Abstract
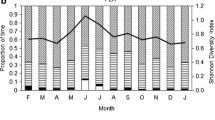
The thin-spined porcupine (Chaetomys subspinosus Olfers, 1818) is an endemic and threatened rodent from the Atlantic Forest biological hotspot. Previous studies have demonstrated it follows a strictly leaf-based diet, limited to a few tree species, although such information is derived from few individuals (n = 7) resident in small forest fragments. We aimed to evaluate whether such dietary specialization persists when animals inhabit larger forest fragments. For this, we assessed the diet composition of 19 radiotracked individuals inhabiting small (< 50 ha; n = 10) and large (> 500 ha; n = 9) forest fragments in southern Bahia state. We compared the composition and diversity of the diet in terms of tree parts and species consumed in these contrasting fragment size environments. Secondly, we aimed to evaluate the influence of leaf chemical composition on the consumption of plant species. Our findings show that the thin-spined porcupine is a strict folivore, with individuals responding to the reduction in forest size by reducing the diversity of plant species consumed, but not by feeding on new plant parts. Although the diet was richer in larger forest fragments, certain tree species were the most consumed in both fragment-size categories and the unique item consumed were leaves. Fiber influenced positively the leaf consumption of the plant species, while there was no effect of other chemical characteristics. The influence of the habitat size reduction on food diversity may be a risk factor for the species.


Similar content being viewed by others
References
Adams JM, Rehill B, Zhang Y, Gower J (2009) A test of the latitudinal defense hypothesis: herbivory, tannins and total phenolics in four North American tree species. Ecol Res 24:697–704. https://doi.org/10.1007/s11284-008-0541-x
Altmann J, Loy J, Wagner S (1974) Observational study of behavior: sampling methods. Behaviour 49:227–266. https://doi.org/10.1163/156853974x00534
AOAC (2005) Official methods of analysis of AOAC. AOAC International, Gaithersburg
Benchimol M, Mariano-Neto E, Faria D, Rocha-Santos L, Pessoa MS, Gomes FS, Talora DC, Cazetta E (2017) Translating plant community responses to habitat loss into conservation practices: forest cover matters. Biol Conserv 209:499–507. https://doi.org/10.1016/j.biocon.2017.03.024
Bonvicino, C.R., D’Andrea, P.S., Bezerra, A.M.R., Percequillo, A., Portella, A., Christoff, A.U., Almeida, A.M., Carmignotto, A.P., Silva, C.R., Raices D.S.L., Medeiros, L., Hingst-Zaher, E., Fernandes, F.A., Ximenes, G.E., Lessa, G., Moreira, J., de Oliveira, J.A., Cherem, J., Tiepolo, L.M., Reis, M.L., Weksler, M., Alvarez, M.R., Faria, M.B., Gonçalves, P.R., Peres, P.H.A.L., Paresque, R., Vilela, R.V., de Freitas, T.O., Leite, Y.R., 2018. Chaetomys subspinosus, in: Instituto Chico Mendes de Conservação da Biodiversidade (Org.), Livro Vermelho da Fauna Brasileira Ameaçada de Extinção: Volume II. Mamíferos, ICMBio, Brasília, pp. 459–462. https://www.icmbio.gov.br/portal/images/stories/comunicacao/publicacoes/publicacoes-diversas/livro_vermelho_2018_vol1.pdf
Burnham KP, Anderson DR (2002) Model selection and inference: a practical information-theoretic approach, 2nd edn. Springer-Verlag, New York
Catzeflis F, Patton J, Percequillo A, Bonvicino C, Weksler M, 2017 Chaetomys subspinosus. https://www.iucnredlist.org/species/4366/22213335. (Accessed on 20 June 2018) [WWW Document]. IUCN Red List Threat. Species.
Charles-Dominique P, Atramentowicz M, Charles-Dominique M, Gérard H, Hladik A, Hladik CM, Prévost MF (1981) Les mammiferes frugivores arboricoles nocturnes d´une forest guyanaise: inter-relations plantes-animaux. Rev. d’écologie. La terre la vie Paris 35:341–435
Chiarello AG, Passamani M, Zortéa M (1997) Field observations on the thin-spined porcupine, Chaetomys subspinosus (Rodentia; Echimyidae). Mammalia 61:29–36. https://doi.org/10.1515/mamm.1997.61.1.29
Collins L, Roberts M (1978) Arboreal folivores in captivity—Maintenance of a delicate minority. In: Montgomery GG (ed) The ecology of arboreal folivores. Smithsonian Institution Press, Washington, D.C., pp 5–12
Coltrane JA (2012) Redefining the north American porcupine (Erethizon dorsatum) as a facultative specialist herbivore. Northwest Nat 93:187–193. https://doi.org/10.1898/12-04.1
Coltrane JA, Barboza PS (2010) Winter as a nutritional bottleneck for North American porcupines (Erethizon dorsatum). J. Comp Physiol B Biochem Syst Environ Physiol 180:905–918. https://doi.org/10.1007/s00360-010-0460-3
Cristóbal-Azkarate J, Arroyo-Rodríguez V (2007) Diet and activity pattern of howler monkeys (Alouatta palliata) in Los Tuxtlas, Mexico: effects of habitat fragmentation and implications for conservation. Am J Primatol 69:1013–1029. https://doi.org/10.1002/ajp.20420
de Abreu TCK, da Rosa CA, Aximoff I (2016) New record of feeding behavior by the porcupine Coendou spinosus (F. Cuvier, 1823) in high-altitude grassland of the Brazilian Atlantic Forest. Mammalia. https://doi.org/10.1515/mammalia-2016-0026
de Souto Lima RB, Oliveira PA, Chiarello AG (2010) Diet of the thin-spined porcupine (Chaetomys subspinosus), an Atlantic forest endemic threatened with extinction in southeastern Brazil. Mamm Biol 75:538–546. https://doi.org/10.1016/j.mambio.2009.09.002
Eisenberg JF (1978) The evolution of arboreal herbivores in the class Mammalia. In: Montgomery GG (ed) The ecology of arboreal folivores. Smithsonian Institution Press, Washington, D.C., pp 135–152
Felicetti LA, Shipley LA, Witmer GW, Robbins CT (2000) Digestibility, nitrogen excretion, and mean retention time by North American porcupines (Erethizon dorsatum) consuming natural forages. Physiol Biochem Zool 73:772–780. https://doi.org/10.1086/318094
Foley WJ, Cork SJ (1992) Use of fibrous diets by small herbivores: how far can the rules be ‘bent’? Tree 7:159–162
Fournier F, Thomas DW (1997) Nitrogen and energy requirements of the North American porcupine (Erethizon dorsatum). Physiol Zool 70:615–620. https://doi.org/10.1086/515874
Freeland WJ, Janzen DH (1974) Strategies in herbivory by mammals: the role of plant secondary compounds. Am Nat 108:269–289. https://doi.org/10.1086/282907
Ganzhorn JU (1995) Low-level forest disturbance effects on primary production, leaf chemistry, and lemur populations. Ecology 76:2084–2096. https://doi.org/10.2307/1941683
Ganzhorn JU, Arrigo- Nelson SJ, Carrai V, Chalise MK, Donati G, Droescher I, Eppley TM, Irwin MT, Koch F, Koenig A, Kowalewski MM, Mowry CB, Patel ER, Pichon C, Ralison J, Reisdorff C, Simmen B, Stalenberg E, Starrs D, Terboven J, Wright PC, Foley WJ (2017) The importance of protein in leaf selection of folivorous primates. Am J Primatol 79:1–13. https://doi.org/10.1002/ajp.22550
Giné GAF, Faria D (2018) Combining species distribution modeling and field surveys to reappraise the geographic distribution and conservation status of the threatened thin-spined porcupine (chaetomys subspinosus). PLoS ONE 13:1–22. https://doi.org/10.1371/journal.pone.0207914
Giné GAF, Duarte JMB, Faria D (2010) Feeding ecology of a selective folivore, the thin-spined porcupine (chaetomys subspinosus) in the Atlantic forest. J Mammal 91:931–941. https://doi.org/10.1644/09-mamm-a-185.1
Giné GAF, De Barros EH, Duarte JMB, Faria D (2015) Home range and multiscale habitat selection of threatened thin-spined porcupine in the Brazilian Atlantic forest. J Mammal 96:1095–1105. https://doi.org/10.1093/jmammal/gyv117
Hutcheson K (1970) A test for comparing diversities based on the Shannon formula. J Theor Biol 29:151–154. https://doi.org/10.1016/0022-5193(70)90124-4
Iason GR, Villalba JJ (2006) Behavioral strategies of mammal herbivores against plant secondary metabolites: the avoidance-tolerance continuum. J Chem Ecol 32:1115–1132. https://doi.org/10.1007/s10886-006-9075-2
Jensen LM, Wallis IR, Foley WJ (2015) The relative concentrations of nutrients and toxins dictate feeding by a vertebrate browser, the greater glider Petauroides volans. PLoS ONE 10:1–12. https://doi.org/10.1371/journal.pone.0121584
Kuniy AA (2005) Análise do conteúdo estomacal e intestinal de dois espécimes de ouriço-preto (Chaetomys subspinosus, olfers, 1818). Acta Biol Lepondensia 27:187–189
Kursar TA, Coley PD (2003) Convergence in defense syndromes of young leaves in tropical rainforests. Biochem Syst Ecol 31:929–949. https://doi.org/10.1016/S0305-1978(03)00087-5
Magurran AE (2004) Measuring biological diversity, 1st edn. Blackwell Publishing Ltd, Oxford, UK
Makkar HPS, Blümmel M, Borowy NK, Becker K (1993) Gravimetric determination of tannins and their correlations with chemical and protein precipitation methods. J Sci Food Agric 61:161–165. https://doi.org/10.1002/jsfa.2740610205
Mekonnen A, Fashing PJ, Bekele A, Hernandez-Aguilar RA, Rueness EK, Stenseth NC (2018) Dietary flexibility of Bale monkeys (Chlorocebus djamdjamensis) in southern Ethiopia: effects of habitat degradation and life in fragments. BMC Ecol 18:1–20. https://doi.org/10.1186/s12898-018-0161-4
Milton K (1979) Factors influencing leaf choice by howler monkeys: a test of some hypotheses of food selection by generalist herbivores. Am Nat 114:362–378. https://doi.org/10.1086/283485
Moojen J (1952) Os roedores do Brasil. Instituto Nacional do Livro, Rio de Janeiro, Brasil
Morante-Filho JC, Arroyo-Rodríguez V, Pessoa MS, Cazetta E, Faria D (2018) Direct and cascading effects of landscape structure on tropical forest and non-forest frugivorous birds. Ecol Appl 28:2024–2032. https://doi.org/10.1002/eap.1791
Mori SA, Boom BM, de Carvalho AM, dos Santos TS (1983) Southern Bahian moist forests. Bot Rev 49:155–232. https://doi.org/10.1007/BF02861011
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, Mcglinn D, Minchin PR, Hara RBO, Simpson GL, Solymos P, Stevens MHH, Szoecs E (2019) vegan: community ecology package. R package version 2.5–6. Available from https://cran.r-project.org/web/packages/vegan/index.html
Parra R (1978) Comparison of foregut and hindgut fermentation in herbivores. In: Montgomery GG (ed) The ecology of arboreal folivores. Smithsonian Institution Press, Washington, D.C., pp 205–230
Passamani M (2010) Use of space and activity pattern of Sphiggurus villosus (F. Cuvier, 1823) from Brazil (Rodentia: Erethizontidae). Mamm Biol 75:455–458. https://doi.org/10.1016/j.mambio.2009.04.005
Pessoa MS, Hambuckers A, Benchimol M, Rocha-Santos L, Bomfim JA, Faria D, Cazetta E (2017a) Deforestation drives functional diversity and fruit quality changes in a tropical tree assemblage. Perspect Plant Ecol Evol Syst 28:78–86. https://doi.org/10.1016/j.ppees.2017.09.001
Pessoa MS, Rocha-Santos L, Talora DC, Faria D, Mariano-Neto E, Hambuckers A, Cazetta E (2017b) Fruit biomass availability along a forest cover gradient. Biotropica 49:45–55. https://doi.org/10.1111/btp.12359
Porter LJ, Hrstich LN, Chan BG (1986) The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 25:223–230. https://doi.org/10.1016/S0031-9422(00)94533-3
R Core Team, 2018. R: a language and environment for statistical computing [WWW Document]. Version 3.0.0. R Found. Stat. Comput. Vienna, Austria.
Ribeiro MC, Metzger JP, Martensen AC, Ponzoni FJ, Hirota MM (2009) The Brazilian Atlantic Forest: how much is left, and how is the remaining forest distributed? Implications for conservation. Biol Conserv 142:1141–1153. https://doi.org/10.1016/j.biocon.2009.02.021
Ripley, B., Venables, B., Bates, D.M., Hornik, K., Gebhardt, A., Firth, D., 2019. MASS: support functions and datasets for venables and Ripley’s MASS. R package version 7.3–51.5. Available from https://cran.r-project.org/web/packages/MASS/index.html
Rivera A, Calmé S (2006) Forest fragmentation and its effects on the feeding ecology of black howlers (Alouatta pigra) from the Calakmul area in Mexico. In: Estrada A, Garber PA, Pavelka MSM (eds) New perspectives in the study of mesoamerican primates. Springer, New York, Boston, pp 189–213
Rocha-Santos L, Pessoa MS, Cassano CR, Talora DC, Orihuela RLL, Mariano-Neto E, Morante-Filho JC, Faria D, Cazetta E (2016) The shrinkage of a forest: landscape-scale deforestation leading to overall changes in local forest structure. Biol Conserv 196:1–9. https://doi.org/10.1016/j.biocon.2016.01.028
Rocha-Santos L, Benchimol M, Mayfield MM, Faria D, Pessoa MS, Talora DC, Mariano-Neto E, Cazetta E (2017) Functional decay in tree community within tropical fragmented landscapes: effects of landscape-scale forest cover. PLoS ONE 12:1–18. https://doi.org/10.1371/journal.pone.0175545
Roze U (1984) Winter foraging by individual porcupines. Can J Zool 62:2425–2428
Roze U (2012) Porcupines: the animal answer guide. John Hopkins University Press, Baltimore, Maryland, USA
Sadee C, Pietrzak M, Seweryn M, Wang C, Rempala G (2019) divo: tools for analysis of diversity and similarity in biological systems. R package version 1.0.1. Available from https://cran.r-project.org/web/packages/divo/index.html
Shannon CE (1948) The Bell system technical journal. Bell Syst Tech J 27:379–423. https://doi.org/10.1016/s0016-0032(23)90506-5
Shipley LA, Forbey JS, Moore BD (2009) Revisiting the dietary niche: when is a mammalian herbivore a specialist. Integr Comp Biol 49:274–290. https://doi.org/10.1093/icb/icp051
Sikes RS, Gannon WL (2011) Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J Mammal 92:235–253. https://doi.org/10.1644/10-mamm-f-355.1
Tabarelli M, Aguiar AV, Girão LC, Peres CA, Lopes AV (2010) Effects of pioneer tree species hyperabundance on forest fragments in Northeastern Brazil. Conserv Biol 24:1654–1663. https://doi.org/10.1111/j.1523-1739.2010.01529.x
Tenneson C, Oring LW (1985) Winter food preferences of porcupines. J Wildl Manage 49:28–33
Thomas W, Carvalho A, Amorim A, Garrison J, Arbeláez A (1998) Plant endemism in two forests in southern Bahia. Brazil Biodivers Conserv 7:311–322. https://doi.org/10.1023/A:1008825627656
Van Soest PJ, Robertson JB, Lewis BA (1991) Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci 74:3583–3597. https://doi.org/10.3168/jds.S0022-0302(91)78551-2
Vilela RV, Machado T, Ventura K, Fagundes V, De J Silva MJ, Yonenaga-Yassuda Y (2009) The taxonomic status of the endangered thin-spined porcupine, Chaetomys subspinosus (Olfers, 1818), based on molecular and karyologic data. BMC Evol Biol 9:1–17. https://doi.org/10.1186/1471-2148-9-29
Vispo C, Hume ID (1995) The digestive tract and digestive function in the North American porcupine and beaver. Can J Zool 73:967–974. https://doi.org/10.1139/z95-113
Westoby M (1978) What are the biological bases of varied diets? Am Nat 112:627–631
Acknowledgements
We thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [Project Rede SISBIOTA 563216/2010-7; Project INCT, IN-Tree 465767/2014-1] and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) [Project CASADINHO/PROCAD 552198/2011-0] for financial support and fellowships for K. F. M. da Silva. We are grateful to Rubens Vieira Lopes and Marcos Alves for field assistance as well as Universidade Estadual of Santa Cruz for logistical support. We are grateful to José Lima da Paixão and Larissa Rocha Santos for identifying and classifying plant species. This research received the legal permit (license number 27021-1) from the Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling editor: Sabine Begall.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Giné, G.A.F., da Silva, K.F.M. & Faria, D. Decreasing dietary diversity following habitat loss: the case of the thin-spined porcupine in the Atlantic forest. Mamm Biol 100, 473–484 (2020). https://doi.org/10.1007/s42991-020-00051-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-020-00051-9