Abstract
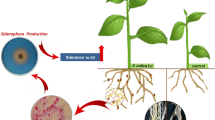
The present study aimed on siderophore and ACC deaminase producing bacteria to mitigate salinity stress and simultaneous mobilization of nutrients. In this study, eight siderophore and ACCD producing bacterial strains selected from a previous study showed that four strains (CWTS 5, CWTS 10, KSBTS 12 and SBTS 12) enhanced mung bean and black gram plant growth in the plate assay (200 mM NaCl stress). Furthermore, evaluation in pot studies (10 g kg− 1 NaCl stress) revealed enhanced root (170.05-237.96% and 16.83-95.74%) and shoot length (139.91-196.96% and 19.87-77.26%) of mung bean and black gram, respectively, compared to the control. Inoculated plants also enhanced the uptake of nitrogen (36.62–41.47 mg g− 1 and 36.19–47.23 mg g− 1), phosphorous (3.29–3.98 mg g− 1 and 3.15–4.21 mg g− 1), Fe (44.36–56.21 µg g− 1 and 41.02–44.31 µg g− 1), Na+ ions (40.65–45.35 µg g− 1 and 20.18–22.11 µg g− 1), and chlorophyll content (12.71–29.33 mg g− 1 and 10.55–26.39 mg g− 1) of mung bean and black gram plants. The Cl− ion uptake by mung bean and black gram plants was below the toxic limit of 94.62–116.6 µg g− 1 and 117.6-150.2 µg g− 1 respectively. The inoculation of bacterial strains also increased the siderophore (259.53-556.05% and 267.26-304.77%) and ACCD (3.02–5.05 µmol mL− 1 and 2.84–4.41 µmol mL− 1) production in the soil and the enhancement was directly correlated with N, P, Fe and chlorophyll content of both the plants. In addition, the inoculated bacteria also enhanced the soil respiration activity such as dehydrogenase enzyme (112.53–167.40 µg g− 1 and 88.92-181.28 µg g− 1), FDA hydrolysis (31.66–44.51 µg g− 1 and 16.94–38.20 µg g− 1) and alkaline phosphatase enzyme (691.53–851.53 µg g− 1 and 511.53-811.53 µg g− 1) of mung bean and black gram soils. Overall, strains CWTS 5 (Bacillus subtilis), SBTS 12 (Rhodococcus sp.), and CWTS 10 (Bacillus albus) can be used to mitigate NaCl stress and mobilize nutrients and Fe in plants.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- ACC:
-
1-aminocyclopropane-1-carboxylic acid
- ACCD:
-
ACC deaminase
- Na+ :
-
Sodium ions
- Cl− :
-
Chloride ions
- Fe:
-
Iron
- N:
-
Nitrogen
- P:
-
Phosphorus
- FDA:
-
Fluorescein diacetate
- TTC:
-
2-3-5-triphenyl tetrazolium chloride
- WHO:
-
World Health Organization
- Ap:
-
Alkaline phosphatase
- NaCl:
-
Sodium chloride
- PGPB:
-
Plant growth promoting bacteria
- CFU:
-
Colony forming unit
- CAS:
-
Chrome azural S
References
Abbas G, Saqib M, Akhtar J, Haq MAU (2015) Interactive effects of salinity and iron deficiency on different rice genotypes. J Plant Nutr Soil Sci 178:306–311. https://doi.org/10.1002/jpln.201400358
Adam G, Duncan H (2001) Development of a sensitive and rapid method for the measurement of total microbial activity using fluorescein diacetate (FDA) in a range of soils. Soil Biol Biochem 33:943–951. https://doi.org/10.1016/S0038-0717(00)00244-3
Agrawal T, Kotasthane AS, Kosharia A, Kushwah R, Zaidi NW, Singh US (2017) Crop specific plant growth promoting effects of ACCd enzyme and siderophore producing and cynogenic fluorescent Pseudomonas. 3 Biotech 7:27. https://doi.org/10.1007/s13205-017-0602-3
Alam A (2014) Soil degradation: a challenge to sustainable agriculture. Int J Sci Res Agric Sci 1:50–55. https://doi.org/10.12983/ijsras-2014-p0050-0055
Barnawal D, Bharti N, Maji D, Chanotiya CS, Kalra A (2014) ACC deaminase-containing Arthrobacter protophormiae induces NaCl stress tolerance through reduced ACC oxidase activity and ethylene production resulting in improved nodulation and mycorrhization in Pisum sativum. J Plant Physiol 171:884–894. https://doi.org/10.1016/j.jplph.2014.03.007
Bessey OA, Lowky OH, Brock MJ (1946) A method for the rapid determination of alkaline phosphatase with five cubic millimeters of serum. J Biol Chem 164:321–329
Brock TD, Madigan MT, Martinko JM, Parker J (2003) Brock biology of microorganisms. Prentice-Hall, Upper Saddle River (NJ)
Casida LE Jr, Klein DA, Santoro T (1964) Soil dehydrogenase activity. Soil Sci 98:371–376
Chandwani S, Amaresan N (2022) Role of ACC deaminase producing bacteria for abiotic stress management and sustainable agriculture production. Environ Sci Pollut Res 29:22843–22859. https://doi.org/10.1007/s11356-022-18745-7
Chandwani S, Chavan SM, Paul D, Amaresan N (2022) Bacterial inoculations mitigate different forms of iron (Fe) stress and enhance nutrient uptake in rice seedlings (Oryza sativa L). Environ Tech Innov 26:102326. https://doi.org/10.1016/j.eti.2022.102326
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560. https://doi.org/10.1093/aob/mcn125
Desai S, Mistry J, Shah F, Chandwani S, Amaresan N, Supriya NR (2023) Salt-tolerant bacteria enhance the growth of mung bean (Vigna radiata L.) and uptake of nutrients, and mobilize sodium ions under salt stress condition. Int J Phytorem 25:66-73. https://doi.org/10.1080/15226514.2022.2057419
Ebrahimi R, Bhatla SC (2011) Effect of sodium chloride levels on growth, water status, uptake, transport, and accumulation pattern of sodium and chloride ions in young sunflower plants. Commun Soil Sci Plant Anal 42:815–831. https://doi.org/10.1080/00103624.2011.552657
Flowers TJ, Munns R, Colmer TD (2015) Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann Bot 115:419–431. https://doi.org/10.1093/aob/mcu217
Gupta A, Bano A, Rai S, Kumar M, Ali J, Sharma S, Pathak N (2021) ACC deaminase producing plant growth promoting rhizobacteria enhance salinity stress tolerance in Pisum sativum. 3 Biotech 11:514. https://doi.org/10.1007/s13205-021-03047-5
Gupta A, Rai S, Bano A, Sharma S, Kumar M, Binsuwaidan R, Pathak N (2022) ACC deaminase produced by PGPR mitigates the adverse effect of osmotic and salinity stresses in Pisum sativum through modulating the antioxidants activities. Plants 11:3419. https://doi.org/10.1080/00103624.2011.552657
Harvey DM, Flowers TJ (1978) Determination of the sodium, potassium and chloride ion concentrations in the chloroplasts of the halophyte Suaeda maritima by non-aqueous cell fractionation. Protoplasma 97:337–349. https://doi.org/10.1007/BF01276291
Hiscox JD, Israelstam GF (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Can J Bot 57:1332–1334. https://doi.org/10.1139/b79-163
Honma M, Shimomura T (1978) Metabolism of 1-aminocyclopropane-1-carboxylic acid. Agric Bio Chem 42:1825–1831. https://doi.org/10.1080/00021369.1978.10863261
Jawad Hassan M, Ali Raza M, Khan I, Ahmad Meraj T, Ahmed M, Abbas Shah G, Ansar M, Afzal Awan S, Khan N, Iqbal N, Peng Y (2020) Selenium and salt interactions in black gram (Vigna mungo L.): ion uptake, antioxidant defense system, and photochemistry efficiency. Plants 9:467. https://doi.org/10.3390/plants9040467
Kang SM, Shahzad R, Bilal S, Khan AL, Park YG, Lee KE, Lee IJ (2019) Indole-3-acetic-acid and ACC deaminase producing Leclercia adecarboxylata MO1 improves Solanum lycopersicum L growth and salinity stress tolerance by endogenous secondary metabolites regulation. BMC Microbiol 19:1–14. https://doi.org/10.1186/s12866-019-1450-6
Karlidag H, Yildirim E, Turan M, Pehluvan M, Donmez F (2013) Plant growth-promoting rhizobacteria mitigate deleterious effects of salt stress on strawberry plants (Fragaria× ananassa). HortScience 48:563–567. https://doi.org/10.21273/HORTSCI.48.5.563
Kaya C, Ashraf M, Dikilitas M, Tuna AL (2013) Alleviation of salt stress-induced adverse effects on maize plants by exogenous application of indole acetic acid (IAA) and inorganic nutrients-A field trial. Aust J Crop Sci 7:249–254
Kumar BS, Prakash M, Narayanan S, Gokulakrishnan J (2012) Breeding for salinity tolerance in mung bean. APCBEE Procedia 4:30–35. https://doi.org/10.1016/j.apcbee.2012.11.006
Lewis RW, Islam A, Opdahl L, Davenport JR, Sullivan TS (2019) Comparative genomics, siderophore production, and iron scavenging potential of root zone soil bacteria isolated from ‘Concord’grape vineyards. Microb Ecol 78:699–713. https://doi.org/10.1007/s00248-019-01324-8
Lu Y, Liu H, Chen Y, Zhang L, Kudusi K, Song J (2022) Effects of drought and salt stress on seed germination of ephemeral plants in desert of northwest China. Front Ecol Evol 10:1026095. https://doi.org/10.3389/fevo.2022.1026095
Omar SA, Fetyan NA, Eldenary ME, Abdelfattah MH, Abd-Elhalim HM, Wrobel J, Kalaji HM (2021) Alteration in expression level of some growth and stress-related genes after rhizobacteria inoculation to alleviate drought tolerance in sensitive rice genotype. Chem Biol Technol Agric 8:1–19. https://doi.org/10.1186/s40538-021-00237-4
Pick U, Rental M, Chitlaru E, Weiss M (1990) Polyphosphate-hydrolysis-a protective mechanism against alkaline stress. FEBS Lett 274:15–18. https://doi.org/10.1016/0014-5793(90)81318-I
Sadeghi A, Karimi E, Dahaji PA, Javid MG, Dalvand Y, Askari H (2012) Plant growth promoting activity of an auxin and siderophore producing isolate of Streptomyces under saline soil conditions. World J Microbiol Biotechnol 28:1503–1509. https://doi.org/10.1007/s11274-011-0952-7
Saravanakumar D, Samiyappan R (2007) ACC deaminase from Pseudomonas fluorescens mediated saline resistance in groundnut (Arachis hypogea) plants. J Appl Microbiol 102:1283–1292. https://doi.org/10.1111/j.1365-2672.2006.03179.x
Shultana R, Kee Zuan AT, Yusop MR, Saud HM (2020) Characterization of salt-tolerant plant growth-promoting rhizobacteria and the effect on growth and yield of saline-affected rice. PLoS ONE 15:e0238537. https://doi.org/10.1371/journal.pone.0238537
Sritongon N, Sarin P, Theerakulpisut P, Riddech N (2022) The effect of salinity on soil chemical characteristics, enzyme activity and bacterial community composition in rice rhizospheres in Northeastern Thailand. Sci Rep 12:20360. https://doi.org/10.1038/s41598-022-24902-2
Sultana S, Alam S, Karim MM (2021) Screening of siderophore-producing salt-tolerant rhizobacteria suitable for supporting plant growth in saline soils with iron limitation. J Agric Food Res 4:100150. https://doi.org/10.1016/j.jafr.2021.100150
Tavakkoli E, Fatehi F, Coventry S, Rengasamy P, McDonald GK (2011) Additive effects of Na+ and Cl– ions on barley growth under salinity stress. J Exp Bot 62:2189–2203. https://doi.org/10.1093/jxb/erq422
Tomaz A, Palma P, Alvarenga P, Gonçalves MC (2020) Soil salinity risk in a climate change scenario and its effect on crop yield. Climate change and soil interactions. Elsevier, Dutch, pp 351–396. https://doi.org/10.1016/B978-0-12-818032-7.00013-8
Turan V (2020) Potential of pistachio shell biochar and dicalcium phosphate combination to reduce pb speciation in spinach, improved soil enzymatic activities, plant nutritional quality, and antioxidant defense system. Chemosphere 245:125611. https://doi.org/10.1016/j.chemosphere.2019.125611
Turan V (2021) Arbuscular mycorrhizal fungi and pistachio husk biochar combination reduces Ni distribution in mungbean plant and improves plant antioxidants and soil enzymes. Physiol Plant 173:418–429. https://doi.org/10.1111/ppl.13490
Turan V, Aydın S, Sönmez O (2022) Production, cost analysis, and marketing of bioorganic liquid fertilizers and plant nutrition enhancers. Industrial microbiology-based entrepreneurship: making money from microbes. Springer Nature, Singapore, pp 193–198. https://doi.org/10.1007/978-981-19-6664-4_13
Vimal SR, Patel VK, Singh JS (2019) Plant growth promoting Curtobacterium albidum strain SRV4: an agriculturally important microbe to alleviate salinity stress in paddy plants. Ecol Indic 105:553–562. https://doi.org/10.1016/j.ecolind.2018.05.014
Wang Q, Dodd IC, Belimov AA, Jiang F (2016) Rhizosphere bacteria containing 1-aminocyclopropane-1-carboxylate deaminase increase growth and photosynthesis of pea plants under salt stress by limiting Na+ accumulation. Funct Plant Biol 43:161–172. https://doi.org/10.1071/FP15200
Youssef MMA, Eissa MFM (2014) Biofertilizers and their role in management of plant parasitic nematodes. A review. J Biotechnol Pharm Res 5:1–6
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
NA: designed the study; SC: performed the experiment and analyzed the data; SC and NA: drafted, reviewed, edited and finalized the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chandwani, S., Amaresan, N. Siderophore and ACC Deaminase Producing Bacteria Enhance the Growth of Vigna spp Under Iron Limited Saline Soils. J Soil Sci Plant Nutr (2024). https://doi.org/10.1007/s42729-024-01795-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42729-024-01795-w