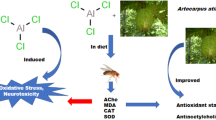
Abstract
Buchholzia coriacea is known for its anti-oxidative, anti-inflammatory, and anti-diabetic properties. This study evaluated the effect of alkaloid-rich extract of Buchholzia coriacea seed (ArEBCs) against copper-induced toxicity in Drosophila melanogaster. The flies were exposed to graded doses (25 to 200 mg/kg diet) of ArEBCs for 12 days and thereafter, a survival test was carried out to determine the safe and therapeutic doses used in the ameliorative study. For this, flies were exposed to CuSO4 (1 mM) alone or in combination with ArEBCs (25 and 50 mg/kg diet) for 5 days. Locomotor activity and biochemical assays such as glucose (GLU) level, acetylcholinesterase (AChE) activity, malonaldehyde (MDA) and antioxidant biomarkers (catalase and reduced glutathione) were performed to assess the effect of ArEBCs on the copper-induced flies. Copper-exposed flies showed reduced survival rates and disrupted locomotor activity when compared to the control flies. In addition, a significant (p < 0.05) increase in AChE activity, GLU and MDA levels and a decrease in the antioxidant biomarkers activities were observed in the copper-exposed flies. Treatment of flies with ArEBCs mitigated the negative effect of copper on the flies by increasing the survival rate of the flies, reducing the activities of AChE, GLU and MDA. The activities of the cellular antioxidant status were also significantly (p < 0.05) increased. This study suggests that ArEBCs could mitigate the effect of copper-induced oxidative damage in Drosophila melanogaster and could be considered a potential therapeutic agent against copper-induced oxidative damage.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Copper (Cu2+) is an important transition element naturally found in the environment and involved in many biochemical processes. It has an oxidation number of + 1 or + 2 depending on its chemical structure. However, in its natural state, the oxidation number is zero (Mustafa and AlSharif 2018). Copper serves as a cofactor in several enzymatic processes involving energy production (cytochrome-c oxidase), pigmentation (tyrosinase), cellular iron transfer (ferroxidase), antioxidant defence (superoxide dismutase), and connective tissue formation (lysyl oxidase) (Gromadzka et al. 2020; Ruiz et al. 2021). Free form of copper is potentially damaging; therefore, its absorption and excretion are tightly regulated in the body (Chen et al. 2022b). Under physiological conditions, the amount of Cu2+ in the cell is tightly regulated, however, excess of it can lead to oxidative stress (OS), mitochondrial dysfunction, hepatic disorders, inflammation, and neurodegenerative diseases (Taylor et al. 2020; Tassone et al. 2021). Increased free Cu2+ levels are highly reactive, and catalyses the Haber-Weiss reaction thereby, promoting the production of hydroxyl radicals which influence the pathogenesis of various diseases (Das et al. 2015). Several literatures have reported the toxicity associated with Cu2+. These include neuropathy, myelopathy, anaemia, and neutropenia (Jaiser and Winston 2010; Lazarchick 2012).
Medicinal plants with antioxidant properties have been reported to inhibit or ameliorate the toxic effect of copper in various experimental studies (Klimaczewski et al. 2018). Medicinal plants possess various phytochemicals which are responsible for their biological activities (Ugboko et al. 2020).
Buchholzia coriacea Engl. (B. coriacea (Capparaceae) known as “wonderful kola’’ is common to the Western regions of Africa including Nigeria and Cameroon. The tree is usually very tall with large glossy leathery leaves bearing a bright cream-white flower. The seed presents the same features as the kola nut, but bigger. It has a pungent taste with a hot spicy flavour (Obembe 2021). In some parts of Nigeria, the plant is known for its native names such as “Oji ogwu” (Igbo) and “Owi” (Bini)’’. The cooked seed is consumed as a snack in Edo State, Nigeria. The plant is widely used in the African traditional medicine practice for the treatment of hypertension, diabetes, psychiatric disorders, and impotence (Izah et al. 2018). The leaves and seeds of the plant have shown great pharmacological potentials including anti-microbial, hypoglycaemic, aphrodisiac, and antimalarial properties (Izah et al. 2018; Kigigha et al. 2018). The bioactive constituents of the plant include saponins, flavonoids, anthraquinone, tannins, cardiac glycosides, steroids, alkaloids, resins, and carbohydrates (Izah et al. 2018). The presence of these phytochemicals exhibits scavenging activities against free radicals. Alkaloids are considered efficient therapeutic constituents of a plant because of their pharmacological properties which include analgesic, antispasmodic, neuroprotective and anti-inflammatory activities (Hussain et al. 2018; Heinrich et al. 2021). Alkaloids have been reported to inhibit the cu2+ toxicity (Huynh 2004). However, to the best of our knowledge, no study has reported the alkaloid-rich fraction of B. coriacea (ArEBCs) to have a protective effect against metal-induced oxidative damage in vivo.
Drosophila melanogaster (fruit fly) has been employed as an alternative animal model in different areas of biological research. This model support the 3 R’s (refinement, replacement, reduction) rule approved by the European Centre for Validation of Alternative Model to lessen the burden associated with the ethical restriction on the number of experimental animals used in research. More so, the nutrient uptake, storage, and metabolism of D. melanogaster are similar to those of humans (Chatterjee and Perrimon 2021). About 75% of genes causing diseases in humans are seen in D. melanogaster, therefore, they are used as a model organism for human diseases (Jennings 2011). Drosophila melanogaster model has also been used to study the toxic effects of compounds including heavy metals (Everman et al. 2021). Copper has high metabolic rate and cell turnover to most organs in Drosophila melanogaster (D. melanogaster) (Everman et al. 2021). Excessive (toxic level) Cu2+ can interrupt the life cycle of flies, as well as disrupt the locomotive activity of the flies (Halmenschelager and da Rocha 2019). Therefore, investigations regarding survival, behaviour and neuronal functions, biochemical and molecular functions are easily performed using this organism model. Oxidative stress and its complications such as diabetes, cancer and neurotoxicity are still a major concern worldwide. In view of this, several approaches need to be explored in combating the deleterious effect. This study, therefore, evaluated the effect of ArEBCs against copper-induced toxicity in D. melanogaster.
Materials and methods
Chemicals and reagents
Analytical grade chemicals such as copper sulphate, acetylthiocholine iodine and glutathione were products of Sigma Aldrich Co. (St Louis, Missouri, USA). Acetic acid, n-hexane and ethanol were products of BDH Chemicals Ltd. (Poole, England).
Plant source and preparation of plant extract
Plant source
Fresh seeds of B. coriacea were obtained from Kings Market Uromi, Edo State. The seeds were thoroughly washed in a clean water, wiped with clean paper towel and shredded into thin pieces. The shredded pieces were air-dried for 14 days and then blended into a fine powder. The powdered sample was air-tight and stored in a plastic container till needed.
Preparation of alkaloid-rich extract
The method described by Oboh et al. (2018) was employed in the extraction of alkaloid-rich portions of B. coriacea seeds. Briefly, 100 g of the powdered sample was soaked in a cold n-hexane (defatted) at room temperature for 24 h. Two hundred (200) mL of 10% alcoholic-acetic acid (v/v) was added to the defatted sample and then was continuously stirred for 72 h. The mixture was filtered, and the filtrate concentrated at 45 °C. Concentrated ammonium hydroxide was added to the filtrate in a drop wise manner till a precipitate was formed. The sample was allowed to settle, and the mixture was separated into two layers. The precipitate was further rinsed with dilute ammonium hydroxide to obtain the ArEBCs.
Experimental animal
Drosophila melanogaster (Harwich strain) flies were sourced from the Drosophila Laboratory, Biochemistry program, Department of Chemical Science, Afe Babalola University Ado Ekiti (ABUAD), Nigeria. The flies were fed on a corn meal medium containing 1% w/v brewer’s yeast, 1% w/v agar, and 0.1% v/w methylparaben. The flies were maintained at 24 ± 2 °C and relative humidity (45–60%) under photoperiod time of 12 h light and 12 h darkness.
Experimental design
Both sexes of flies (3–5 days old) were assigned into five groups, (n = 4 independent vials; 50 flies per vial) and were exposed to ArEBCs (0–200 mg/kg diet) for 12 days (survival test). The flies were kept under daily observation for the number of mortalities and changes in the behavioural activities of the flies. The survival rate of the flies was determined by counting the number of dead flies for 12 days. The ArEBCs (25 and 50 mg/kg diet) used for the ameliorative study were choices made from the data obtained from the survival study. Flies were assigned into four groups, (n = 4 independent vial; 50 flies per vial) and were exposed to CuSO4 (1mM) and ArEBCs supplemented basal diet for 5 days as shown below (Table 1):
The CuSO4 (1mM) was an IC50 value dose adopted from a study by Abolaji et al. (2020). All experiments were performed in triplicate.
Negative geotaxis measurement
The locomotor activity (negative geotaxis) of the control and treated flies was determined using the method described by Adedara et al. (2016). Briefly, 10 immobilized (ice) flies from each group were transferred to a clean and sterilized (70% alcohol) 50 ml vial. The vials were placed on the bench for 5 min for the flies to recover from immobilization and then were gently tapped at the bottom to wake the flies (active state). The locomotor activity of the flies was recorded by counting the flies that successfully crossed the 6 cm marked line on the vial within 6 s.
Preparation of homogenate for biochemical assays
Immobilized flies were transferred to a clean Eppendorf tube, weighed, and homogenized in 0.1 M phosphate buffer, pH 7.4. The homogenates were centrifuged at 4000 × g for 10 min at 4 °C and the supernatants were transferred to new tubes and stored in a − 20 °C freezer till needed.
Biochemical parameter
The following biochemical analyses were carried out in the whole organism’s homogenate: glucose (GLU) level was evaluated using the method by Barham and Trinder (1972). The nitric oxide level was measured using the Greiss reaction Green et al. (1982). Lipid peroxidation was determined using the described method by Varshney and Kale (1990). Total thiols, catalase, and AChE activities were evaluated using the described methods by Ellman (1959), Claiborne (2018) and Ellman et al. (1961), respectively.
Statistical analysis
Statistical analysis was performed using one-way analysis of variance (ANOVA). Multiple comparison of intergroup variables was performed using the Tukey post hoc comparison test on Graphpad Prism version 8.1. Results were expressed as mean ± standard deviation (SD), n = 4. p < 0.05 was considered statistically significant in all cases.
Results
Effect of alkaloid-rich extract of Buchholzia coriacea seed on the survival rate of flies
Figure 1a shows the survival rate of flies fed with basal diet supplemented with ArEBCs at doses of 0 to 200 mg/kg diet. Statistically, no significant (p > 0.05) changes were observed in the flies treated with the different doses of ArEBCs supplemented diet when compared with the control. In Fig. 1b, a significant (p < 0.05) decrease in the survival rate of flies was observed in the D. melanogaster fed with basal diet supplemented with CuSO4 when compared to the control and ArEBCs supplemented-diet fed D. melanogaster.
Negative geotaxis (locomotor) activity of D. Melanogaster exposed to copper
Exposure of flies to CuSO4 significantly (p < 0.05) decreased the % climbing activity of the flies as shown in Fig. 2. Treatment of flies with ArEBCs (50 mg/kg diet) however, restored the locomotive movement of the flies.
Legend: CuSO4-copper sulphate; ArEBCs-alkaloid-rich extract of Buchholzia coriacea seeds.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 significantly different from the copper group.
Biochemical parameters
Alkaloid-rich extract of Buchhlozia coriacea seeds restored glucose level in copper-induced toxicity in D. Melanogaster
Figure 3 reveals the effect of copper and ArEBCs on glucose level in D. melanogaster. The glucose levels of flies exposed to copper fortified diet was significantly (p < 0.05) increased when compared with the control. However, treatment of flies with ArEBCs (25 and 50 mg/kg diet) significantly (p < 0.05) restored the elevated glucose level implicated by copper comparatively to the control.
Legend: CuSO4-copper sulphate; ArEBCs-alkaloid-rich extract of Buchholzia coriacea seeds.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 when significantly different from the copper group.
3.3.2 Alkaloid-rich extract of B. coriacea mitigates nitric oxide level in copper-induced toxicity in D. Melanogaster
Figure 4 shows the effect of ArEBCs and copper on the nitric oxide levels of D. melanogaster. The level of nitric oxide in copper-induced flies was significantly (p < 0.05) increased when compared to the control flies. However, treatment of flies with ArEBCs supplemented diet significantly (p < 0.05) decreased the level of nitric oxide when compared with the copper-induced group.
Legend: CuSO4-copper sulphate; ArEBCs-alkaloid-rich extract of Buchholzia coriacea seeds.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 significantly different from the copper group.
Alkaloid-rich extract of B. Coriacea reduced malondialdehyde levels in copper-induced D. Melanogaster
As shown in Fig. 5, flies exposed to copper showed a significant (p < 0.05) increase in MDA level when compared with the control flies. However, co-treatment with ArEBCs (25 and 50 mg/kg diet) supplemented diet significantly (p < 0.05) reduced the elevated MDA level caused by copper.
Legend: CuSO4-copper sulphate; ArEBCs-alkaloid-rich extract of Buchholzia coriacea seeds.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 significantly different from the copper group.
Alkaloid-rich extract of B. coriacea restores catalase activity in copper-induced toxicity in D. Melanogaster
Figure 6 depicts the effect of ArEBCs on catalase activity in copper-induced D. melanogaster. Copper significantly (p < 0.05) reduced the catalase activity of flies when compared with the control flies. Co-treatment with doses of ArEBCs (25 and 50 mg/kg diet) restored the enzyme activity of the flies.
Legend: CuSO4-copper sulphate; ArEBCs-alkaloid-rich extract of Buchholzia coriacea seed.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 significantly different from the copper group.
Alkaloid-rich extract of B. coriacea reverse total thiol levels in copper-induced toxicity in D. melanogaster
Figure 7 displays the effect of ArEBCs and copper on the total thiol level in D. melanogaster. In comparison with the control, copper significantly (p < 0.05) reduced the total thiol levels in the flies. However, co-treatment with ArEBCs supplemented diet significantly (p < 0.05) increased the total thiol levels when compared with the copper-induced group.
Legend: CuSO4-copper sulphate; ArEBCs-alkaloid-rich extract of Buchholzia coriacea seeds.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 significantly different from the copper group.
Alkaloid-rich extract of B. coriacea reversed reduced glutathione level in copper-induced toxicity in D. melanogaster
Figure 8 shows the effect of ArEBCs on reduced glutathione levels in copper-induced toxicity in D. melanogaster. Comparably with the control, the GSH level was significantly (p < 0.05) reduced in the copper-induced flies. Treatment with ArEBCs however, mitigated the negative effect of copper in the flies.
Legend: CuSO4 - copper sulphate; ArEBCs - alkaloid-rich extract of Buchholzia coriacea.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 significantly different from the copper group.
Neuroprotective role of alkaloid-rich extract of B. coriacea in copper-induced toxicity in D. melanogaster
Effect of ArEBCs on acetylcholinesterase activity in copper-induced toxicity in D. melanogaster. Comparatively, with the control, copper significantly (p < 0.05) increased the acetylcholinesterase activity in flies. However, co-treatment with the ArEBCs (50 mg/kg diet) mitigated the toxic effect of copper in the flies (Fig. 9).
Legend: CuSO4-copper sulphate; AChE-acetylcholinesterase activity ArEBCs-alkaloid-rich extract of Buchholzia coriacea seeds.
Data are expressed as mean ± SD (n = 4 replicates and 50 flies per vial); ap < 0.05 significantly different from the control; bp < 0.05 significantly different from the copper group.
Discussion
Copper is one of the friendliest metals found in the environment with several biological functions including promoting growth, lung elasticity, neuroendocrine function, and iron metabolism. However, exposure to copper in excessive amounts can result in harmful effects (Taylor et al. 2020). Like other experimental animals, D. melanogaster is considered a model to study various biological diseases and the toxicity of chemical elements. This is because D. melanogaster has high reproductive rate, and can easily be genetically manipulated (Walters et al. 2020; Everman et al. 2021). In addition, D. melanogaster model is cost-effective, easy to maintain and validates the use of 3Rs in biological research (Rocha 2013). Buchholzia coriacea is known for its pharmacological activities. Taking the evidence regarding its potency into consideration, we sought to evaluate its effect against copper-induced toxicity using the D. melanogaster model.
Flies in this study were exposed to a basal diet supplemented with ArEBCs at different doses to establish a safe dose for the ameliorative study. The non-significant changes observed in the flies exposed to the different doses of ArEBCs suggest that the plant extract was non-toxic to the flies. For the ameliorative study, CuSO4 caused drastic decline in the survival rates of the flies. This was however, not the case for the flies treated with ArEBCs as no significant decrease was noted in the survival rate of the flies, thus, suggesting that the ArEBCs could have a protective effect against copper toxicity. The decreased locomotor activity of the flies exposed to CuSO4 indicates that copper caused impaired locomotor in the flies. In addition, the copper-exposed flies exhibit more mortality which may be due to OS. Abolaji et al. (2020) reported similar findings where copper caused impairment in the upward movement of the flies thereby affecting their climbing ability. Furthermore, the flies treated with ArEBCs (25 and 50 mg/kg diet) showed that the life expectancy of the flies was maintained, and the impaired locomotive activity was restored. Therefore, suggesting that the ArEBCs could possess neuroprotective potential and were able to scavenge free radicals generated due to copper toxicity.
Excessive intake of copper can promote the generation of OS, which is one of the risk factors in the development of type II diabetes mellitus (Eljazzar et al. 2023). Poor glycaemic control has been relatively linked to high levels of copper in the serum (Cui et al. 2022). Abnormal Cu metabolism could result in type II diabetes as well as disrupt antioxidant homeostasis (Gembillo et al. 2022). The elevated glucose level observed in this study on exposure of the flies to Cu was significantly reduced by the administration of ArEBCs suggesting that the ArEBCs possess a protective effect against oxidative damage instigated by copper toxicity.
Nitric oxide is a natural gas produced by the human body. Its diffusion rate is very high and has a short half-life and therefore, can easily be converted to more stable metabolites, nitrite, and nitrate (Oyetayo et al. 2020). Nitric oxide is a pro-inflammatory mediator that interacts with superoxide anion to produce free radicals which leads to organ toxicity (Abolaji et al. 2020). In this study, the ArEBCs were able to mitigate the toxicity caused by copper on the flies, thus suggesting the protective effect of the ArEBCs on the flies.
The increased MDA levels observed in flies exposed to copper were drastically reduced by the ArEBCs, therefore, it can be suggested that the ArEBCs modulated the levels of MDA, and thus, reduced the free radicals generated. This observation confirms that the B. coriacea plant demonstrates antioxidant and radical scavenging ability (Fakoya and Olusola 2020).
Catalase, a heme-containing enzyme is responsible for catalysing hydrogen peroxide to oxygen and water (Abolaji et al. 2020). Reduced level of catalase in flies fed with copper suggests a compromised antioxidant status, thereby increasing the level of hydrogen peroxide and ultimately inducing OS. The ArEBCs were able to assuage the oxidative damage caused by copper, therefore, suggesting the scavenging ability of the plant extract.
Reduced glutathione (GSH) is a non-enzymatic antioxidant found in high concentrations in the cellular system. It plays a role in the detoxification of xenobiotics via electrophilic molecules (Averill-Bates 2023). Low level of GSH exposes cells to OS attack. In this study, the reduced level of GSH caused by copper induction was notably elevated by ArEBCs, thus, suggesting that the ArEBCs protected the organs in the D. melanogaster from OS by enhancing the cellular GSH level.
Acetylcholinesterase is an enzyme that breaks down acetylcholine, a neurotransmitter involved in the regulation of motor function and neuronal activities of the brain to acetic acid and choline (Anadozie et al. 2019; Chen et al. 2022b). In this study, the ArEBCs ameliorated the elevated acetylcholinesterase activity caused by exposure of flies to copper. This suggests that the ArEBCs have neuroprotective ability. This also complements the result of locomotor activity of this study where ArEBCs restored the impaired locomotor activity caused by copper toxicity.
Alkaloid-rich extract of Buchholzia coriacea has demonstrated antioxidant, neuroprotective and radical scavenging effects against copper-induced toxicity in D. melanogaster by significantly, enhancing the various biochemical parameters assayed in this study.
Conclusion
In conclusion, this study showed that ArEBCs possesses antioxidant activity and could protect the flies from oxidative damage caused by copper toxicity. The ArEBCs restored the locomotive activity, antioxidant status and neuronal activity of the copper-fed flies. Alkaloid-rich extract of Buchholzia coriacea could be considered a potential therapeutic agent in the prevention and management of diseases associated with metal toxicity.
Data availability
All data supporting this research will be made available on request.
References
Abolaji AO, Fasae KD, Iwezor CE, Aschner M, Farombi EO (2020) Curcumin attenuates copper-induced oxidative stress and neurotoxicity in Drosophila melanogaster. Toxicol Rep 7:261–268
Adedara IA, Abolaji AO, Rocha JB, Farombi EO (2016) Diphenyl diselenide protects against mortality, locomotor deficits and oxidative stress in Drosophila melanogaster model of manganese-induced neurotoxicity. Neurochem Res 41(6):1430–1438. https://doi.org/10.1007/s11064-016-1852-x
Anadozie SO, Akinyemi JA, Adewale OB, Isitua CC (2019) Prevention of short-term memory impairment by Bryophyllum pinnatum (lam.) Oken and its effect on acetylcholinesterase changes in ccl4-induced neurotoxicity in rats. J Basic Clin Physiol Pharmacol 30:5. https://doi.org/10.1515/jbcpp-2018-0161
Averill-Bates DA (2023) Chapter five—the antioxidant glutathione. Vitamins and hormones. G Litwack Acad Press 121:109–141
Barham D, Trinder P (1972) An improved colour reagent for the determination of blood glucose by the oxidase system. Analyst 97(1151):142–145
Chatterjee N, Perrimon N (2021) What fuels the fly: energy metabolism in drosophila and its application to the study of obesity and Diabetes. Sci Adv 7:24. https://doi.org/10.1126/sciadv.abg4336
Chen Z-R, Huang J-B, Yang S-L, Hong F-F (2022) Role of cholinergic signaling in Alzheimer’s disease. Molecules 27(6):1816
Chen L, Min J, Wang F (2022) Copper homeostasis and cuproptosis in health and disease. Signal Transduct Target Ther 7(1):378. https://doi.org/10.1038/s41392-022-01229-y
Claiborne A (2018) Catalase activity. CRC handbook of methods for oxygen radical research. CRC Press, Boca Raton, Florida, pp 283–284
Cui Z, Chen H, Lu W, Wang P, Zhou Z, Zhang N, Wang Z, Lin T, Song Y, Liu L (2022) The association between plasma copper concentration and prevalence of diabetes in Chinese adults with hypertension. Front Public Health 10:888219. https://doi.org/10.3389/fpubh.2022.888219
da Halmenschelager PT (2019) Biochemical cuso 4 toxicity in Drosophila melanogaster depends on sex and developmental stage of exposure. Biol Trace Elem Res 189:574–585
Das TK, Wati MR, Fatima-Shad K (2015) Oxidative stress gated by Fenton and haber weiss reactions and its association with Alzheimer’s disease. Arch Neurosci 2:2
Eljazzar S, Abu-Hijleh H, Alkhatib D, Sokary S, Ismail S, Al-Jayyousi GF, Tayyem R (2023) The role of copper intake in the development and management of type 2 diabetes: a systematic review. Nutrients 15:1655
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82(1):70–77
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Everman ER, Cloud-Richardson KM, Macdonald SJ (2021) Characterizing the genetic basis of copper toxicity in drosophila reveals a complex pattern of allelic, regulatory, and behavioral variation. Genetics 217(1):1–20. https://doi.org/10.1093/genetics/iyaa020
Fakoya A, Olusola A (2020) Transcriptional evaluation of antioxidant and anti-inflammatory potential of Buchholzia coriacea in acetaminophen-induced sub-chronic renal and hepatic toxicity. J Biochem Anal Stud 4:3
Gembillo G, Labbozzetta V, Giuffrida AE, Peritore L, Calabrese V, Spinella C, Stancanelli MR, Spallino E, Visconti L, Santoro D (2022) Potential role of copper in diabetes and diabetic kidney Disease. Metabolites 13:17
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15n] nitrate in biological fluids. Anal Biochem 126(1):131–138
Gromadzka G, Tarnacka B, Flaga A, Adamczyk A (2020) Copper dyshomeostasis in neurodegenerative diseases-therapeutic implications. Int J Mol Sci. https://doi.org/10.3390/ijms21239259
Heinrich M, Mah J, Amirkia V (2021) Alkaloids used as medicines: structural phytochemistry meets biodiversity—an update and forward look. Molecules 26(7):1836
Hussain G, Rasul A, Anwar H, Aziz N, Razzaq A, Wei W, Ali M, Li J, Li X (2018) Role of plant derived alkaloids and their mechanism in neurodegenerative disorders. Int J Biol Sci 143:341
Huynh NH (2004) The inhibition of copper corrosion in aqueous environments with heterocyclic compounds. Doctoral dissertation, Queensland University of Technology
Izah SC, Uhunmwangho E, Eledo BO (2018) Medicinal potentials of Buchholzia coriacea (wonderful kola). Med Plant Res 8
Jaiser SR, Winston GP (2010) Copper deficiency myelopathy. J Neurol 257(6):869–881. https://doi.org/10.1007/s00415-010-5511-x
Jennings BH (2011) Drosophila – a versatile model in biology & medicine. Mater Today 14(5):190–195. https://doi.org/10.1016/S1369-7021(11)70113-4
Kigigha LT, Selekere RE, Izah SC (2018) Antibacterial and synergistic efficacy of acetone extracts of Garcinia kola (bitter kola) and Buchholzia coriacea (wonderful kola). J Basic Pharmacol Toxicol 2(1):13–17
Klimaczewski CV, Ecker A, Piccoli B, Aschner M, Barbosa NV, Rocha JBT (2018) Peumus boldus attenuates copper-induced toxicity in Drosophila melanogaster. Biomed Pharmacother 97:1–8
Lazarchick J (2012) Update on anemia and neutropenia in copper deficiency. Curr Opin Hematol 19(1):58–60
Mustafa SK, AlSharif MA (2018) Copper (cu) an essential redox-active transition metal in living system—a review article. Am J Anal Chem 9(01):15
Obembe OO (2021) Buchholzia coriacea (wonderful kola) seeds induce male reproductive toxicity by suppressing the pituitary-gonadal axis in Wistar rats. Braz J Pharm Sci 57:e19016
Oboh G, Ogunsuy OB, Awonyemi OI, Atoki VA (2018) Effect of alkaloid extract from African jointfir (Gnetum africanum) leaves on manganese-induced toxicity in Drosophila melanogaster. Oxid Med Cell Longev 2018: 8952646. https://doi.org/10.1155/2018/8952646
Oyetayo BO, Abolaji AO, Fasae KD, Aderibigbe A (2020) Ameliorative role of diets fortified with curcumin in a Drosophila melanogaster model of aluminum chloride-induced neurotoxicity. J Funct Foods 71:104035. https://doi.org/10.1016/j.jff.2020.104035
Rocha J (2013) Drosophila melanogaster as a promising model organism in toxicological studies. Arch Basic Appl Med 1(1):33–38
Ruiz LM, Libedinsky A, Elorza AA (2021) Role of copper on mitochondrial function and metabolism. Front Mol Biosci 8:711227
Tassone G, Kola A, Valensin D, Pozzi C (2021) Dynamic interplay between copper toxicity and mitochondrial dysfunction in alzheimer’s Disease. Life (Basel) 11:5. https://doi.org/10.3390/life11050386
TaylorAA, Tsuji JS, Garry MR, McArdle ME, Goodfellow WL, Jr-Adams WJ, Menzie CA (2020) Critical review of exposure and effects: implications for setting regulatory health criteria for ingested copper. Environ Manage 65(1):131–159. https://doi.org/10.1007/s00267-019-01234-y
Ugboko HU, Nwinyi OC, Oranusi SU, Fatoki TH, Omonhinmin CA (2020) Antimicrobial importance of medicinal plants in nigeria. Sci World J
Varshney R, Kale R (1990) Effects of calmodulin antagonists on radiation-induced lipid peroxidation in microsomes. Int J Radiat Biol 58(5):733–743
Walters AW, Hughes RC, Call TB, Walker CJ, Wilcox H, Petersen SC, Rudman SM, Newell PD, Douglas AE, Schmidt PS (2020) The microbiota influences the drosophila melanogaster life history strategy. Mol Ecol 29(3):639–653
Acknowledgements
The authors are grateful to the Founder and management of Afe Babalola University Ado Ekiti for assisting with the APC fee and for Dr A.O Abolaji for providing the flies used in this study.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
SOA: Conceptualized, draft the original manuscrpit, provided resources and supervised the project, AUA: performed analysis. OBA: supervised, provided resources and performed formal analysis. All authors read and approved the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors wish to declare that there is no competing interest related to this work.
Ethical approval
Ethical approval was not applicable in this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Anadozie, S.O., Aduma, A.U. & Adewale, O.B. Alkaloid-rich extract of Buchholzia coriacea seed mitigate the effect of copper-induced toxicity in Drosophila melanogaster. Vegetos 37, 460–468 (2024). https://doi.org/10.1007/s42535-023-00760-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42535-023-00760-9