Abstract
Cropping systems in many sub-tropical and tropical regions rely on phosphorus (P) fertilisers to maintain crop yields. However, crop responses are often spatially and temporally inconsistent. This study evaluated P availability from dispersed applications of fresh phosphate fertiliser and their residual effect in two contrasting Vertisols - a major soil type for crop production. We collected soils that had received mono-ammonium phosphate (MAP) fertiliser four years prior to sampling (residual), as well as unfertilised soils to which we applied MAP at equivalent rates (‘fresh’). Soil P tests quantified the potential P availability arising from the residual and fresh P applications, and P sorption curves were generated to describe the P sorption/desorption dynamics. A 45-day pot trial evaluated wheat growth and P uptake from fresh and residual P applications in both soils. Soil tests and sorption curves reflected differences in P addition in the two soils, although the sorption characteristics did not differ appreciably in response to residual P. Crop growth and P uptake strongly increased with increasing P availability, with greater responses for the freshly applied P. Low P recovery from aged fertiliser applications demonstrated that the availability of residual P for crop uptake after dispersed applications is limited in Vertisols. P-tests and sorption measurements could collectively capture potential soil P reactions and plant P availability in two contrasting Vertisols. Residual P benefits from dispersed applications remained low. Enrichment in a defined soil volume (i.e., banding) merits further investigation in this soil type.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Vertisols (WRB 2015) are an important soil type that support food production systems in sub-tropical and tropical regions, especially for the cropping systems in India, Australia, Sudan, United States, Chad, and China. Vertisols from northern Australia have historically been able to supply enough phosphorus (P) for crop production (Dalal 1997; Raymond et al. 2021; Wang et al. 2007), therefore have received little or no phosphate fertiliser to support crop production. This continuous removal of native soil P in harvested products has led to a decline in P fertility, with P depletion most notable in the subsoils (Bell et al. 2012; Bell et al. 2013). Consequently, cropping systems on these soils have become increasingly reliant on mineral phosphate fertiliser (Angus et al. 2019; McLaren et al. 2014).
To support agronomic decision making, “critical soil P concentrations” have been established, below which application of P is likely to elicit a crop response. The most commonly used soil test to predict P responsiveness is a bicarbonate-extractable P test, Colwell-P (Colwell 1963). Additional insights into soil P reserves and short term availability are derived from (i) a dilute acid P test, BSES-P (Bureau of Sugar Experiment Stations (Kerr and Von Stieglitz 1938)), and (ii) a CaCl2-extractable P test. Combined, these three soil tests are assumed to extract various soil P pools of different lability (Colwell: P in the soil solution and weakly adsorbed P; BSES: P in the soil solution, some amorphous calcium-phosphate (Ca-P) and most adsorbed P; and CaCl2: P in the soil solution (McLaren et al. 2014; Moody et al. 2013)). However, the ability of soil P tests to predict fertiliser P requirements is based on empirical relationships with crop fertiliser responses and there are no available tests which provide a ‘universally’ useful predictor across all soil types (Moody et al. 2013; Speirs et al. 2013). For Vertisols, Colwell-P and BSES-P measurements are typically interpreted with the assumption that additional P measured in the BSES-P test reflects a potential supply of moderately labile P forms (i.e., calcium phosphates). These minerals are often present in high concentrations in Vertisols but are only sparingly accessible to plants as a P source (McLaren et al. 2014). However, uncertainty remains as to the appropriate interpretation of these soil test results, in terms of likely crop responses to applied P fertiliser. For example, current recommendations are to apply deep phosphate fertilisers bands (15-25 cm depth) every three to five years in Vertisols with limited subsoil P reserves (typically < 10 mg kg-1 Colwell P and < 50 mg kg-1 BSES-P for both tests) but crop responses to these P applications have been variable (Angus et al. 2019). While part of this variability is likely related to fluctuating seasonal soil-water availability and root access to more P-enriched topsoil layers (Angus et al. 2019), there is an increasing need to better understand the underlying bioavailability of P fertiliser applied to P-depleted Vertisols.
In recent years, substantial research has been conducted on the behaviour of P fertiliser in P-depleted Vertisols in NE Australia. For example, Raymond et al. (2023) conducted short-term sorption assays on a wide range of Vertisols with low P availability, which indicated that at typical commercial application rates much of the applied P is rapidly sorbed, with little subsequently returning to the soil solution through desorption. In contrast, a much greater proportion of applied P was desorbed at high concentrations of added P (i.e., concentrations close to those observed within P fertiliser bands). The further loss of P availability with time has been interpreted as initially metastable adsorbed or precipitated P gradually becoming less soluble (Barrow 2008; Lindsay et al. 1989; Penn and Camberato 2019). This decline may be countered by greater P application rates, but when large amounts of P are applied in concentrated fertiliser bands, the solution chemistry in the vicinity of these concentrated bands can lead to precipitation of added P as a variety of taranakites and calcium-phosphate minerals (Meyer et al. 2021). While these laboratory studies clarified how different amounts and concentrations of P affected the short and medium-term reactivity and potential availability of banded P fertilisers, little is known about the bioavailability of this P in the longer term in relation to the level of P enrichment and intrinsic soil properties. Crops often only take up 10–20 % of the P fertiliser applied in the first year after application (Holford 1997), thus a substantial part of applied P accumulates in the soil as residual P. This residual P can be taken up by crops in subsequent years, depending on a range of soil and plant factors. The main dominant soil characteristics that regulate residual P supply include soil P sorption capacity (Arai and Sparks 2007). Whilst initial sorption of applied P is very rapid (Barrow 1980), the process continues over time. Thus, in soils with a high P sorption capacity, often little P remains in solution after long periods following fertiliser application. At the plant root surface, rapid uptake of inorganic P results in a depletion of soil solution P concentrations around the root, with this concentration gradient extending out through the rhizosphere to the bulk soil and providing the driver for diffusive supply of P to the plant root system. This depletion of soil solution P contributes to the slow release of inorganic P by the processes of desorption or dissolution (Hinsinger 2001), with the capacity of soils to release P being inversely related to their sorption capacity. Plant roots may also interact with less labile P pools in soil through the secretion of exudates (protons and organic anions) which mobilise less available P forms (Hinsinger 2001; Wang and Lambers 2020; Yli-Halla 2016). These soil and plant factors can collectively influence the residual value of applied fertiliser P, further highlighting the need to better understand the availability of P in the context of the soil-plant system.
Our overall aim was to evaluate the P status and residual availability of P fertilisers mixed into two contrasting Vertisols. We hypothesized that the intrinsic soil properties and fertiliser history affect P availability and P status. The specific objectives were to (i) evaluate the existing soil P tests against plant responses to freshly applied P fertiliser; (ii) determine whether these soil P tests also reflect plant responses to residual P; (iii) assess the efficacy of the soil tests for providing a common basis for assessing soil P responsiveness; and (iv) determine the key soil characteristics which influence P fertiliser recovery by plants in high P sorption soils.
2 Materials and Methods
2.1 Soil collection
We collected soils from two field trials that investigated the fate and residual effects of phosphate fertilisers in Vertisols. The first trial was located at Hopeland (south-east Queensland, Australia; from hereon referred to as ‘Hopeland Vertisol’), and the second at Gindie (Central Queensland, Australia; from hereon referred to as ‘Gindie Vertisol’). Both soils are typical examples of cropped Vertisols found in the northeast of Australia, with a long cropping history and historically low fertiliser P inputs. The soils differed in soil pH, clay content, parent material and P status (Table 1). The Hopeland soil is derived from a mix of basalt and alluvial parent materials, whereas the Gindie soil was weathered in situ from basalt.
In the field trials, the P fertiliser treatments were applied as mono-ammonium phosphate (MAP, NH4H2PO4) solutions sprayed onto the soil surface before incorporation with conventional tillage equipment. Farmers in North-East Australia generally use MAP as the preferred P source on these soils. The fertilisers were applied in late 2017 (Gindie) and late 2018 (Hopeland), with prolonged dry conditions resulting in both sites only being cropped three times in the period leading up to soil collection in 2021. Crops sown at the Hopeland site included maize (2019 - not harvested), barley (2019), and chickpea (2020). At Gindie the crop succession differed slightly with chickpea (2018), wheat (2020), and mungbean (2021). The Hopeland soil was collected in August 2021 from blocks sown to winter wheat (33 months after fertiliser application). The Gindie soil was collected in June 2021 after the mungbean crop (43 months after fertiliser application).
Soil was collected from the unfertilised buffer zone of each field (i.e., no P fertiliser application) and from treatments to which P was applied at rates that represented ~30 kg P ha-1 (soil concentration of 27.6 mg P kg-1), ~100 kg P ha-1 (82.8 mg P kg-1), or ~165 kg P ha-1 (137.9 mg P kg-1) in the top 10 cm soil profile layer. The soils that had previously received MAP applications in the field are referred to as ‘residual P’ treatments in this study, while the soil from the unfertilised buffer zone subsequently received fresh MAP applications at rates that matched those in the original field applications.
2.2 Soil Preparations
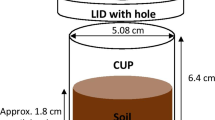
All soils were air-dried at room temperature, crushed (< 4 mm), and separated into bags of 3 kg of soil. Each bag received basal mineral nutrients, except P (K2SO4 370 mg kg−1, CaCl2 57 mg kg−1, MnSO4 9.4 mg kg−1, ZnSO4 3 mg kg−1, CuSO4 1.3 mg kg−1, Na2MoO4 15 μg kg−1, NH4NO3 286 mg kg−1 (100 mg N kg−1) and MgSO4 198 mg kg−1 (Raymond et al. 2018). ‘Fresh’ MAP fertiliser was applied at increasing rates to bags of the previously unfertilised soil (i.e., soil from buffer zones) to achieve fertiliser P concentrations of 0, 6.9, 13.8, 20.7, 27.6, 34.5, 55.2, 82.8, 110.3 or 137.9 mg P kg-1 (i.e., a response curve). The residual treatments (0, 27.6, 82.8 and 137.9 mg added P kg soil-1) and corresponding fresh (27.6, 82.8, and 137.9 mg P kg-1) treatments were replicated five times. The other unreplicated fertiliser P concentrations served as additional points along the entire response curve. Apart from P, MAP contains 120 mg N g-1. Therefore, variable amounts of urea were added to ensure equal N concentrations amongst the different fresh MAP applications. The MAP and urea were sprayed onto the soil in the fresh treatments, air-dried, and thoroughly mixed to ensure an even distribution. This approach mimicked the fertiliser applications in the field trials (i.e., the now ‘residual’ treatments). Thoroughly mixed soil from all the freshly applied P and the residual P treatments was placed in PVC pots that were 90 mm diameter and 50 cm high. Soil water content was adjusted to 70 % of field capacity (Table 1) and all treatments were then incubated in a temperature-controlled glasshouse for one week with day/night temperatures of 22°C and 18°C.
2.3 Pot trial
A pot experiment was conducted in a air-conditioned glasshouse (equal duration day/night temperature of 22/18 °C) with wheat (Triticum aestivum cv Sunmax) grown in both soils. The fresh and residual P treatments were randomized across five replicate blocks. To ensure uniform establishment, wheat seeds were germinated in a Petri dish for 24 h at room temperature and three seeds were sown per pot. After emergence, the plants were thinned to two plants per pot. The soil water content was adjusted by weighing and watering the pots every two days to reach 80 % of field capacity (Table 1).
Forty-five days after sowing, the plant shoots were cut as close as possible to the soil surface and oven-dried at 65 °C until stable weight. The whole shoot samples were then milled before digesting 100 mg of plant material with 2.5 mL 70% nitric acid and 1 mL 30% hydrogen peroxide (McQuaker et al. 1979). The P concentration in the extracts was measured using inductively coupled plasma optical emission spectroscopy (Thermo ICAP 6500 ICP OES Radial View, Massachusetts, United States). Total N in the plant shoot was measured on selected samples to ensure that any plant responses had not been limited by inadequate N availability. Tissue N concentration was determined on a total N Analyzer (Leco FP528, NSW, Australia) after Dumas high temperature combustion (Rayment and Lyons 2011). Plant roots were recovered by carefully washing away the soil, with roots then oven-dried at 65 °C and stable dry weight recorded.
2.4 Phosphorus Sorption Curves
Jars containing 100 g soil from the fresh and residual P treatments were prepared in the same manner as the treatments in the pot trial, but replicated three times. The soil in each jar was brought to 70 % of field capacity and incubated for one week at room temperature (20 °C). After incubation, the soils were air-dried, crushed to < 2mm, and split into four subsamples. The first three subsamples were used for measurement of CaCl2-P, Colwell-P, and BSES-P (Rayment and Lyons 2011). The fourth subsample was used to produce P sorption curves, following a modified procedure used by Barrow and Debnath (2014). For each treatment, 2 g of soil was weighed into a 50 mL tube and mixed with 20 mL 0.01 M CaCl2 containing either: 0, 10, 20, 50, 75, 100, 150 and 200 mg P L-1 (prepared from 500 mg P L-1 (KH2PO4 stock solution)). The analyses were performed in triplicate for each soil. Tubes were shaken on a rotary shaker at 25 °C for 17 h. Orthophosphate concentration in the extracts was then measured in the supernatant after centrifugation (5 min, 4000 rpm), using the malachite green method (Ohno and Zibilske 1991).
2.5 Data Analysis
The relationships between the rates of ‘fresh’ and ‘residual’ MAP applications and the concentrations of plant available P (CaCl2-P, Colwell-P, and BSES-P extractions) were analysed by linear regression.
Plant growth responses (shoot biomass and P uptake) to MAP applications and plant available P (CaCl2-P, Colwell-P, and BSES-P extractions) were described using Mitscherlich non-linear curves in R (nls package (Baty et al. 2015)) by use of the following equation:
Where “y” is the plant response to P fertiliser addition (shoot biomass or shoot P content), “a”, “b” and “c” are equation coefficients and “x” is either the rate of P fertiliser added or the concentration of potentially plant available P (CaCl2-P, Colwell-P or BSES-P). The critical P application rate was defined as the amount of P applied to achieve 90 % of maximum yield for that soil (Dyson and Conyers 2013).
Phosphorus sorption curves were described using a three coefficient Freundlich equation, from which sorption and desorption were derived:
Where PS is the P sorbed in mg P kg-1, PSol is the P concentration in mg P L-1 at equilibrium after the sorption step, and “a” and “b” are the Freundlich coefficients. The intercept term is “c” (Barrow 2008).
We calculated the degree of P saturation (DPS) as the ratio between Colwell P concentration and the P sorption index PBIColwell-P as described in Rayment and Lyons (2011), with data expressed as a percentage.
All data analyses were performed in R version 4.1.2 (Core 2015) using the RStudio development environment, with graphical illustrations made using the ggplot package (Wickham 2016).
3 Results
3.1 P Availability as Assessed by Soil Extractions
Both soils showed clear linear relationships between added P and extractable P for Colwell-P and BSES-P tests in the fresh and residual treatments (Fig. 1B, C, E, F and Table S2). However, the rate of increase in extractable P for each additional amount of P added was greater for the Hopeland soil, with this effect particularly evident for the BSES-P extractions. In the Gindie soil, the application of fresh MAP increased Colwell-P more than BSES-P. Interestingly, the relationship between MAP addition and CaCl2-P was not linear in either soil (Fig. 1A&D), with the commencement of more rapid increases in CaCl2-P occurring at rates of P addition > 30 mg P kg-1 in the Hopeland soil. This curvilinearity was less pronounced in the Gindie soil but was still clearly evident for rates of P addition > 55 mg P kg-1.
Potentially plant available phosphorus indicated by CaCl2-P (0.005 M calcium chloride 1:10 (w/v)) (A&D), Colwell-P (0.5 M sodium bicarbonate, pH8.5 1:100 (w/v)) (B&E), and BSES-P (0.005 M sulfuric acid 1:200 (w/v)) (C&F) soil tests following fresh and in-field (residual) mono-ammonium phosphate fertiliser (MAP) applications. Relationship between the amount of fertiliser added and soil tests was modelled with a linear model with P < 0.05 indicating a significant linear relationship. For the CaCl2-P (A&D), an exponential model was used to explain the relationship between fresh fertiliser application and CaCl2 extractable P, with P < 0.05 indicating a significant relationship
Colwell-P and BSES-P were always greater in the residual P fertiliser treatments than in the unfertilised soils but the increases at the lowest residual P rate (27.6 mg P kg-1) were small (Fig. 1B&E). In contrast, CaCl2-P values were similar for all the residual P treatments and the unfertilised control soils (Fig. 1A&D). Further, Colwell-P and BSES-P were much lower in the residual treatments than their corresponding rates of fresh P fertiliser application. As an example, Colwell-P in the 27.6 mg P kg-1 treatment for both soils was approximately 50 % of that in the same treatment with freshly applied P.
In the Gindie soil, the relative increases in Colwell-P concentrations from residual P compared to the same rate of fresh P tended to increase with the rate of P addition, rising from 48 % to 56 % of the freshly applied P value (i.e., as P addition increased from 27.6 mg P kg-1 to 137.9 mg P kg-1 (Fig. 2A)). However, in the Hopeland soil, the relative increases of residual versus freshly applied P decreased with increasing rates of P application, from 50 % at 27.6 mg P kg-1 to only 27 % at 137.9 mg P kg-1 (Fig. 2A). Similar patterns were observed for BSES-P, with the recovery of applied P from residual treatments increasing from 45 % to 62 % of that from freshly applied P in the Gindie soil but decreasing from 60% to 39% in the Hopeland soil (Fig. 2A).
(A) Phosphorus recovery in the residual treatments for Colwell-P (0.5 M sodium bicarbonate, pH8.5 1:100 (w/v)) and BSES-P (0.005 M sulfuric acid 1:200 (w/v)) soil tests; (B) Relationship between Colwell and BSES-P tests for the Gindie and Hopeland soils (both fresh and residual mono-ammonium phosphate fertiliser (MAP) applications). Relationship between the Colwell-P and BESES-P test was modelled with a linear model with P < 0.05 indicating a significant linear relationship
Colwell-P and BSES-P were closely correlated for both soils (Fig. 2B). However, BSES-P was significantly less effective at desorbing/solubilizing P in the Gindie soil than in the Hopeland soil, where an almost 1:1 relationship was observed between the two extraction solutions.
3.2 Effect of P-fertiliser Addition on P-sorption Capacity
The three-parameter Freundlich equation accurately described the P sorption behaviour in both soils (Fig. 3 and Table S2). For a given solution P concentration, the amount of sorbed P was smaller in soils which had received P applications than in the 0P control, with less P sorbed across the range of solution P concentrations. In the fresh P treatments, the proportion of sorbed P decreased with increasing fertiliser P application rate (Fig. 3B, 3D), but for the residual treatments there were no measurable differences between the original application rates (Fig. 3A, 3C). The sorption curve equations indicated that the amounts of P sorbed were similar for the 27.6 mg P kg-1 residual and fresh applications, and consistently greater for the residual P treatments than for the equivalent rates of freshly applied P. For example, at an equilibrium solution P concentration of 10 mg P l-1, the 137.9 mg P kg-1 treatment in the Gindie soil sorbed 349.1 mg P kg-1 in the residual treatment but only 292.5 mg P kg-1 for the freshly applied equivalent.
The PBIColP only marginally varied with increasing amounts of fertiliser P addition from either freshly applied fertiliser P or residual applications in both soils (Fig 4A & B). The addition of P fertiliser increased DPS in both soils in a manner consistent with the increase in Colwell-P concentration (Fig. 4C & D). In the Hopeland soil, the P sorption sites reached apparent saturation (DPS > 100%) for rates of fresh P addition > 110 mg P kg-1, while in the Gindie soil the DPS had only reached ~85% saturation at a rate of 137.9 mg P kg-1. The DPS values for the residual P treatments increased with increasing P addition but the values were lower than the corresponding rates of fresh addition, particularly in the Hopeland soil.
Effect of P fertiliser (mono-ammonium phosphate (MAP)) addition on measured phosphorus buffering capacity corrected with Colwell-P soil test (0.5 M sodium bicarbonate, pH8.5 1:100 (w/v)) (PBIColP) (A, B) and the degree of P saturation (DPS – C, D) as a function of the quantity of P added. Treatments receiving either freshly applied MAP (dark blue) or residual MAP in the field experiment (light blue) are shown. Relationship between the DPS and added P test was modelled with a linear model with P < 0.05 indicating a significant linear relationship
3.3 Plant Response to P-addition
Increases in plant biomass, shoot P concentrations, and P-uptake were observed in response to increasing rates of fresh P fertiliser addition in both soils (Figs. 5, 6 & Table S2). The response curve for shoot biomass showed a clear plateau, with maximum biomass achieved at rates of fresh P addition between 40 and 50 mg P kg-1 (Fig. 5A & B). However, the accumulation of P in plant aboveground biomass had not reached a plateau at the highest the rates of P addition in either soil (Fig. 5C & D). Unfortunately, plants grown in the highest rate of residual P in the Gindie soil were compromised due to a missed basal Zn application, and so could not be considered in this analysis. Nonetheless, it was clear that shoot biomass production and P uptake in both soils were significantly lower in the residual P fertiliser treatments than in the equivalent rate of freshly applied P (p > 0.0001).
Plant biomass and plant phosphorus uptake in response to increasing rates of mono-ammonium phosphate fertiliser (MAP) (A, B, C, D) and the resulting Colwell-P (0.5 M sodium bicarbonate, pH8.5 1:100 (w/v)) concentration (E, F, G, H). The MAP was applied fresh (dark blue) or was residual from the field experiment (light blue). The fitted curves use a non-linear regression model (Mitscherlich) (y = a - b × (e-c×x)). For the addition rates of 27.6, 82.8, and 137.9 mg P kg-1 the average of four replicates is presented. Error bars are standard errors.
Relative shoot biomass production in response to increasing Colwell-P (0.5 M sodium bicarbonate, pH8.5 1:100 (w/v)) following mono-ammonium phosphate fertiliser (MAP) addition mixed through soil volume (fresh and residual application). The maximum biomass was based on the shoot biomass obtained from the highest rate of freshly applied P fertiliser (i.e. 137.9 mg P kg-1). The fitted curves use a non-linear regression model (Mitscherlich) (y = a - b × (e-c×x))
In both soils, wheat biomass and P uptake increased with increasing rates of residual P. Although plants grew poorly in the residual 27.6 mg P kg-1 treatment of the Gindie soil, plant growth was similar in both soil types at the residual 82.8 mg P kg-1 rate (i.e., producing 40%-46% of the biomass and 26-28 % of the P uptake compared to the freshly applied P equivalent; Fig. 5A & B). The residual 137.9 mg P kg-1 rate in the Hopeland soil produced ~65% of the maximum biomass and 35% of the maximum P uptake achieved in the equivalent fresh P treatment. For comparison, this biomass and P uptake was only equivalent to that achieved with additions of 20.7 - 27.6 mg P kg-1 of freshly applied P. While plant responses to residual P were relatively small, a clear trend for biomass production and plant P uptake with Colwell P concentrations measured in both fresh and residual treatments was observed (Fig. 5).
Relative yield curves fitted to each soil showed clear differences between soils in wheat responses to increasing P availability at lower Colwell-P concentrations (Fig. 6). Wheat grown in the Hopeland soil demonstrated a greater relative biomass production than wheat grown in the Gindie soil for Colwell-P concentrations < 40 mg P kg-1. The Colwell-P concentration required to reach 90% of maximum biomass (Y90%, derived from Fig. 6 and shown in Table 2) was 41 mg P kg-1 in the Hopeland soil (requiring the addition of about 40 mg P kg-1), but was 66 mg P kg-1 in the Gindie soil (requiring the addition of 80 mg P kg-1). The equivalent BSES-P concentration for Y90% was similar to that for Colwell-P in the Hopeland soil but was lower than the equivalent for Colwell-P in the Gindie soil (Table 2). The critical CaCl2-P concentrations for Y90% were extremely low compared to the equivalent Colwell-P and BSES-P concentrations and were much lower in the Gindie soil than in the Hopeland soil (Table 2).
4 Discussion
This study extends previous laboratory incubations that describe short and medium-term reactivity and potential P availability in similar soils (Dorahy et al. 2007; Meyer et al. 2021; Raymond et al. 2023) by considering the long-term residual P value several years after fertiliser application. Furthermore, we link the potential P availability as indicated by soil tests to plant P uptake by a globally agronomically-relevant crop. This relationship has been previously unclear in high P-sorbing Vertisol soils. These data are relevant to agronomic decision-making, providing information on the residual value of historical P fertiliser applications and improving the reliability of using soil test measurements to underpin P nutrition management.
Our first objective was to determine whether commonly used soil P tests reflected plant responses to recently applied P fertiliser. Unfertilised soils were characterised by low plant available P concentrations, indicated by Colwell-P, BSES-P and CaCl2-P, and each had P buffer indices (PBIColP) that would place them in the low to moderate PBI category (Burkitt et al. 2002; Moody 2007). Subsequently, P additions were expected to elicit plant biomass responses and increases in plant P uptake. All three soil tests demonstrated increasing amounts of extractable P with larger additions of fresh P fertiliser, which correlated with plant responses. However, differences in the total P addition required to achieve Y90% between the two soils for Colwell-P measurements (but not BSES-P) suggests that there was potentially little formation of sparingly soluble forms of P in the short-term in both soils. Both extraction solutions were therefore effective at measuring the (presumably) same desorbable P pool. Although CaCl2-P also exhibited good correlation with plant responses (Fig. S2), this relationship occurred at much lower P concentrations than for Colwell and BSES-P. These values are likely indicative of a much smaller and more reactive P pool that is in dynamic equilibrium with both sorbed P and sparingly soluble mineral P, considering CaCl2-P is the weakest extracting solution among the three methods and is known to correlate with the concentration of P in soil solution (Moody et al. 2013). Overall, our data shows that all three P tests were able to predict wheat responses to P fertiliser that was freshly mixed through soil. Over this short duration of soil incubation, correlations between the different P tests were similar and so any individual test was able to satisfactorily predict plant P availability in the short-medium term from these two Vertisols.
Our second and third objectives were to evaluate (i) whether common soil P tests can predict plant responses to historic rates of P addition (i.e., residual P availability) and (ii) to determine if soil P tests of residual P availability demonstrate the same relationships to plant responses as those of freshly applied P (i.e., do soil P tests deliver consistent results between fresh and aged P applications?). Like the fresh P applications, soil P test values increased with increasing rates of historical P fertiliser application. As expected, the concentrations of extractable P were always much lower than those from freshly added P, but the proportion of extractable P over the differing rates of P addition varied from that of fresh P applications. In particular, the CaCl2 test showed little sensitivity to residual P, with very small amounts of extractable P across all the residual P rates in both soils. Thus, the low CaCl2-P values in the residual P treatments likely reflect the effects of continued sorption and seasonal uptake, which would have resulted in low solution P concentrations that are hard to distinguish from those found in unfertilised soils. The inability of the CaCl2 test to adequately reflect plant P availability, combined with analytical difficulties due to P contents close to or below detection limits (Börling et al. 2004; Six et al. 2012), make this extracting solution less suited to measuring residual fertiliser P benefits. In contrast, our results demonstrate that both Colwell-P and BSES-P tests can provide measures of residual P availability that correlate with wheat responses. The amounts of extractable P and plant responses to residual P were always smaller than those observed for the same rates of freshly added P. This would be expected given (i) seasonal uptake of P during the cropping seasons removes applied P from soil, and (ii) the longer duration of contact between the soil and applied fertiliser P increases the potential for in situ adsorption and precipitation reactions, thus removing P from solution (Barrow 1983). The relative contributions of these processes is unknown, but the observations are unlikely to be strongly driven by crop P removal. Both sites were cropped for three growing seasons, in which apparent fertiliser P removal in harvested grain was 7.4 kg P ha-1 (removal of ~6.2 mg P kg-1 assuming a soil bulk density of 1.2 g cm3) in the Gindie soil and 10.3 kg P ha-1 (removal of ~8.6 mg P kg-1 assuming a soil bulk density of 1.2 g cm3) in the Hopeland soil for the highest rates of P addition (>80 mg P kg-1) (unpublished data). These relatively small additional crop P removals indicate that the vast majority of P remained in soil as residual P, and thus that sorption or precipitation of P (i.e., soil-driven P reactions) should be the major driver of plant P availability in residual treatments (Raymond et al. 2023).
Specific soil properties were likely to influence P reactions and the efficacy of soil P tests to predict plant P availability. Specifically, properties related to parent material of these soils showed marked differences. Soils were derived from either basaltic (Gindie) or a mixture of basaltic and alluvial parent materials (Hopeland), with the greater concentrations of crystalline and amorphous Al and Fe oxides in the Gindie soil (Raymond et al. 2023) consistent with the slightly higher initial P sorption capacity observed in this soil. This was also reflected in the increases in extractable P, which were proportionally greater with increasing rate of P addition in the Hopeland soil compared with the Gindie soil. Of particular interest was the greater increase in BSES-P compared to Colwell-P when P was applied to the Hopeland soil (cf. Gindie soil). Given BSES-P is thought to be mostly related to a combination of sorbed P and sparingly soluble P minerals (e.g. calcium phosphates (Ca-P)) the proportionally greater increase of BSES-P recovery suggests that a greater amount of applied P may undergo precipitation in this soil (McLaren et al. 2014; Moody et al. 2013). The likelihood of precipitation of P minerals in the Hopeland soil is also supported by other soil characteristics which predispose to Ca-P precipitation, including a more alkaline pH and the presence of carbonates. This was also confirmed in the P sorption assay. When the sorbed P concentrations were plotted against concentration raised to a fractional power, to test if the Freundlich equation applies (Raymond et al. 2023) (Fig. S4), it was clear that the two soils had contrasting behaviour to increasing P concentration in the soil solution. The Gindie soil showed a linear relationship between solution P concentration and sorbed P, whereas the Hopeland soil showed an inflection point that is indicative of precipitation reactions. The Hopeland soil also demonstrated rapid exponential increases in CaCl2-P with increasing rates of fresh P addition, and achieved 100% DPS at lower rates of P addition (cf. Gindie soil). This suggests that there is a greater likelihood of P saturation in the solution of this soil with increasing rates of added P, potentially inducing precipitation reactions.
The smaller proportion of added P that was recovered from residual P applications in the Hopeland soil (cf. Gindie soil) is consistent with greater proportions of fertiliser P becoming tightly bound to the soil solid phase; this may also be an indication of formation of more stable precipitates such as octo-calcium phosphate or apatite (Arai and Sparks 2007; Castro and Torrent 1998). In contrast, the addition of P fertiliser increased Colwell-P and BSES-P recoveries in a similar manner in the Gindie soil. There was no evidence of saturation of P sorption sites in this soil (DPS only reached ~85 % at the highest rate of P addition). Consequently, a larger proportion of P was likely retained on sorption sites and was potentially plant available in the residual treatments.
In both soils, freshly and historically applied P had little effect on the PBIColP, although it had a clear effect on the sorption curves, by reducing the amounts of P sorbed at the higher concentrations of solution P. Other studies have shown that P fertiliser applications can affect the P sorption isotherms (Barrow and Debnath 2014; Barrow et al. 1998), but the lack of PBIColP change after P fertiliser addition may be the result of the relatively small concentrations of added P in these studies, which were chosen to mimic the range of P rates applied in the field experiments.
The relatively large decreases in available P over time, and the low P recovery in plant biomass (especially the residual treatments), illustrate the risks associated with dispersing fertiliser P through large soil volumes. Dispersing P fertiliser is often used in an effort to increase the proportion of the root system exposed to P-enriched soil (van der Bom et al. 2023a). The application of dispersed P fertiliser has been shown to be more effective in terms of P uptake for durum wheat (Triticum durum L.) compared to freshly applied concentrated P fertiliser bands under optimal soil water conditions (van der Bom et al. 2023b). While banded P application decreases the proportion of the root system exposed to P-enriched soil, it was also shown that banded P fertiliser application often resulted in the precipitation of P as Al-P minerals and Ca-P minerals (for calcareous soils) - which greatly reduced P availability (Meyer et al. 2021). However, the water availability through the soil profile can greatly influence the performance of the various P applications on crop productivity (van der Bom et al. 2023b). This illustrates the complexity of P applications in these sub-tropical cropping systems. Further investigation of the implications of different degrees of soil-P fertiliser mixing (e.g. band frequency and in-band concentrations, limiting P application to specific locations using zonal tillage and full fertiliser incorporation) on recovery of applied P by crops grown in Vertisols with a range of mineralogies and P sorption capacities is needed to guide the development of soil-specific fertiliser P management strategies.
5 Conclusions and Perspectives
Overall, this study confirmed that intrinsic soil properties and fertiliser history influence P reactions and efficacy of soil P tests. The use of Colwell-P (0.5 M sodium bicarbonate, pH8.5 1:100 (w/v)) and BSES-P (0.005 M sulfuric acid 1:200 (w/v)) measurements in combination with the phosphorus buffering index (PBIColP) assessments (as currently recommended by industry guidelines in Australia) is an appropriate strategy for making P nutrition decisions. These soil tests adequately reflect the dynamics of incorporated fertiliser P applications and the availability of that P to meet the short-medium term demand of wheat in two high-P sorbing and chemically contrasting Vertisols. The utility of these tests to reflect P availability from both fresh and residual fertiliser P provides confidence in the robustness of this approach for these soils, which is especially important given the increasing prevalence of residual P in crop nutrition programs.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Angus J, Bell M, McBeath T, Scanlan C (2019) Nutrient-management challenges and opportunities in conservation agriculture. In: Pratley J, Kirkegaard J (eds) Australian Agriculture in 2020: From Conservation to Automation. Charles Sturt University, Wagga Wagga NSW, Australia, Australian Society of Agronomy http://agronomyaustraliaproceedings.org/index.php/special-publications
Arai Y, Sparks D (2007) Phosphate reaction dynamics in soils and soil components: A multiscale approach. Adv Agron 94:135–179. https://doi.org/10.1016/S0065-2113(06)94003-6
Barrow N (1980) Evaluation and utilization of residual phosphorus in soils. In: The role of phosphorus in agriculture, pp 333–359. https://doi.org/10.2134/1980.roleofphosphorus.c14
Barrow NJ (1983) On the Reversibility of Phosphate Sorption by Soils. Eur J Soil Sci 34:751–758. https://doi.org/10.1111/j.1365-2389.1983.tb01069.x
Barrow NJ (2008) The description of sorption curves. Eur J Soil Sci 59:900–910. https://doi.org/10.1111/j.1365-2389.2008.01041.x
Barrow N, Debnath A (2014) Effect of phosphate status on the sorption and desorption properties of some soils of northern India. Plant Soil 378:383–395. https://doi.org/10.1007/s11104-014-2042-8
Barrow NJ, Bolland MDA, Allen DG (1998) Effect of previous additions of superphosphate on sorption of phosphate. Soil Research 36:359–372. https://doi.org/10.1071/S97110
Baty F, Ritz C, Charles S, Brutsche M, Flandrois J, Delignette-Muller M (2015) A Toolbox for Nonlinear Regression in R: The Package nlstools. J Stat Softw 66:1–21. https://doi.org/10.18637/jss.v066.i05
Bell M, Lester D, Smith L, Want P (2012) Increasing complexity in nutrient management on clay soils in the northern grain belt–nutrient stratification and multiple nutrient limitations. In: Proceedings of 16th Australian Agronomy Conference
Bell MJ, Moody PW, Anderson GC, Strong W (2013) Soil phosphorus—crop response calibration relationships and criteria for oilseeds, grain legumes and summer cereal crops grown in Australia. Crop Pasture Sci 64:499–513. https://doi.org/10.1071/CP12428
Burkitt LL, Moody PW, Gourley CJP, Hannah MC (2002) A simple phosphorus buffering index for Australian soils. J Soil Research 40:497–513. https://doi.org/10.1071/SR01050
Börling K, Otabbong E, Barberis E (2004) Soil Variables for Predicting Potential Phosphorus Release in Swedish Noncalcareous Soils. J Environ Qual 33:99–106. https://doi.org/10.2134/jeq2004.9900
Castro B, Torrent J (1998) Phosphate sorption by calcareous Vertisols and Inceptisols as evaluated from extended P-sorption curves. Eur J Soil Sci 49:661–667. https://doi.org/10.1046/j.1365-2389.1998.4940661.x
Colwell J (1963) The estimation of the phosphorus fertilizer requirements of wheat in southern New South Wales by soil analysis. Aust J Exp Agric 3:190–197. https://doi.org/10.1071/EA9630190
Core TR (2015) R: A language and environment for statistical computing. R Core Team
Dalal RC (1997) Long-term phosphorus trends in Vertisols under continuous cereal cropping. Aust J of Soil Res 35:327–339. https://doi.org/10.1071/S96052
Dorahy C, Blair G, Rochester I, Till A (2007) Availability of P from 32P-labelled endogenous soil P and 33P-labelled fertilizer in an alkaline soil producing cotton in Australia. Soil use manage 23:192–199. https://doi.org/10.1111/j.1475-2743.2007.00083.x
Dyson CB, Conyers MK (2013) Methodology for online biometric analysis of soil test–crop response datasets. Crop Pasture Sci 64:435–441. https://doi.org/10.1071/CP13009
Hinsinger P (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant Soil 237:173–195. https://doi.org/10.1023/A:1013351617532
Holford ICR (1997) Soil phosphorus: its measurement, and its uptake by plants. Soil Res 35:227–240. https://doi.org/10.1071/S96047
Kerr HW, Von Stieglitz C (1938) Laboratory determination of soil fertility. Sugar Experiment Stations Board, Queensland
Lindsay WL, Vlek PL, Chien SH (1989) Phosphate minerals) Minerals in soil environments, pp 1089–1130. https://doi.org/10.2136/sssabookser1.2ed.c22
McLaren T, Guppy CN, Tighe MK, Moody P, Bell M (2014) Dilute Acid Extraction is a useful Indicator of the Supply of Slowly Available Phosphorus in Vertisols. Soil Sci Soc Am J 78:139–146. https://doi.org/10.2136/sssaj2013.05.0188
McQuaker NR, Brown DF, Kluckner PDJAC (1979) Digestion of environmental materials for analysis by inductively coupled plasma-atomic emission spectrometry. Anal Chem 51:1082–1084
Meyer G, Bell MJ, Lombi E, Doolette CL, Brunetti G, Novotny EH, Klysubun W, Zhang Y, Kopittke PM (2021) Phosphorus speciation in the fertosphere of highly concentrated fertilizer bands. Geoderma 403:115208. https://doi.org/10.1016/j.geoderma.2021.115208
Moody P (2007) Interpretation of a single-point P buffering index for adjusting critical levels of the Colwell soil P test. Soil Res 45:55–62. https://doi.org/10.1071/SR06056
Moody PW, Speirs SD, Scott BJ, Mason SDJC, Science P (2013) Soil phosphorus tests I: What soil phosphorus pools and processes do they measure? Crop Pasture Sci 64:461–468. https://doi.org/10.1071/CP13112
Ohno T, Zibilske LM (1991) Determination of Low Concentrations of Phosphorus in Soil Extracts Using Malachite Green. Soil Sci Soc Am J 55:892–895. https://doi.org/10.2136/sssaj1991.03615995005500030046x
Penn CJ, Camberato JJ (2019) A Critical Review on Soil Chemical Processes that Control How Soil pH Affects Phosphorus Availability to Plants. Agriculture 9:120. https://doi.org/10.3390/agriculture9060120
Rayment GE, Lyons DJ (2011) Soil chemical methods: Australasia. CSIRO publishing
Raymond NS, Jensen LS, Müller Stöver D (2018) Enhancing the phosphorus bioavailability of thermally converted sewage sludge by phosphate-solubilising fungi. Ecol Eng 120:44–53. https://doi.org/10.1016/j.ecoleng.2018.05.026
Raymond N, Kopittke PM, Wang E, Lester D, Bell MJ (2021) Does the APSIM model capture soil phosphorus dynamics? A case study with Vertisols. Field Crops Res 273:108302. https://doi.org/10.1016/j.fcr.2021.108302
Raymond NS, Kopittke PM, Fvd B, Barrow NJ, Bell MJ (2023) Short-term phosphorus sorption and desorption in contrasting cropped Vertisols. Eur J Soil Sci 74(5):e13418. https://doi.org/10.1111/ejss.13418
Six L, Pypers P, Degryse F, Smolders E, Merckx R (2012) The performance of DGT versus conventional soil phosphorus tests in tropical soils - An isotope dilution study. Plant Soil 359:267–279. https://doi.org/10.1007/s11104-012-1192-9
Speirs SD, Scott BJ, Moody PW, Mason SD (2013) Soil phosphorus tests II: A comparison of soil test–crop response relationships for different soil tests and wheat. J Crop Pasture Sci 64:469–479. https://doi.org/10.1071/CP13111
van der Bom FJT, Williams A, Borrell AK, Raymond N, Bell MJ (2023a) Phosphorus management is key to effective deployment of root ideotypes in complex soil environments. Plant Soil. https://doi.org/10.1007/s11104-023-06020-8
van der Bom FJT, Williams A, Raymond NS, Alahmad S, Hickey LT, Singh V, Bell MJ (2023b) Root angle, phosphorus, and water: Interactions and effects on durum wheat genotype performance in drought-prone environments. Plant Soil. https://doi.org/10.1007/s11104-023-05966-z
Wang Y, Lambers H (2020) Root-released organic anions in response to low phosphorus availability: recent progress, challenges and future perspectives. Plant Soil 447:135–156. https://doi.org/10.1007/s11104-019-03972-8
Wang X, Lester DW, Guppy CN, Lockwood PV, Tang C (2007) Changes in phosphorus fractions at various soil depths following long-term P fertiliser application on a black vertosol from south-eastern Queensland. Aust J Soil Res 45:524–532. https://doi.org/10.1071/SR07069
Wickham H (2016) ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York. https://doi.org/10.1007/978-3-319-24277-4
WRB IWG (2015) World Reference Base for Soil Resources 2014, update 2015. International soil classification system for naming soils and creating legends for soil maps. WRB IWG, Rome, Italy
Yli-Halla M (2016) Fate of fertilizer P in soils: inorganic pathway. In: Schnug E, De Kok LJ (eds) Phosphorus in Agriculture: 100% Zero. Springer, pp 27–40. https://doi.org/10.1007/978-94-017-7612-7
Acknowledgements
The authors acknowledge funding from the Grains Research and Development Corporation of Australia (Project no. UOQ1905-009RTX) that enabled this research to be conducted. The authors would also like to thank David Lester, Doug Sands, and Tim Rossignol for providing information about the field trials and assistance in collecting soils for these studies. The authors also thank Meghan Barnard for soil processing and laboratory work, and Brigid McKenna for her ongoing support throughout the project.
Funding
Open access funding provided by Copenhagen University. The authors acknowledge funding from the Grains Research and Development Corporation of Australia (Project no. UOQ1905-009RTX) that enabled this research to be conducted.
Author information
Authors and Affiliations
Contributions
Conceptualization: NR, MB; Methodology: NR, FB, CJ, MB; Investigation: NR, FB, CJ; Formal Analysis: FB, NR; Writing – Original Draft Preparation: NR; Writing – Review & Editing: all.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 203 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Raymond, N.S., van der Bom, F.J.T., Janke, C. et al. Impact of Contrasting Rates of Phosphate Fertiliser Addition in Two Vertisols: Effects of Fresh Application and Residual Fertiliser on Sorption and Plant Uptake. J Soil Sci Plant Nutr 24, 4373–4384 (2024). https://doi.org/10.1007/s42729-024-01841-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-024-01841-7