Abstract
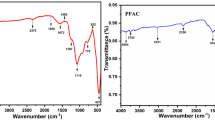
Adsorption of hexavalent chromium from the wastewater by activated carbon (AC) synthesized from biomass was extensively studied. The AC was prepared by a cost-effective way from abundantly available biomass IPOMOEA CARNEA (morning glory). The adsorption behaviors of Cr (VI) containing wastewater from chrome plating industry in the range of 100 to 300 mg/L, on biomass were systematically investigated. The formation, morphology, structure, and surface areas were analyzed by FTIR, SEM-EDS, XRD analytical techniques thus to substantiate the suitability of the adsorbent material. Batch experiments studies were carried out by varying adsorptions factors including contact time, temperature, pH, adsorbent dose, initial concentration etc., to resolve the adsorption properties of the AC prepared from biomass. The optimum dosage of 50 g/L at pH 3.5 and contact time 90 min was found to be suitable for adsorptive removal complying the government of India discharge standard of 0.1 and 2 mg/L for hexavalent chromium and total chromium respectively. Kinetically the Freundlich and Langmuir isotherms were found to be suitable for the purpose. The absorption data set closely resembles with Langmuir and Freundlich isotherm. Thus, the biomass IPOMOEA CARNEA used in this work proved to be effective adsorbent materials for the treatment of chromium bearing wastewater.









Similar content being viewed by others
References
Saha R, Nandi R, Saha B Sources and toxicity of hexavalent chromium (2011).J. Coord. Chem.64:1782–1806
Pan C, Troyer LD, Liao P, Catalano JG, Li W, Giammar DE (2017) Effect of humic acid on the removal of chromium (VI) and the production of solids in iron electrocoagulation. Environ Sci Technol 51:6308–6318
Mondal MH, Begum W, Nasrollahzadeh M, Ghorobannezhad F, Antoniadis V, Levizou E, Saha (2021) A comprehensive review on chromium chemistry along with detection speciation, extraction and remidiation of hexavalent chromium in contemprorary science and technology. Vietnam J Chem 59:711–732
Amin S, Abbas M, Tahir A, Ghani N, Abrar A, Aslam F, Ahmad S (2022) SEM and XRD for removal of heavy metals from industrial wastewater and characterization of chicken eggshell. Microscopy Res Technique 85:2587–2595
Zhang X, Ren B, Wu X, Yan X, Sun Y, Gao H, Qu F (2021) Efficient removal of chromium (VI) using a novel waste biomass chestnut shell-based carbon electrode by electrosorption. ACS omega 6:25389–25396
Dong H, Zhang L, Shao L, Wu Z, Zhan P, Zhou X, Chen J (2022) Versatile Strategy for the Preparation of Woody Biochar with Oxygen-Rich Groups and Enhanced Porosity for Highly Efficient Cr (VI) Removal. ACS omega 7:863–874
Qi W, Zhao Y, Zheng X, Ji M, Zhang Z (2016) Adsorption behavior and mechanism of Cr (VI) using Sakura waste from aqueous solution. Appl Surf Sci 360:470–476
Belcaid A, Beakou B, El Hassani K, Bouhsina S, Anouar A (2021) Efficient removal of Cr (VI) and Co (II) from aqueous solution by activated carbon from Manihot esculenta Crantz agricultural bio-waste. Water Sci Technol 83:556–566
Khazaei I, Aliabadi M, HAMED MH (2011) Use of agricultural waste for removal of Cr (VI) from aqueous solution. Iran J Chem Eng 8:11–23
Mouhamadou S, Dalhatou S, Dobe N, Djakba R, Fasanya OO, Bansod ND, Fita G, Ngayam CH, Tejeogue JPN, Harouna M (2022) Linear and Non-linear Modelling of Kinetics and Equilibrium Data for Cr(VI) Adsorption by Activated Carbon Prepared from Piliostigma reticulatum. Chem Afr. doi.https://doi.org/10.1007/s42250-022-00324-5
Yang J, Yu M, Chen W (2015) Adsorption of hexavalent chromium from aqueous solution by activated carbon prepared from longan seed: Kinetics, equilibrium and thermodynamics. J Ind Eng Chem 21:414–422
Kumar A, Kumar V (2022) A comprehensive review on application of lignocellulose derived nanomaterials in heavy metals removal from waste water. Chemistry Africa. https://doi.org/10.1007/s42250-022-00367-8
Javadian H, Ahmadi M, Ghiasvand M, Kahrizi S, Katal R (2013) Removal of Cr (VI) by modified brown algae Sargassum bevanom from aqueous solution and industrial wastewater. J Taiwan Inst Chem Eng 44:977–989
Yadav S, Srivastava V, Banerjee S, Weng C-H, Sharma YC (2013) Adsorption characteristics of modified sand for the removal of hexavalent chromium ions from aqueous solutions: Kinetic, thermodynamic and equilibrium studies. CATENA 100:120–127
Chen F, Guo S, Wang Y, Ma L, Li B, Song Z, Huang L, Zhang W (2022) Concurrent adsorption and reduction of chromium(VI) to chromium(III) using nitrogen-doped porous carbon adsorbent derived from loofah sponge. Front Environ Sci Eng 16(5):57. https://doi.org/10.1007/s11783-021-1491-6
Malkoc E, Nuhoglu Y (2007) Potential of tea factory waste for chromium (VI) removal from aqueous solutions: Thermodynamic and kinetic studies. Sep Purif Technol 54:291–298
Mondal MH, Malik S, Garain A, Mandal S, Saha B (2017) Extraction of Natural Surfactant Saponin from Soapnut (Sapindus mukorossi) and its Utilization in the Remediation of Hexavalent Chromium from Contaminated Water. Tenside Surfactants Detergent 54:519–529
Narayan R, Meena RP, Patel AK, Prajapati AK, Srivastava S, Mondal MK (2016) Characterization and application of biomass gasifier waste material for adsorptive removal of Cr (VI) from aqueous solution. Environ Prog Sustain Energy 35:95–102
Anupam K, Dutta S, Bhattacharjee C, Datta S (2011) Adsorptive removal of chromium (VI) from aqueous solution over powdered activated carbon: Optimisation through response surface methodology. Chem Eng J 173:135–143
Pathania D, Sharma A, Srivastava A (2020) Modelling studies for remediation of Cr (VI) from wastewater by activated Mangifera indica bark. Curr Res Green Sustainable Chem 3:100034
Deng M, Wang X, Li Y, Wang F, Jiang Z, Liu Y, Gu Z, Xia S, Zhao J (2020) Reduction and immobilization of Cr (VI) in aqueous solutions by blast furnace slag supported sulfidized nanoscale zerovalent iron. Sci Total Environ 743:140722
Rice EW, Baird R, Eaton AD (2017) Standard methods for the examination of water and waste water, 23rd edition. American Public Health Association (APHA)
Crini G, Peindy HN, Gimbert F, Robert C (2007) Removal of CI Basic Green 4 (Malachite Green) from aqueous solutions by adsorption using cyclodextrin-based adsorbent: kinetic and equilibrium studies. Sep Purif Technol 53:97–110
Shawabkeh RA (2006) Adsorption of chromium ions from aqueous solution by using activated carbo-aluminosilicate material from oil shale. J Colloid Interface Sci 299:530–536
Saha B, Orvig C (2010) Biosorbents for hexavalent chromium elimination from Industrial and Municipal effluents. Coord Chem Rev 254:2959–2972
Alshehri SM, Naushad M, Ahamad T, Alothman ZA, Aldalbahi A (2014) Synthesis, characterization of curcumin based ecofriendly antimicrobial bio-adsorbent for the removal of phenol from aqueous medium. Chem Eng J 254:181–189
Kennedy LJ, Vijaya JJ, Kayalvizhi K, Sekaran G (2007) Adsorption of phenol from aqueous solutions using mesoporous carbon prepared by two-stage process. Chem Eng J 132:279–287
Sahoo SK, Hota G (2019) Amine-functionalized GO decorated with ZnO-ZnFe2O4 nanomaterials for remediation of Cr (VI) from water. ACS Appl Nano Mater 2:983–996
Das B, Kumar B, Begum W, Bhattarai A, Mondal MH, Saha B (2022) Comprehensive Review on Applications of Surfactants in Vaccine Formulation, Therapeutic and Cosmetic Pharmacy and prevention of Pulmonary Failure due to COVID-19. Chem Afr 5:459–480
He C, Yang Z, Ding J, Chen Y, Tong X, Li Y (2017) Effective removal of Cr (VI) from aqueous solution by 3-aminopropyltriethoxysilane-functionalized graphene oxide. Colloids Surf A 520:448–458
Singh DK, Kumar V, Mohan S, Hasan SH (2017) Polylysine functionalized graphene aerogel for the enhanced removal of Cr (VI) through adsorption: kinetic, isotherm, and thermodynamic modeling of the process. J Chem Eng Data 62:1732–1742
Huang Z, Xiong C, Ying L, Wang W, Wang S, Ding J, Lu J (2022) Facile synthesis of a MOF-derived magnetic CoAl-LDH@chitosan composite for Pb(II) and Cr(VI) adsorption. Chem Eng J 449:137722. https://doi.org/10.1016/j.cej.2022.137722
EP Act (1986) India, Schedule-VI, General Standards for Discharge of Environmental Pollutants. Part-A
Kumar A, Jena HM (2017) Adsorption of Cr (VI) from aqueous phase by high surface area activated carbon prepared by chemical activation with ZnCl2. Process Saf Environ Prot 109:63–71
Norouzi S, Heidari M, Alipour V, Rahmanian O, Fazlzadeh M, Mohammadi-moghadam F, Nourmoradi H, Goudarzi B, Dindarloo K (2018) Preparation, characterization and Cr(VI) adsorption evaluation of NaOH activated carbon produced from Date Press Cake; an agro-industrial waste. Bioresour Technol 258:48–56
Mohit Nigam S, Rajoriya SR, Singh P, Kumar (2019) Adsorption of Cr (VI) ion from tannery wastewater on tea waste: Kinetics, equilibrium and thermodynamics studies. J Environ Chem Eng 7:103188
Labied R, Benturki O, Hamitouche A, Donnot A (2018) Adsorption of hexavalent chromium by activated carbon obtained from a waste lignocellulosic material (Ziziphus jujuba cores): Kinetic, equilibrium, and thermodynamic study. Adsorpt Sci Technol 36(3–4):1006–1099
Dehghani MH, Sanaei D, Ali I, Bhatnagar A (2015) Removal of Cr (VI) from aqueous solution using treated waste newspaper as a low cost adsorbent: Kinetic modeling and isotherm studies. J Mol Liq 215:671–679
Song X, Wang Y, Zhou L, Luo X, Liu J (2021) Halloysite nanotubes stabilized polyurethane foam carbon coupled with iron oxide for high-efficient and fast treatment of arsenic (III/V) wastewater. Chem Eng Res Des 165:298–307
Acknowledgements
The authors deeply acknowledge the institute authorities of ITER, SOA University and NIST for providing the infrastructural and laboratory support to execute the work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors hereby announce that no conflicts of interest of any kind with reference to this research work and the consequent publishing of this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Biswal, S.S., Panda, C., Parida, S.R. et al. Removal of Hexavalent Chromium from Industrial Wastewater Using Adsorbent Prepared from Biomass IPOMOEA CARNEA. Chemistry Africa 5, 1823–1835 (2022). https://doi.org/10.1007/s42250-022-00472-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42250-022-00472-8