Abstract
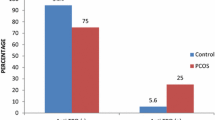
To compare the rate of positive thyroid peroxidase antibodies (TPO Ab) between women with different polycystic ovary syndrome (PCOS) phenotypes and women without PCOS. This is a retrospective cohort study. Women with PCOS at My Duc Hospital between June 1, 2020, and March 27, 2021, were matched with non-PCOS women by age. TPO Ab (cut-off: 34 IU/mL) and thyroid-stimulating hormone (TSH) levels were measured as markers of Hashimoto thyroiditis and thyroid function, respectively. One thousand eight hundred eight infertile women were included, 904 with PCOS (mean age 29.0 ± 3.58 years) and 904 without PCOS (29.1 ± 3.4 years; controls). Women with PCOS had a higher body mass index (22.8 ± 3.84 vs. 19.9 ± 2.23 kg/m2, p < 0.001), but most were not overweight/obese. Rates of positive TPO Ab in women with versus without PCOS were 8.2% and 8.4%, respectively (p = 0.932). Rates of positive TPO Ab in patients with PCOS phenotype A, B, C, or D were not statistically different (7.5%, 2.9%, 20.0%, and 7.8%, respectively). Median TSH concentrations were similar in the PCOS and control groups (1.84 mIU/L vs. 1.78 mIU/L, respectively; p = 0.194). Based on a linear regression model, there was no correlation between either BMI or the estradiol to progesterone ratio and TPO Ab status. In a large population of infertile women with PCOS who were mostly lean patients, rates of positive TPO Ab across all four PCOS phenotypes did not differ significantly from those in women without PCOS. These findings did not support the hypothesis that PCOS is a risk factor for Hashimoto thyroiditis.

Similar content being viewed by others
Data Availability
The datasets generated and/or analyzed during the current study are not publicly available due to patient privacy but are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Kennedy RL, Malabu UH, Jarrod G, Nigam P, Kannan K, Rane A. Thyroid function and pregnancy: before, during and beyond. J Obstet Gynaecol. 2010;30(8):774–83.
Poppe K, Glinoer D, Van Steirteghem A, Tournaye H, Devroey P, Schiettecatte J, et al. Thyroid dysfunction and autoimmunity in infertile women. Thyroid. 2002;12(11):997–1001.
Medenica S, Nedeljkovic O, Radojevic N, Stojkovic M, Trbojevic B, Pajovic B. Thyroid dysfunction and thyroid autoimmunity in euthyroid women in achieving fertility. Eur Rev Med Pharmacol Sci. 2015;19(6):977–87.
Lee HJ, Li CW, Hammerstad SS, Stefan M, Tomer Y. Immunogenetics of autoimmune thyroid diseases: a comprehensive review. J Autoimmun. 2015;64:82–90.
Feldthusen AD, Pedersen PL, Larsen J, ToftKristensen T, Ellervik C, Kvetny J. Impaired fertility associated with subclinical hypothyroidism and thyroid autoimmunity: the Danish General Suburban Population Study. J Pregnancy. 2015;2015:1–6.
Birjandi B, Ramezani Tehrani F, Amouzegar A, Tohidi M, BidhendiYarandi R, Azizi F. The association between subclinical hypothyroidism and TPOAb positivity with infertility in a population-based study: Tehran thyroid study (TTS). BMC Endocr Disord. 2021;21(1):108.
Yuan N, Sun J, Li Z, Chai S, Zhang X, Ji L. Relationship between anti-thyroid peroxidase antibody positivity and pregnancy-related and fetal outcomes in Euthyroid women: a single-center cohort study. BMC Pregnancy Childbirth. 2020;20(1):491.
Lizneva D, Suturina L, Walker W, Brakta S, Gavrilova-Jordan L, Azziz R. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. 2016;106(1):6–15.
Zhao Y, Fu L, Li R, Wang LN, Yang Y, Liu NN, et al. Metabolic profiles characterizing different phenotypes of polycystic ovary syndrome: plasma metabolomics analysis. BMC Med. 2012;10(1):153.
Guo M, Chen ZJ, Eijkemans MJE, Goverde AJ, Fauser BCJM, Macklon NS. Comparison of the phenotype of Chinese versus Dutch Caucasian women presenting with polycystic ovary syndrome and oligo/amenorrhoea. Hum Reprod. 2012;27(5):1481–8.
Romitti M, Fabris VC, Ziegelmann PK, Maia AL, Spritzer PM. Association between PCOS and autoimmune thyroid disease: a systematic review and meta-analysis. Endocr Connect. 2018;7(11):1158–67.
Kim JJ, Yoon JW, Kim MJ, Kim SM, Hwang KR, Choi YM. Thyroid autoimmunity markers in women with polycystic ovary syndrome and controls. Hum Fertil. 2022;25(1):128–34.
Adamska A, Łebkowska A, Krentowska A, Hryniewicka J, Adamski M, Leśniewska M, et al. Ovarian reserve and serum concentration of thyroid peroxidase antibodies in euthyroid women with different polycystic ovary syndrome phenotypes. Front Endocrinol. 2020;28(11):440.
Arduc A, AycicekDogan B, Bilmez S, ImgaNasiroglu N, Tuna MM, Isik S, et al. High prevalence of Hashimoto’s thyroiditis in patients with polycystic ovary syndrome: does the imbalance between estradiol and progesterone play a role? Endocr Res. 2015;40(4):204–10.
Janssen O, Mehlmauer N, Hahn S, Offner A, Gartner R. High prevalence of autoimmune thyroiditis in patients with polycystic ovary syndrome. Eur J Endocrinol. 2004;1:363–9.
Song Rh, Wang B, Yao Qm, Li Q, Jia X, Zhang Ja. The impact of obesity on thyroid autoimmunity and dysfunction: a systematic review and meta-analysis. Front Immunol. 2019;10:2349.
Matarese G, Sanna V, Giacomo AD, Lord GM, Howard JK, Bloom SR, et al. Leptin potentiates experimental autoimmune encephalomyelitis in SJL female mice and confers susceptibility to males. Eur J Immunol. 2001;31(5):1324–32.
Zhang HY, Guo CX, Zhu FF, Qu PP, Lin WJ, Xiong J. Clinical characteristics, metabolic features, and phenotype of Chinese women with polycystic ovary syndrome: a large-scale case–control study. Arch Gynecol Obstet. 2013;287(3):525–31.
Kim JJ, Choi YM. Phenotype and genotype of polycystic ovary syndrome in Asia: ethnic differences. J Obstet Gynaecol Res. 2019;45(12):2330–7.
Cai J, Zhang Y, Wang Y, Li S, Wang L, Zheng J, et al. High thyroid stimulating hormone level is associated with hyperandrogenism in euthyroid polycystic ovary syndrome (PCOS) women, independent of Age, BMI, and thyroid autoimmunity: a cross-sectional analysis. Front Endocrinol. 2019;10(10):222.
Acknowledgements
We are grateful to My Duc Hospital and HRC staffs for their contribution and supports in this study.
Author information
Authors and Affiliations
Contributions
The study was designed by LDL, VTTT, LNV, and TDP. Statistical analysis was performed by TDP and LTHT. The draft of this manuscript was first written by VTTT and LDL. The manuscript was reviewed and edited by all of the authors. VTTT, LDL, TDP, and LTHT had full access to all the data in the study. All authors had final responsibility for the decision to submission and assume responsibility for the accuracy and completeness of the analyses and the consistent adherence of this report to the research protocol.
Corresponding author
Ethics declarations
Ethics Approval
02/21/DD-BVMD.
Consent to Participate
Not applicable.
Consent for Publication
All the authors have consented to the publication.
Conflict of Interest
LNV has received grant, speaker, and conference fees from Merck Sharp and Dohme and grant, speaker, conference, and scientific board fees from Ferring. LDL has received speaker fees from Novo Nordisk. All other authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tran, V.T.T., Ly, L.D., Nguyen, M.H.N. et al. Thyroid Peroxidase Antibodies in Infertile Women with Polycystic Ovary Syndrome. Reprod. Sci. 30, 3071–3076 (2023). https://doi.org/10.1007/s43032-023-01261-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01261-5