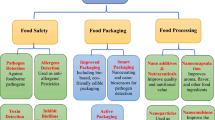
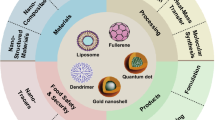
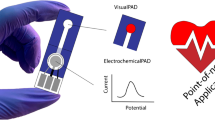
Abstract
Over the years, food-borne illnesses possess a major public health concern around the world, especially those in low-income countries and rural areas. In spite of all safety precautions and control measures to prevent the cause of food-borne bacterial pathogens, there are global demands for rapid detection approaches. Although traditional methods for detecting food-borne bacteria are very accurate, they are very time-consuming, expensive, and also need trained personnel. In recent years, nanotechnology-based approaches with different types of organic, inorganic, and hybrid nanomaterials have been proposed to devise novel detection methods for food pathogens. Among these, metal nanoparticles with surface plasmonic properties, semiconducting nanoparticles with photo-luminescence properties, and their hybrids with different polymeric systems have shown popularity in the design of point-of-care testing (POCT)–based food biosensors, and this is the focus of our current review. This review is majorly divided into two sections; the first one is based on the various approaches as quick detection schemes using different types of nanoparticles and their hybrids. Here, we discuss the properties of different types of plasmonic, semiconducting nanoparticles and how these properties have been used to devise the scheme for rapid detection methods. The second part of the review discusses the use of different receptors on bacterial surfaces enabling quick identification. This includes advantages and disadvantages of different types of receptors such as synthetic antibodies, tailspike proteins, and lectins that have been conjugated onto nanoparticles for binding onto targeted pathogens in the detection process.







Similar content being viewed by others
Data availability
Not applicable.
References
H. Gourama, in Food Engineering Series. Foodborne Pathogens, (Springer, 2020) pp. 25–49. https://doi.org/10.1007/978-3-030-42660-6_2
T. Bintsis, Microbial pollution and food safety. AIMS Microbiol. 4(3), 377 (2018)
World Health Organization, WHO estimates of the global burden of foodborne diseases: foodborne disease burden epidemiology reference group 2007-2015. (World Health Organization, Geneva, Switzerland, 2015). https://apps.who.int/iris/handle/10665/199350
S.M. Pires et al., Burden of foodborne diseases: think global, act local. Curr. Opin. Food Sci. 39, 152–159 (2021)
X. Zhang et al., Recent progress in the construction of nanozyme-based biosensors and their applications to food safety assay. TrAC Trends Anal. Chem. 121, 115668 (2019)
E. Abebe, G. Gugsa, M. Ahmed, Review on major food-borne zoonotic bacterial pathogens. J. Trop. M. 2020, 1–19 (2020). https://doi.org/10.1155/2020/4674235
M. Marin, M.V. Nikolic, J. Vidic, Rapid point-of-need detection of bacteria and their toxins in food using gold nanoparticles. Compr. Rev. Food Sci. Food Saf. 20(6), 5880–5900 (2021)
W. Hussain, M.W. Ullah, U. Farooq, A. Aziz, S. Wang, Bacteriophage-based advanced bacterial detection: concept, mechanisms, and applications. Biosens. Bioelectron. 177, 112973 (2021)
V. Velusamy, K. Arshak, O. Korostynska, K. Oliwa, C. Adley, An overview of foodborne pathogen detection: In the perspective of biosensors. Biotechnol. Adv. 28(2), 232–254 (2010)
D.S.P. Silva, T. Canato, M. Magnani, J. Alves, E.Y. Hirooka, T.C.R.M. de Oliveira, Multiplex PCR for the simultaneous detection of Salmonella spp. and Salmonella Enteritidis in food. Int. J. food Sci. Technol. 46(7), 1502–1507 (2011)
J. Chen, L. Zhang, G.C. Paoli, C. Shi, S.-I. Tu, X. Shi, A real-time PCR method for the detection of Salmonella enterica from food using a target sequence identified by comparative genomic analysis. Int. J. Food Microbiol. 137(2–3), 168–174 (2010)
D.H. D’souza, L. Jaykus, Nucleic acid sequence based amplification for the rapid and sensitive detection of Salmonella enterica from foods. J. Appl. Microbiol. 95(6), 1343–1350 (2003)
L. Wang, L. Shi, J. Su, Y. Ye, Q. Zhong, Detection of Vibrio parahaemolyticus in food samples using in situ loop-mediated isothermal amplification method. Gene 515(2), 421–425 (2013)
N. Singhal, M. Kumar, P.K. Kanaujia, J.S. Virdi, MALDI-TOF mass spectrometry: an emerging technology for microbial identification and diagnosis. Front. Microbiol. 6, 791 (2015)
Y. Wang, E.C. Alocilja, Gold nanoparticle-labeled biosensor for rapid and sensitive detection of bacterial pathogens. J. Biol. Eng. 9(1), 1–7 (2015)
L.-L. Tay et al., Silica encapsulated SERS nanoprobe conjugated to the bacteriophage tailspike protein for targeted detection of Salmonella. Chem. Commun. 48(7), 1024–1026 (2012)
C. Park, J. Lee, Y. Kim, J. Kim, J. Lee, S. Park, 3D-printed microfluidic magnetic preconcentrator for the detection of bacterial pathogen using an ATP luminometer and antibody-conjugated magnetic nanoparticles. J. Microbiol. Methods 132, 128–133 (2017)
J. Chen, S.D. Alcaine, Z. Jiang, V.M. Rotello, S.R. Nugen, Detection of Escherichia coli in drinking water using T7 bacteriophage-conjugated magnetic probe. Anal. Chem. 87(17), 8977–8984 (2015)
A. Abbaspour, F. Norouz-Sarvestani, A. Noori, N. Soltani, Aptamer-conjugated silver nanoparticles for electrochemical dual-aptamer-based sandwich detection of staphylococcus aureus. Biosens. Bioelectron. 68, 149–155 (2015)
M. Imai et al., Dark-field microscopic detection of bacteria using bacteriophage-immobilized SiO2@ AuNP core–shell nanoparticles. Anal. Chem. 91(19), 12352–12357 (2019)
Y.-L. Bai, M. Shahed-Al-Mahmud, K. Selvaprakash, N.-T. Lin, Y.-C. Chen, Tail fiber protein-immobilized magnetic nanoparticle-based affinity approaches for detection of acinetobacter baumannii. Anal. Chem. 91(15), 10335–10342 (2019)
Y. Lin, A.T. Hamme II., Targeted highly sensitive detection/eradication of multi-drug resistant Salmonella DT104 through gold nanoparticle–SWCNT bioconjugated nanohybrids. Analyst 139(15), 3702–3705 (2014)
N. Duan, S. Wu, S. Dai, T. Miao, J. Chen, Z. Wang, Simultaneous detection of pathogenic bacteria using an aptamer based biosensor and dual fluorescence resonance energy transfer from quantum dots to carbon nanoparticles. Microchim. Acta 182(5–6), 917–923 (2015)
G. Sai-Anand et al., Recent progress on the sensing of pathogenic bacteria using advanced nanostructures. Bull. Chem. Soc. Jpn. 92(1), 216–244 (2019)
H. Ding, Y. Ma, Computational approaches to cell–nanomaterial interactions: keeping balance between therapeutic efficiency and cytotoxicity. Nanoscale Horizons 3(1), 6–27 (2018)
X. Huang, M.A. El-Sayed, Gold nanoparticles: optical properties and implementations in cancer diagnosis and photothermal therapy. J. Adv. Res. 1(1), 13–28 (2010)
S. Mukherji et al., Synthesis and characterization of size- and shape-controlled silver nanoparticles. Phys. Sci. Rev. 4(1), 20170082 (2018). https://doi.org/10.1515/psr-2017-0082
J. Belza, A. Opletalová, K. Poláková, Carbon dots for virus detection and therapy. Microchim. Acta 188(12), 1–23 (2021)
F.L. Nobrega et al., Targeting mechanisms of tailed bacteriophages. Nat. Rev. Microbiol. 16(12), 760–773 (2018)
S. Hameed, L. Xie, Y. Ying, Conventional and emerging detection techniques for pathogenic bacteria in food science: a review. Trends Food Sci. Technol. 81, 61–73 (2018)
P. Wang, D. Tanaka, S. Ryuzaki, S. Araki, K. Okamoto, K. Tamada, Silver nanoparticles with tunable work functions. Appl. Phys. Lett. 107(15), 151601 (2015)
A.A. Yaqoob et al., Recent advances in metal decorated nanomaterials and their various biological applications: a review. Front. Chem. 8, 341 (2020)
H. Katas et al. Biosynthesis and potential applications of silver and gold nanoparticles and their chitosan-based nanocomposites in nanomedicine. J. Nanotechnol. 2018, 1–13 (2018). https://doi.org/10.1155/2018/4290705
H.-S. Kim et al., Two-stage label-free aptasensing platform for rapid detection of Cronobacter sakazakii in powdered infant formula. Sensors Actuators B Chem. 239, 94–99 (2017)
Y.-J. Kim, H.-S. Kim, J.-W. Chon, D.-H. Kim, J.-Y. Hyeon, K.-H. Seo, New colorimetric aptasensor for rapid on-site detection of Campylobacter jejuni and Campylobacter coli in chicken carcass samples. Anal. Chim. Acta 1029, 78–85 (2018)
X. Xia, S.M. Mugo, Q. Zhang, Responsive microgels-based wearable devices for sensing multiple health signals. Chem Eng J 427, 130903 (2022). https://doi.org/10.1016/j.cej.2021.130903
Y. Xiao, J. Du, Superparamagnetic nanoparticles for biomedical applications. J. Mater. Chem. B 8(3), 354–367 (2020)
Z. Zhou, L. Yang, J. Gao, X. Chen, Structure–relaxivity relationships of magnetic nanoparticles for magnetic resonance imaging. Adv. Mater. 31(8), 1804567 (2019)
O. Prakash, S. Sil, T. Verma, S. Umapathy, Direct detection of bacteria using positively charged Ag/Au bimetallic nanoparticles: a label-free surface-enhanced Raman scattering study coupled with multivariate analysis. J. Phys. Chem. C 124(1), 861–869 (2019)
M. Türk, C. Erkey, Synthesis of supported nanoparticles in supercritical fluids by supercritical fluid reactive deposition: current state, further perspectives and needs. J. Supercrit. Fluids 134, 176–183 (2018)
A. Ali, M.Z. Hira Zafar, I. ulHaq, A.R. Phull, J.S. Ali, A. Hussain, Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnol. Sci. Appl 9, 49 (2016)
A.P. Tiwari, S.S. Rohiwal, Synthesis and bioconjugation of hybrid nanostructures for biomedical applications. Hybrid Nanostruct. Cancer Theranostics 17–41 (2019). https://doi.org/10.1016/b978-0-12-813906-6.00002-0
I. Khan, A. Abdalla, A. Qurashi, Synthesis of hierarchical WO3 and Bi2O3/WO3 nanocomposite for solar-driven water splitting applications. Int. J. Hydrogen Energy 42(5), 3431–3439 (2017)
N. Kazemifard, A.A. Ensafi, Z.S. Dehkordi, A review of the incorporation of QDs and imprinting technology in optical sensors–imprinting methods and sensing responses. New J. Chem. 45(23), 10170–10198 (2021)
P. Liu et al., Green InP/ZnSeS/ZnS core multi-shelled quantum dots synthesized with aminophosphine for effective display applications. Adv. Funct. Mater. 31(11), 2008453 (2021)
A.L. Efros, L.E. Brus, Nanocrystal quantum dots: from discovery to modern development. ACS Nano 15(4), 6192–6210 (2021)
M. Bhattacharya et al., in Biosensors in food safety and quality. Nanobiosensors: principles, techniques, and innovation in nanobiosensors, (CRC Press, 2022) pp. 85–110. https://doi.org/10.1201/9780429259890-7
B.G. Jeong et al., Interface polarization in heterovalent core–shell nanocrystals. Nat. Mater. 21(2), 246–252 (2022)
G. Kedarnath, in Handbook on synthesis strategies for advanced materials. Synthesis of advanced inorganic materials through molecular precursors, (Springer, Singapore, 2021), pp. 467–501. https://doi.org/10.1007/978-981-16-1807-9_15
X. Gao, S. Nie, Molecular profiling of single cells and tissue specimens with quantum dots. Trends Biotechnol. 21(9), 371–373 (2003)
S. Joshi et al., A review on peptide functionalized graphene derivatives as nanotools for biosensing. Microchim. Acta 187(1), 1–15 (2020)
D.C.F. Soares, S.C. Domingues, D.B. Viana, M.L. Tebaldi, Polymer-hybrid nanoparticles: current advances in biomedical applications. Biomed. Pharmacother. 131, 110695 (2020)
M. Manoswini, D. Bhattacharya, P. Sen, N. Ganguly, P.S. Mohanty, Antibacterial and cytotoxic activity of polymer-metal hybrid nanoparticle. Adv. Nat. Sci. Nanosci. Nanotechnol. 12(2), 025003 (2021)
A. Bordat, T. Boissenot, J. Nicolas, N. Tsapis, Thermoresponsive polymer nanocarriers for biomedical applications. Adv. Drug Deliv. Rev. 138, 167–192 (2019)
M. Sponchioni, U.C. Palmiero, D. Moscatelli, Thermo-responsive polymers: applications of smart materials in drug delivery and tissue engineering. Mater. Sci. Eng. C 102, 589–605 (2019)
B. Mutharani, P. Ranganathan, S.-M. Chen, Stimuli-enabled reversible switched aclonifen electrochemical sensor based on smart PNIPAM/PANI-Cu hybrid conducting microgel. Sensors Actuators B Chem. 304, 127232 (2020)
P. Bhol, M. Mohanty, P.S. Mohanty, Polymer-matrix stabilized metal nanoparticles: aynthesis, characterizations and insight into molecular interactions between metal ions, atoms and polymer moieties. J. Mol. Liq. 325, 115135 (2021)
A. De Backer, L. Jones, A. Varambhia, P.D. Nellist, S. Van Aert, Measuring dynamic structural changes of nanoparticles at the atomic scale using scanning transmission electron microscopy. Phys. Rev. Lett. 124(10), 106105 (2020)
D. Ma, in Noble metal-metal oxide hybrid nanoparticles. Hybrid nanoparticles, (Elsevier Inc., 2019), pp. 3–6. https://doi.org/10.1016/b978-0-12-814134-2.00001-2
J. Zhou, L. Rao, G. Yu, T.R. Cook, X. Chen, F. Huang, Supramolecular cancer nanotheranostics. Chem. Soc. Rev. 50(4), 2839–2891 (2021)
R.-V. Kalaydina, K. Bajwa, B. Qorri, A. Decarlo, M.R. Szewczuk, Recent advances in ‘smart’ delivery systems for extended drug release in cancer therapy. Int. J. Nanomedicine 13, 4727 (2018)
M.A. Wahab, E.Y. Erdem, Multi-step microfludic reactor for the synthesis of hybrid nanoparticles. J. Micromechanics Microengineering 30(8), 85006 (2020)
T. Shu et al., Multi-responsive micro/nanogels for optical sensing. Adv. Phys. X 7(1), 2043185 (2022)
N. Duan et al., A dual-color flow cytometry protocol for the simultaneous detection of Vibrio parahaemolyticus and Salmonella typhimurium using aptamer conjugated quantum dots as labels. Anal. Chim. Acta 804, 151–158 (2013)
J. Hu et al., Colorimetric-fluorescent-magnetic nanosphere-based multimodal assay platform for Salmonella detection. Anal. Chem. 91(1), 1178–1184 (2018)
E. Mohamadi, M. Moghaddasi, A. Farahbakhsh, A. Kazemi, A quantum-dot-based fluoroassay for detection of food-borne pathogens. J. Photochem. Photobiol. B Biol. 174, 291–297 (2017)
K. Yamada, W. Choi, I. Lee, B.-K. Cho, S. Jun, Rapid detection of multiple foodborne pathogens using a nanoparticle-functionalized multi-junction biosensor. Biosens. Bioelectron. 77, 137–143 (2016)
S. Wu, Y. Wang, N. Duan, H. Ma, Z. Wang, Colorimetric aptasensor based on enzyme for the detection of Vibrio parahemolyticus. J. Agric. Food Chem. 63(35), 7849–7854 (2015)
J. Zhang, L. Mou, X. Jiang, Surface chemistry of gold nanoparticles for health-related applications. Chem. Sci. 11(4), 923–936 (2020)
X. Han, K. Xu, O. Taratula, K. Farsad, Applications of nanoparticles in biomedical imaging. Nanoscale 11(3), 799–819 (2019)
C.I. Colino, J.M. Lanao, C. Gutierrez-Millan, Recent advances in functionalized nanomaterials for the diagnosis and treatment of bacterial infections. Mater. Sci. Eng. C. 121, 111843 (2021). https://doi.org/10.1016/j.msec.2020.111843
S. Singh, V. Kumar, D.S. Dhanjal, S. Datta, R. Prasad, J. Singh, in Microbial biotechnology: basic research and applications. Environmental and microbial biotechnology, eds., J. Singh, A. Vyas, S. Wang, R. Prasad. Biological biosensors for monitoring and diagnosis, (Springer, Singapore, 2020). https://doi.org/10.1007/978-981-15-2817-0_14
T. Ozer, B.J. Geiss, C.S. Henry, Chemical and biological sensors for viral detection. J. Electrochem. Soc. 167(3), 37523 (2019)
P. D’Orazio, Biosensors in clinical chemistry. Clin. Chim. acta 334(1–2), 41–69 (2003)
R. Zhang, T. Belwal, L. Li, X. Lin, Y. Xu, Z. Luo, Nanomaterial-based biosensors for sensing key foodborne pathogens: advances from recent decades. Compr. Rev. Food Sci. Food Saf. 19(4), 1465–1487 (2020)
T. Vo-Dinh, B. Cullum, Biosensors and biochips: advances in biological and medical diagnostics. Fresenius. J. Anal. Chem. 366(6), 540–551 (2000)
S. Ko, T.J. Park, H.-S. Kim, J.-H. Kim, Y.-J. Cho, Directed self-assembly of gold binding polypeptide-protein A fusion proteins for development of gold nanoparticle-based SPR immunosensors. Biosens. Bioelectron. 24(8), 2592–2597 (2009)
N. Verdoodt, C.R. Basso, B.F. Rossi, V.A. Pedrosa, Development of a rapid and sensitive immunosensor for the detection of bacteria. Food Chem. 221, 1792–1796 (2017)
Y. Wang, Z. Ye, Y. Ying, New trends in impedimetric biosensors for the detection of foodborne pathogenic bacteria. Sensors 12(3), 3449–3471 (2012)
K.J. Lee et al., Simple and rapid detection of bacteria using a nuclease-responsive DNA probe. Analyst 143(1), 332–338 (2018)
A. Singh, D. Arutyunov, C.M. Szymanski, S. Evoy, Bacteriophage based probes for pathogen detection. Analyst 137(15), 3405–3421 (2012)
H. Peng, I.A. Chen, Rapid colorimetric detection of bacterial species through the capture of gold nanoparticles by chimeric phages. ACS Nano 13(2), 1244–1252 (2018)
Z. AliakbarAhovan, A. Hashemi, L.M. De Plano, M. Gholipourmalekabadi, A. Seifalian, Bacteriophage based biosensors: trends, outcomes and challenges. Nanomater 10(3), 501 (2020)
A. Singh, D. Arutyunov, M.T. McDermott, C.M. Szymanski, S. Evoy, Specific detection of Campylobacter jejuni using the bacteriophage NCTC 12673 receptor binding protein as a probe. Analyst 136(22), 4780–4786 (2011)
A. Singh et al., Bacteriophage tailspike proteins as molecular probes for sensitive and selective bacterial detection. Biosens. Bioelectron. 26(1), 131–138 (2010)
S. Poshtiban, A. Singh, G. Fitzpatrick, S. Evoy, Bacteriophage tail-spike protein derivitized microresonator arrays for specific detection of pathogenic bacteria. Sensors Actuators B Chem. 181, 410–416 (2013)
E. Nakata, S. Nakano, A. Rajendran, and T. Morii, “Covalent bond formation by modular adaptors to locate multiple enzymes on a DNA scaffold,” in Kinetic Control in Synthesis and Self-Assembly, Elsevier, 2019, pp. 163–183.
N. Wongkaew, M. Simsek, C. Griesche, A.J. Baeumner, Functional nanomaterials and nanostructures enhancing electrochemical biosensors and lab-on-a-chip performances: recent progress, applications, and future perspective. Chem. Rev. 119(1), 120–194 (2018)
M.S. Draz, H. Shafiee, Applications of gold nanoparticles in virus detection. Theranostics 8(7), 1985 (2018)
S. Kumar, J. Aaron, K. Sokolov, Directional conjugation of antibodies to nanoparticles for synthesis of multiplexed optical contrast agents with both delivery and targeting moieties. Nat. Protoc. 3(2), 314–320 (2008)
G.T. Hermanson, in Bioconjugate techniques. Isotopic labeling techniques, (Elsevier, 2013), pp. 507–534. https://doi.org/10.1016/b978-0-12-382239-0.00012-1
L. Zhang, Y. Mazouzi, M. Salmain, B. Liedberg, S. Boujday, Antibody-gold nanoparticle bioconjugates for biosensors: synthesis, characterization and selected applications. Biosens. Bioelectron. 165, 112370 (2020)
J.H.T. Luong, S.K. Vashist, Chemistry of biotin–streptavidin and the growing concern of an emerging biotin interference in clinical immunoassays. ACS Omega 5(1), 10–18 (2019)
J. Conde, J.T. Dias, V. Grazú, M. Moros, P.V. Baptista, J.M. de la Fuente, Revisiting 30 years of biofunctionalization and surface chemistry of inorganic nanoparticles for nanomedicine. Front. Chem. 2, 48 (2014)
J. Chen, S.M. Andler, J.M. Goddard, S.R. Nugen, V.M. Rotello, Integrating recognition elements with nanomaterials for bacteria sensing. Chem. Soc. Rev. 46(5), 1272–1283 (2017)
I. Gessner, I. Neundorf, Nanoparticles modified with cell-penetrating peptides: conjugation mechanisms, physicochemical properties, and application in cancer diagnosis and therapy. Int. J. Mol. Sci. 21(7), 2536 (2020)
A. Heuer-Jungemann et al., The role of ligands in the chemical synthesis and applications of inorganic nanoparticles. Chem. Rev. 119(8), 4819–4880 (2019)
L. Guerrini, R.A. Alvarez-Puebla, N. Pazos-Perez, Surface modifications of nanoparticles for stability in biological fluids. Mater (Basel) 11(7), 1154 (2018)
G. Sanità, B. Carrese, A. Lamberti, Nanoparticle surface functionalization: How to improve biocompatibility and cellular internalization. Front. Mol. Biosci. 7, 587012 (2020). https://doi.org/10.3389/fmolb.2020.587012
M. Bilal, H.M.N. Iqbal, Chemical, physical, and biological coordination: an interplay between materials and enzymes as potential platforms for immobilization. Coord. Chem. Rev. 388, 1–23 (2019)
G.R. Rudramurthy, M.K. Swamy, Potential applications of engineered nanoparticles in medicine and biology: An update. JBIC J. Biol. Inorg. Chem. 23(8), 1185–1204 (2018)
K. Kalimuthu et al., Gold nanoparticles stabilize peptide-drug-conjugates for sustained targeted drug delivery to cancer cells. J. Nanobiotechnol 16(1), 1–13 (2018)
S.J. Amina, B. Guo, A review on the synthesis and functionalization of gold nanoparticles as a drug delivery vehicle. Int. J. Nanomedicine 15, 9823 (2020)
C.M. Goodman, et al. Toxicity of gold nanoparticles functionalized with cationic and anionic side chains. Bioconjug Chem. 15(4), 897–900 (2004). https://doi.org/10.1021/bc049951i
Z.U.H. Khan et al., Biomedical applications of green synthesized Nobel metal nanoparticles. J. Photochem. Photobiol. B Biol. 173, 150–164 (2017)
B. Nasseri, N. Soleimani, N. Rabiee, A. Kalbasi, M. Karimi, M.R. Hamblin, Point-of-care microfluidic devices for pathogen detection. Biosens. Bioelectron. 117, 112–128 (2018)
M. Kotepui, K.U. Kotepui, G. De Jesus Milanez, F.R. Masangkay, Summary of discordant results between rapid diagnosis tests, microscopy, and polymerase chain reaction for detecting Plasmodium mixed infection: a systematic review and meta-analysis. Sci. Rep 10(1), 1–17 (2020)
S. Li, Z. Ma, Z. Cao, L. Pan, Y. Shi, Advanced wearable microfluidic sensors for healthcare monitoring. Small 16(9), 1903822 (2020)
A.I. Akinyemi, A.F. Fagbamigbe, E. Omoluabi, O.M. Agunbiade, S.O. Adebayo, Diarrhoea management practices and child health outcomes in Nigeria: sub-national analysis. Adv. Integr. Med. 5(1), 15–22 (2018)
D.M. Anderson, D.I. Rees, T. Wang, The phenomenon of summer diarrhea and its waning, 1910–1930. Explor. Econ. Hist. 78, 101341 (2020)
A. Kamath, K. Shetty, B. Unnikrishnan, S. Kaushik, S.N. Rai, Prevalence, patterns, and predictors of diarrhea: a spatial-temporal comprehensive evaluation in India. BMC Public Health 18(1), 1–10 (2018)
I. Khan, K. Saeed, I. Khan, Nanoparticles: properties, applications and toxicities. Arab. J. Chem. 12(7), 908–931 (2019)
S. Förster, M. Konrad, From self-organizing polymers to nano-and biomaterials. J. Mater. Chem. 13(11), 2671–2688 (2003)
M. Zakharzhevskii, A.S. Drozdov, D.S. Kolchanov, L. Shkodenko, V.V. Vinogradov, Test-system for bacteria sensing based on peroxidase-like activity of inkjet-printed magnetite nanoparticles. Nanomater 10(2), 313 (2020)
R. Torensma, M.J. Visser, C.J. Aarsman, M.J. Poppelier, A.C. Fluit, J. Verhoef, Monoclonal antibodies that react with live Listeria spp. Appl. Environ. Microbiol. 59(8), 2713–2716 (1993)
H.R. Hoogenboom, Selecting and screening recombinant antibody libraries. Nat. Biotechnol. 23, 1105–1116 (2005)
N. Zambrano et al., High-throughput monoclonal antibody discovery from phage libraries: challenging the current preclinical pipeline to keep the pace with the increasing mAb demand. Cancers (Basel) 14(5), 1325 (2022)
E. Stone, K. Campbell, I. Grant, O. McAuliffe, Understanding and exploiting phage–host interactions. Viruses 11(6), 567 (2019)
A. Freiberg et al., The tailspike protein of Shigella phage Sf6: a structural homolog of Salmonella phage P22 tailspike protein without sequence similarity in the β-helix domain. J. Biol. Chem. 278(3), 1542–1548 (2003)
Funding
This work is partially supported by BIRAC, COE, DBT, Govt. of India and ICMR (Indian Council of Medical Research), Government of India.
Author information
Authors and Affiliations
Contributions
PSM designed the studies, supervise, and formulate the work plan of review and its revision, writing, and corrections. BRS provided valuable suggestions and idea to improve the review, writing, editing, and corrections. MM: original draft preparation, writing, editing, and referencing. AGM and BP contribute significantly to the writing, editing, and referencing of the section on the use of nanomaterials for the detection of food pathogens.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Manoswini, M., Majumdar, A.G., Pany, B. et al. Rapid detections of food pathogens using metal, semiconducting nanoparticles, and their hybrids: a review. emergent mater. 6, 15–30 (2023). https://doi.org/10.1007/s42247-022-00441-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42247-022-00441-4