H, Sánchez A, Hettmann T, Ossipov D, et al. Monoclonal antibody blockade of the human Eag1 potassium channel function exerts antitumor activity. Cancer Res. 2007;67:7343–49.
García-Becerra R, Díaz L, Camacho J, Barrera D, Ordaz-Rosado D, Morales A, et al. Calcitriol inhibits Ether-à go-go potassium channel expression and cell proliferation in human breast cancer cells. Exp Cell Res. 2010;316:433–42.
Bernal-Ramos G, Hernández-Gallegos E, Vera E, Chávez-López M de G, Zúñiga-García V, Sánchez-Pérez Y, et al. Astemizole inhibits cell proliferation in human prostate tumorigenic cells expressing ether à-go-go-1 potassium channels. Cell Mol Biol. 2017;63:11–3.
Wang X, Chen Y, Li J, Guo S, Lin X, Zhang H, et al. Tetrandrine, a novel inhibitor of ether-à-go-go-1 (Eag1), targeted to cervical cancer development. J Cell Physiol. 2019;234:7161–73.
Gavrilova-Ruch O, Schönherr K, Gessner G, Schönherr R, Klapperstück T, Wohlrab W, et al. Effects of imipramine on ion channels and proliferation of IGR1 melanoma cells. J Membr Biol. 2002;188:137–49.
García-Quiroz J, Camacho J. Astemizole: an old anti-histamine as a new promising anti-cancer drug. Anticancer Agents Med Chem. 2012;11(3):307-14.
Wu J, Zhong D, Fu X, Liu Q, Kang L, Ding Z. Silencing of Ether à go-go 1 by shRNA inhibits osteosarcoma growth and cell cycle progression. Int J Mol Sci. 2014;15(4) 5570–81.
Yu HB, Li M, Wang WP, Wang XL. High throughput screening technologies for ion channels. Acta Pharmacologica Sinica. 2016;37:34-43.
Philippaert K, Kerselaers S, Voets T, Vennekens R. A thallium-based screening procedure to identify molecules that modulate the activity of Ca2+-activated monovalent cation-selective channels. SLAS Discov. 2018;23:341–52.
Huang XP, Mangano T, Hufeisen S, Setola V, Roth BL. Identification of human Ether-à-go-go related gene modulators by three screening platforms in an academic drug-discovery setting. Assay Drug Dev Technol. 2010;8:727–42.
Weaver CD, Harden D, Dworetzky SI, Robertson B, Knox RJ. A thallium-sensitive, fluorescence-based assay for detecting and characterizing potassium channel modulators in mammalian cells. J Biomol Screen. 2004;9:671–77.
Zhang J-H, Chung TDY, Oldenburg KR. A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J Biomol Screen. 1999;4(2):67–73.
Visan V, Fajloun Z, Sabatier JM, Grissmer S. Mapping of maurotoxin binding sites on hKv1.2, hKv1.3, and hlKCa1 channels. Mol Pharmacol. 2004;66:1103–12.
García-Ferreiro RE, Kerschensteiner D, Major F, Monje F, Stühmer W, Pardo LA. Mechanism of block of hEag1 K+ channels by imipramine and astemizole. J Gen Physiol. 2004;124:301-17.
Cazorla O, Lacampagne A, Fauconnier J, Vassort G. SR33805, a Ca2+ antagonist with length-dependent Ca2+-sensitizing properties in cardiac myocytes. Br J Pharmacol. 2003;139:99–108.
Church J, Fletcher EJ, Abdel-Hamid K, MacDonald JF. Loperamide blocks high-voltage-activated calcium channels and N-methyl-d-aspartate-evoked responses in rat and mouse cultured hippocampal pyramidal neurons. Mol Pharmacol. 1994;45(4):747–57.
Wu Y, Zou B, Liang L, Li M, Tao YX, Yu H, et al. Loperamide inhibits sodium channels to alleviate inflammatory hyperalgesia. Neuropharmacology. 2017;117:282-291.
Hagiwara K, Nakagawasai O, Murata A, Yamadera F, Miyoshi I, Tan-No K, et al. Analgesic action of loperamide, an opioid agonist, and its blocking action on voltage-dependent Ca2+ channels. Neurosci Res. 2003;46(4):493–7.
Hanauer SB. The role of loperamide in gastrointestinal disorders. Rev Gastroenterol Disord. 2008;8(1):15–20.
Zahradník I, Minarovič I, Zahradníková A. Inhibition of the cardiac l-type calcium channel current by antidepressant drugs. J Pharmacol Exp Ther. 2008;324:977–84.
Song JH, Ham SS, Shin YK, Lee CS. Amitriptyline modulation of Na+ channels in rat dorsal root ganglion neurons. Eur J Pharmacol. 2000;401:297-305.
Garg V, Sachse FB, Sanguinetti MC. Tuning of EAG K+ channel inactivation: Molecular determinants of amplification by mutations and a small molecule. J Gen Physiol. 2012;140:307–24.
Vetter I, Carter D, Bassett J, Deuis JR, Tay B, Jami S, et al. High-throughput fluorescence assays for ion channels and GPCRs. In: Advances in experimental medicine and biology. 2020;27–72.
Ufartes R, Schneider T, Mortensen LS, Romero C de J, Hentrich K, Knoetgen H, et al. Behavioural and functional characterization of Kν10.1 (Eag1) knockout mice. Hum Mol Genet. 2013;22(11):2247–62.
Gharagozlou P, Demirci H, Clark JD, Lameh J. Activation profiles of opioid ligands in HEK cells expressing δ opioid receprtors. BMC Neurosci. 2002;3:1-8.
Gharagozlou P, Demirci H, Clark JD, Lameh J. Activity of opioid ligands in cells expressing cloned μ opioid receptors. BMC Pharmacol. 2003;3:1-8.
Gómez-Lagunas F, Carrillo E, Pardo LA, Stühmer W. Gating modulation of the tumor-related Kv10.1 channel by Mibefradil. J Cell Physiol. 2017;232(8):2019–32.
Moreels L, Bhat C, Voráčová M, Peigneur S, Goovaerts H, Mäki-Lohiluoma E, et al. Synthesis of novel purpurealidin analogs and evaluation of their effect on the cancer-relevant potassium channel KV10.1. PLoS One. 2017;12(12):1-18.
Humphries ESA, Dart C. Neuronal and cardiovascular potassium channels as therapeutic drug targets: promise and pitfalls. J Biomol Screen. 2015;20:1055–73.
Urrego D, Tomczak AP, Zahed F, Stühmer W, Pardo LA. Potassium channels in cell cycle and cell proliferation. Philos Trans R Soc B Biol Sci. 2014;369(1638):1–9.
Rao VR, Perez-Neut M, Kaja S, Gentile S. Voltage-gated ion channels in cancer cell proliferation. Cancers. 2015;7:849–75.
Urrego D, Movsisyan N, Ufartes R, Pardo LA. Periodic expression of Kv10.1 driven by pRb/E2F1 contributes to G2/M progression of cancer and non-transformed cells. Cell Cycle. 2016;15:799-811.
Zhou Y, Wong CO, Cho KJ, Van Der Hoeven D, Liang H, Thakur DP, et al. Membrane potential modulates plasma membrane phospholipid dynamics and K-Ras signaling. Science (80-). 2015;349(6250):873–6.
Yang M, Brackenbury WJ. Membrane potential and cancer progression. Front Physiol. 2013;4:1-10.
Kadir LA, Stacey M, Barrett-Jolley R. Emerging roles of the membrane potential: Action beyond the action potential. Front Physiol. 2018;9:1–10.
King GF. Venoms as a platform for human drugs: translating toxins into therapeutics. Expert Opinion on Biological Therapy. 2011;11(11):1469–84. Available from: https://doi.org/10.1517/14712598.2011.621940
Moreels L, Peigneur S, Galan DT, De Pauw E, Béress L, Waelkens E, et al. APETx4, a novel sea anemone toxin and a modulator of the cancer-relevant potassium channel KV10.1. Mar Drugs. 2017;15(9) 1-17.
Twiner MJ, Doucette GJ, Rasky A, Huang XP, Roth BL, Sanguinetti MC. Marine algal toxin azaspiracid is an open-state blocker of hERG potassium channels. Chem Res Toxicol. 2012; 25, 1975-1984.
Moreels L, Peigneur S, Yamaguchi Y, Vriens K, Waelkens E, Zhu S, et al. Expanding the pharmacological profile of κ-hefutoxin 1 and analogues: a focus on the inhibitory effect on the oncogenic channel Kv101. Peptides. 2017;98:43–50.
Funding
This work was supported by Grants: 314839, 21887 and 315803 from Consejo Nacional de Ciencia y Tecnología (CONACYT) and PAPIIT-UNAM IG200119 to AH-C, and SEP-CONACYT CB2017-2018-A1-S-13646 to E.L. We thank Fis. Cesar Oliver Lara Figueroa and Dr. Luisa Duran Pasten for technical assistance. The English revision by Dr. Bristol Denlinger is also acknowledged.
Author information
Authors and Affiliations
Contributions
AL–H, EM and EL carried out the experiments; EL and AH-C designed, analyzed, and interpreted the results; EL wrote the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Loza-Huerta, A., Milo, E., Picones, A. et al. Thallium-sensitive fluorescent assay reveals loperamide as a new inhibitor of the potassium channel Kv10.1. Pharmacol. Rep 73, 1744–1753 (2021). https://doi.org/10.1007/s43440-021-00304-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43440-021-00304-5