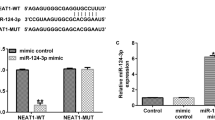
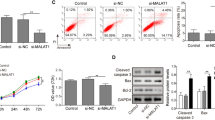
Abstract
Endometriosis (EMs) is a systemic and chronic disease with cancer-like feature, namely, distant implantation, which caused heavy healthy burden of nearly 200 million females. LncRNAs have been proved as new modulators in epithelial–mesenchymal transition (EMT) and EMs. Quantitative real-time PCR was conducted to measure the expression level of long intergenic non-protein coding RNA, regulator of reprogramming (Linc-ROR), and miR-204-5p in ectopic endometrium (n = 25), eutopic endometrium (n = 20), and natural control endometrium (n = 22). Overexpression of Linc-ROR, knockdown or overexpression of miR-204-5p in End1/E6E7 and Ishikawa cells, was conducted to detect the function of Linc-ROR and miR-204-5p in EMs. Furthermore, luciferase reports were used to confirm the combination of Linc-ROR and miR-204-5p and the combination between miR-204-5p and SMAD4. Cell-Counting Kit-8, EdU assay, transwell assays, and Western blotting were used to detect the function of Linc-ROR and miR-204-5p in EMs cancer-like behaviors and EMT process. Linc-ROR was up-regulated in ectopic endometrium. Overexpressed Linc-ROR promotes cell proliferation, invasion, and EMT process. Linc-ROR regulated the EMT process, cellular proliferation, and invasion of EMs via binding to miR-204-5p. In addition, overexpression of Linc-ROR up-regulated SMAD4, a target protein of miR-204-5p, with which regulated EMT process and cancer-like behaviors in EMs together. Linc-ROR/miR-204-5p/SMAD4 axis plays a vital role in regulation EMT process in EMs, which might become a novel therapeutic targets and powerful biomarkers in EMs therapy.





Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
Abbreviations
- EMs:
-
Endometriosis
- EMT:
-
Epithelial–mesenchymal transition
- ncRNA:
-
Non-coding RNA
- ISK:
-
Ishikawa
- E6E7:
-
End1/E6E7
- E-cad:
-
E-cadherin
- VIM:
-
Vimentin
- ceRNA:
-
Competing endogenous RNA
- LncRNA:
-
Long non-coding RNA
- PBS:
-
Phosphate-buffered saline
- CCK-8:
-
Cell counting kit-8
- qRT-PCR:
-
Quantitative reverse-transcription polymerase chain reaction
- IHC:
-
Immunohistochemistry
- EC:
-
Ectopic endometrium
- EU:
-
Eutopic endometrium
- NC:
-
Endometrium from the control group
- R-SMAD:
-
Receptor-regulated SMAD
References
Eisenberg VH, Weil C, Chodick G, Shalev V. Epidemiology of endometriosis: a large population-based database study from a healthcare provider with 2 million members. BJOG. 2018;125:55–62.
de Ziegler D, Borghese B, Chapron C. Endometriosis and infertility: pathophysiology and management. Lancet. 2010;376:730–8.
Bulun SE, Yilmaz BD, Sison C, Miyazaki K, Bernardi L, Liu S, Kohlmeier A, Yin P, Milad M, Wei J. Endometriosis. Endocr Rev. 2019;40:1048–79.
Taylor HS, Giudice LC, Lessey BA, Abrao MS, Kotarski J, Archer DF, Diamond MP, Surrey E, Johnson NP, Watts NB, Gallagher JC, Simon JA, Carr BR, Dmowski WP, Leyland N, Rowan JP, Duan WR, Ng J, Schwefel B, Thomas JW, Jain RI, Chwalisz K. Treatment of Endometriosis-Associated Pain with Elagolix, an Oral GnRH Antagonist. New Engl J Med. 2017;377:28–40.
Chapron C, Marcellin L, Borghese B, Santulli P. Rethinking mechanisms, diagnosis and management of endometriosis. Nat Rev Endocrinol. 2019;15:666–82.
Guo SW. Cancer-associated mutations in endometriosis: shedding light on the pathogenesis and pathophysiology. Hum Reprod Update. 2020;26:423–49.
Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol. 2019;20(2):69–84.
Bilyk O, Coatham M, Jewer M, Postovit LM. Epithelial-to-Mesenchymal Transition in the Female Reproductive Tract: From Normal Functioning to Disease Pathology. Front Oncol. 2017;7:145.
Saunders PTK, Horne AW. Endometriosis: Etiology, pathobiology, and therapeutic prospects. Cell. 2021;184:2807–24.
Tay Y, Rinn J, Pandolfi PP. The multilayered complexity of ceRNA crosstalk and competition. Nature. 2014;505:344–52.
Thomson DW, Dinger ME. Endogenous microRNA sponges: evidence and controversy. Nat Rev Genet. 2016;17(5):272–83.
Abba ML, Patil N, Leupold JH, Moniuszko M, Utikal J, Niklinski J, Allgayer H. MicroRNAs as novel targets and tools in cancer therapy. Cancer Lett. 2017;387:84–94.
Zondervan KT, Becker CM, Koga K, Missmer SA, Taylor RN, Vigano P. Endometriosis. Nat Rev Dis Primers. 2018;4:9.
Laganà AS, Garzon S, Götte M, Viganò P, Franchi M, Ghezzi F, Martin DC. The pathogenesis of endometriosis: molecular and cell biology insights. Int J Mol Sci. 2019;20(22):5615.
Wang X, Zhang J, Liu X, Wei B, Zhan L. Long noncoding RNAs in endometriosis: Biological functions, expressions, and mechanisms. J Cell Physiol. 2021;236:6–14.
Lin D, Huang Q, Wu R, Dai S, Huang Z, Ren L, Huang S, Chen Q. Long non-coding RNA AFAP1-AS1 promoting epithelial-mesenchymal transition of endometriosis is correlated with transcription factor ZEB1. Am J Reprod Immunol. 2019;81(1):e13074.
Mai H, Wei Y, Yin Y, Huang S, Lin H, Liao Y, Liu X, Chen X, Shi H, Liu C, Xu H. LINC01541 overexpression attenuates the 17 beta-Estradiol-induced migration and invasion capabilities of endometrial stromal cells. Syst Biol Reprod Med. 2019;65:214–22.
Liu H, He H, Zhang Z, Wang L, Zhang L, Liu Y, Xiong W. Upregulation of the long noncoding RNA UBOX5 antisense RNA 1 (UBOX5-AS1) under hypoxic conditions promotes epithelial-mesenchymal transition in endometriosis. Ann Transl Med. 2021;9(9):790.
Liang Z, Chen Y, Zhao Y, Xu C, Zhang A, Zhang Q, Wang D, He J, Hua W, Duan P. miR-200c suppresses endometriosis by targeting MALAT1 in vitro and in vivo. Stem Cell Res Ther. 2017;8:251.
Du Y, Zhang Z, Xiong W, Li N, Liu H, He H, Li Q, Liu Y, Zhang L. Estradiol promotes EMT in endometriosis via MALAT1/miR200s sponge function. Reproduction. 2019;157:179–88.
Wang S, Yi M, Zhang X, Zhang T, Jiang L, Cao L, Zhou Y, Fang X. Effects of CDKN2B-AS1 on cellular proliferation, invasion and AKT3 expression are attenuated by miR-424-5p in a model of ovarian endometriosis. Reprod Biomed Online. 2021;42:1057–66.
Taylor HS, Kotlyar AM, Flores VA. Endometriosis is a chronic systemic disease: clinical challenges and novel innovations. Lancet. 2021;397:839–52.
Wang S, Zhang M, Zhang T, Deng J, Xia X, Fang X. microRNA-141 inhibits TGF-β1-induced epithelial-to-mesenchymal transition through inhibition of the TGF-β1/SMAD2 signalling pathway in endometriosis. Arch Gynecol Obstet. 2020;301:707–14.
Derynck R, Zhang YE. Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature. 2003;425:577–84.
Derynck R, Budi EH. Specificity, versatility, and control of TGF-beta family signaling. Sci Signal. 2019;12(570):eaav5183.
Azar R, Alard A, Susini C, Bousquet C, Pyronnet S. 4E-BP1 is a target of Smad4 essential for TGFbeta-mediated inhibition of cell proliferation. EMBO J. 2009;28:3514–22.
Zhang J, Guan W, Xu X, Wang F, Li X, Xu G. A novel homeostatic loop of sorcin drives paclitaxel-resistance and malignant progression via Smad4/ZEB1/miR-142-5p in human ovarian cancer. Oncogene. 2021;40:4906–18.
Ping S, Ma C, Liu P, Yang L, Yang X, Wu Q, Zhao X, Gong B. Molecular mechanisms underlying endometriosis pathogenesis revealed by bioinformatics analysis of microarray data. Arch Gynecol Obstet. 2016;293:797–804.
Xu XY, Zhang J, Qi YH, Kong M, Liu SA, Hu JJ. Linc-ROR promotes endometrial cell proliferation by activating the PI3K-Akt pathway. Eur Rev Med Pharmacol Sci. 2018;22:2218–25.
Inoue S, Hirota Y, Ueno T, Fukui Y, Yoshida E, Hayashi T, Kojima S, Takeyama R, Hashimoto T, Kiyono T, Ikemura M, Taguchi A, Tanaka T, Tanaka Y, Sakata S, Takeuchi K, Muraoka A, Osuka S, Saito T, Oda K, Osuga Y, Terao Y, Kawazu M, Mano H. Uterine adenomyosis is an oligoclonal disorder associated with KRAS mutations. Nat Commun. 2019;10:5785.
Gao H, Wang T, Zhang P, Shang M, Gao Z, Yang F, Liu R. Linc-ROR regulates apoptosis in esophageal squamous cell carcinoma via modulation of p53 ubiquitination by targeting miR-204-5p/MDM2. J Cell Physiol. 2020;235:2325–35.
Zhi Y, Abudoureyimu M, Zhou H, Wang T, Feng B, Wang R, Chu X. FOXM1-Mediated LINC-ROR Regulates the Proliferation and Sensitivity to Sorafenib in Hepatocellular Carcinoma. Mol Ther Nucleic Acids. 2019;16:576–88.
Zhan HX, Wang Y, Li C, Xu JW, Zhou B, Zhu JK, Han HF, Wang L, Wang YS, Hu SY. LincRNA-ROR promotes invasion, metastasis and tumor growth in pancreatic cancer through activating ZEB1 pathway. Cancer Lett. 2016;374:261–71.
Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67:817–821.
Xu J, Zhu X, Wu L, Yang R, Yang Z, Wang Q, Wu F. MicroRNA-122 suppresses cell proliferation and induces cell apoptosis in hepatocellular carcinoma by directly targeting Wnt/β-catenin pathway. Liver Int. 2012;32:752–60.
Sun Y, Li Q, Gui H, Xu DP, Yang YL, Su DF, Liu X. MicroRNA-124 mediates the cholinergic anti-inflammatory action through inhibiting the production of pro-inflammatory cytokines. Cell Res. 2013;23:1270–83.
Matsuzaki S, Pouly JL, Canis M. In vitro and in vivo effects of MK2206 and chloroquine combination therapy on endometriosis: autophagy may be required for regrowth of endometriosis. Br J Pharmacol. 2018;175:1637–53.
Bayoglu Tekin Y, Guven S, Kirbas A, Kalkan Y, Tumkaya L, Guvendag Guven ES. Is resveratrol a potential substitute for leuprolide acetate in experimental endometriosis? Eur J Obstet Gynecol Reprod Biol. 2015;184:1–6.
Chen J, Zhong Y, Li L. miR-124 and miR-203 synergistically inactivate EMT pathway via coregulation of ZEB2 in clear cell renal cell carcinoma (ccRCC). J Transl Med. 2020;18:69.
Wang JR, Liu B, Zhou L, Huang YX. MicroRNA-124-3p suppresses cell migration and invasion by targeting ITGA3 signaling in bladder cancer. Cancer Biomark. 2019;24:159–72.
Luo L, Chi H, Ling J. MiR-124–3p suppresses glioma aggressiveness via targeting of Fra-2. Pathol Res Pract. 2018;214:1825–1834.
Zeng T, Luo L, Huang Y, Ye X, Lin J. Upregulation of miR-138 Increases Sensitivity to Cisplatin in Hepatocellular Carcinoma by Regulating EZH2. Biomed Res Int. 2021;2021:6665918.
Zhu D, Gu L, Li Z, Jin W, Lu Q, Ren T. MiR-138–5p suppresses lung adenocarcinoma cell epithelial-mesenchymal transition, proliferation and metastasis by targeting ZEB2. Pathol Res Pract. 2019;215:861–872.
Zhang F, Li T, Han L, Qin P, Wu Z, Xu B, Gao Q, Song Y. TGFβ1-induced down-regulation of microRNA-138 contributes to epithelial-mesenchymal transition in primary lung cancer cells. Biochem Biophys Res Commun. 2018;496:1169–75.
Li D, He C, Wang J, Wang Y, Bu J, Kong X, Sun D. MicroRNA-138 Inhibits Cell Growth, Invasion, and EMT of Non-Small Cell Lung Cancer via SOX4/p53 Feedback Loop. Oncol Res. 2018;26:385–400.
Si H, Chen P, Li H, Wang X. Long non-coding RNA H19 regulates cell growth and metastasis via miR-138 in breast cancer. Am J Transl Res. 2019;11:3213–25.
Zhao C, Ling X, Li X, Hou X, Zhao D. MicroRNA-138-5p inhibits cell migration, invasion and EMT in breast cancer by directly targeting RHBDD1. Breast Cancer. 2019;26:817–25.
Tang N, Dong Y, Liu J, Zhao H. Silencing of Long Non-coding RNA NEAT1 Upregulates miR-195a to Attenuate Intervertebral Disk Degeneration via the BAX/BAK Pathway. Front Mol Biosci. 2020;7:147.
Bai J, Xu J, Zhao J, Zhang R. lncRNA SNHG1 cooperated with miR-497/miR-195-5p to modify epithelial-mesenchymal transition underlying colorectal cancer exacerbation. J Cell Physiol. 2020;235:1453–68.
Zhao X, Dai L, Yue Q, Wang H, Wang XU, Li Y, Chen R. MiR-195 inhibits migration, invasion and epithelial-mesenchymal transition (EMT) of endometrial carcinoma cells by targeting SOX4. J Biosci (Bangalore). 2019:44(6):146.
Liang M, Huang G, Liu Z, Wang Q, Yu Z, Liu Z, Lin H, Li M, Zhou X, Zheng Y. Elevated levels of hsa_circ_006100 in gastric cancer promote cell growth and metastasis via miR-195/GPRC5A signalling. Cell Prolif. 2019;52: e12661.
Zhao L, Tian C, Xiao E, Du J, Liang J, Chen X, Chi W. Clinical significance and potential mechanisms of miR-223-3p and miR-204-5p in squamous cell carcinoma of head and neck: a study based on TCGA and GEO. Open Med (Wars). 2020;15:728–38.
Wang H, Qian J, Xia X, Ye B. Long non-coding RNA OIP5-AS1 serves as an oncogene in laryngeal squamous cell carcinoma by regulating miR-204-5p/ZEB1 axis. Naunyn-Schmiedeberg’s Arch Pharmacol. 2020;393:2177–84.
Sui H, Fan S, Liu W, Li Y, Zhang X, Du Y, Bao H. LINC00028 regulates the development of TGFbeta1-treated human tenon capsule fibroblasts by targeting miR-204–5p. Biochem Biophys Res Commun. 2020;525(1):197–203.
Liang C, Yang Y, Guan J, Lv T, Qu S, Fu Q, Zhao H. LncRNA UCA1 sponges miR-204–5p to promote migration, invasion and epithelial-mesenchymal transition of glioma cells via upregulation of ZEB1. Pathol Res Pract. 2018;214:1474–1481.
Huang J, Wang X, Wen G, Ren Y. miRNA-205-5p functions as a tumor suppressor by negatively regulating VEGFA and PI3K/Akt/mTOR signaling in renal carcinoma cells. Oncol Rep 2019;42(5):1677–88.
Lu J, Lin Y, Li F, Ye H, Zhou R, Jin Y, Li B, Xiong X, Cheng N. MiR-205 suppresses tumor growth, invasion, and epithelial-mesenchymal transition by targeting SEMA4C in hepatocellular carcinoma. FASEB J. 2018;32(11):6123–34.
Dai B, Zhou G, Hu Z, Zhu G, Mao B, Su H, Jia Q. MiR-205 suppresses epithelial-mesenchymal transition and inhibits tumor growth of human glioma through down-regulation of HOXD9. Biosci Rep. 2019;39(5):BSR20181989.
Vosgha H, Ariana A, Smith RA, Lam AK. miR-205 targets angiogenesis and EMT concurrently in anaplastic thyroid carcinoma. Endocr Relat Cancer. 2018;25:323–37.
Chen W, Kong KK, Xu XK, Chen C, Li H, Wang FY, Peng XF, Zhang Z, Li P, Li JL, Li FC. Downregulation of miR205 is associated with glioblastoma cell migration, invasion, and the epithelial-mesenchymal transition, by targeting ZEB1 via the Akt/mTOR signaling pathway. Int J Oncol. 2018;52:485–95.
Gulei D, Magdo L, Jurj A, Raduly L, Cojocneanu-Petric R, Moldovan A, Moldovan C, Florea A, Pasca S, Pop LA, Moisoiu V, Budisan L, Pop-Bica C, Ciocan C, Buiga R, Muresan MS, Stiufiuc R, Ionescu C, Berindan-Neagoe I. The silent healer: miR-205-5p up-regulation inhibits epithelial to mesenchymal transition in colon cancer cells by indirectly up-regulating E-cadherin expression. Cell Death Dis. 2018;9:66.
Standart N, Jackson RJ. MicroRNAs repress translation of m7G ppp-capped target mRNAs in vitro by inhibiting initiation and promoting deadenylation. Genes Dev. 2007;21:1975–82.
Vasudevan S, Tong Y, Steitz JA. Switching from repression to activation: microRNAs can up-regulate translation. Science. 2007;318:1931–4.
Eiring AM, Harb JG, Neviani P, Garton C, Oaks JJ, Spizzo R, Liu S, Schwind S, Santhanam R, Hickey CJ, Becker H, Chandler JC, Andino R, Cortes J, Hokland P, Huettner CS, Bhatia R, Roy DC, Liebhaber SA, Caligiuri MA, Marcucci G, Garzon R, Croce CM, Calin GA, Perrotti D. miR-328 functions as an RNA decoy to modulate hnRNP E2 regulation of mRNA translation in leukemic blasts. Cell. 2010;140:652–65.
Weis-Banke SE, Lerdrup M, Kleine-Kohlbrecher D, Mohammad F, Sidoli S, Jensen ON, Yanase T, Nakamura T, Iwase A, Stylianou A, Abu-Rustum NR, Aghajanian C, Soslow R, Da Cruz Paula A, Koche RP, Weigelt B, Christensen J, Helin K, Cloos PAC. Mutant FOXL2(C134W) Hijacks SMAD4 and SMAD2/3 to Drive Adult Granulosa Cell Tumors. Cancer Res. 2020;80:3466–79.
Peng C, Wang Y, Ji L, Kuang L, Yu Z, Li H, Zhang J, Zhao J. LncRNA-MALAT1/miRNA-204–5p/Smad4 axis regulates epithelial-mesenchymal transition, proliferation and migration of lens epithelial cells. Curr Eye Res. 2021;46(8):1137–47.
Qu J, Zhu Y, Wu X, Zheng J, Hou Z, Cui Y, Mao Y, Liu J. Smad3/4 Binding to Promoter II of P450arom So As to Regulate Aromatase Expression in Endometriosis. Reprod Sci. 2017;24:1187–94.
Zheng J, Qu J, Lu P, Hou Z, Cui Y, Mao Y, Qi X, Ji H, Liu J. Activin A Stimulates Aromatase via the ALK4-Smad Pathway in Endometriosis. Biomed Res Int. 2016;2016:5791510.
Acknowledgements
The authors thank AiMi Academic Services (www.aimieditor.com) for the English language editing and review services.
Funding
This work was supported by the National Natural Science Foundation of China (81671437, 81771558) and the Natural Science Foundation of Hunan Province, China (2016JC2049, 2020JJ4814).
Author information
Authors and Affiliations
Contributions
Mingyu Yi: Data curation, methodology, software, writing—original draft preparation. Sixue Wang: Data curation, Writing—Original draft preparation. Xinyue Zhang: Data curation, writing—original draft preparation. Li Jiang: Resources, Investigation, validation. Xiaomeng Xia: Resources, investigation, validation. Tingting Zhang: Resources, investigation, validation. Xiaoling Fang: Supervision, writing—reviewing and editing, funding acquisition.
Corresponding author
Ethics declarations
Ethics Approval
This study was approved by the Institutional Ethics Review Committee of Second Xiangya Hospital of Central South University (#2016243).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Fig. S1
Validation of transfection plasmid Linc-ROR, miR-204-5p mimic and miR-204-5p Inhibitor. a Plasmid map of Linc-ROR. b 1 μg plasmid Linc-ROR and vector was transfected into End1/E6E7 and Ishikawa cells respectively, and qRT-PCR was conducted to measure the relatively expression of Linc-ROR. Two-tailed Student’s t-test was conducted for statistical analysis. c 50 nM miR-204-5p mimic or mimic NC was transfected into End1/E6E7 and Ishikawa cells, 80 nM miR-204-5p Inhibitor or Inhibitor NC was transfected into Ishikawa cells, and 30 nM miR-204-5p Inhibitor or Inhibitor NC was transfected into End1/E6E7 cells, the transfection was confirmed using qRT-PCR. Two-tailed Student’s t-test was conducted for statistical analysis. E6E7, End1/E6E7; ISK, Ishikawa; mimic, miR-204-5p mimic; inhibitor, miR-204-5p Inhibitor; NC, natural control. (PNG 214 kb)

Fig. S2
Expression of miR-124-3p, miR-138-5p, miR-195-5p and miR-205-5p, and their regulation by Linc-ROR. a-d Left panel shows expression levels of miR-124-3p, miR-138-5p, miR-195-5p and miR-205-5p in ectopic endometrium (EC, n = 10), eutopic endometrium (EU, n = 10), and control group (NC, n = 10) by qRT-PCR. Kruskal–Wallis test followed by pot-hoc Dunn’s test was conducted for statistical analysis. Right panel shows expression of miR-124-3p, miR-138-5p, miR-195-5p and miR-205-5p in End1/E6E7 and Ishikawa cells transfected with Linc-ROR plasmid or control vector using qRT-PCR. Two-tailed Student’s t-test was conducted for statistical analysis. E6E7, End1/E6E7; ISK, Ishikawa. (PNG 236 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yi, M., Wang, S., Zhang, X. et al. Linc-ROR Promotes EMT by Targeting miR-204-5p/SMAD4 in Endometriosis. Reprod. Sci. 30, 2665–2679 (2023). https://doi.org/10.1007/s43032-023-01204-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01204-0