Abstract
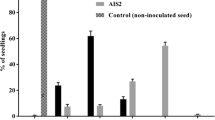
Almond witches’-broom (AlmWB) is a serious phytoplasma disease of stone fruits; thus far reported from Iran and Lebanon. In the present study the host range of two Iranian AlmWB phytoplasma strains, known as PEAB and AlKI, were examined through graft-inoculation assay. Two-year-old stone fruit seedlings of almond (three cultivars), peach, plum, Myrobalan plum, apricot and sweet cherry were separately graft-inoculated by scions obtained from symptomatic almond seedlings infected by PEAB or AlKI phytoplasma strains. The presence of phytoplasma in the inoculated seedlings, and the type and development of symptoms were evaluated and recorded monthly over one year. Results showed that both phytoplasma strains could be transmitted to almond (‘Pooya’, ‘Mamaei’ and ‘Talkhe’ cultivars), peach and plum seedlings. The disease symptoms were more obvious in almond cultivars. AlmWB phytoplasma strains could not be detected in the inoculated sweet cherry and the Myrobalan plum seedlings. On the other hand, infection of apricot seedlings could only be detected in seedlings inoculated with the PEAB strain. The homologous of the AYWB SAP11 effector protein, known to play a crucial role in phytoplasma infection and symptom expression, was sequence-analyzed in both PEAB and AlKI strains. The SAP11 nucleotide sequence of AlKI strain was identical to that of the SA213 AlmWB phytoplasma strain from Lebanon. Analysis showed high SAP11 sequence diversity between AlmWB phytoplasmas and phytoplasma strains of other 16Sr groups. Despite poor sequence conservation, the structural features and the motifs of SAP11 protein were conserved between AlmWB and distantly related phytoplasma strains; speculating a similar function for this gene in diverse phytoplasmas.



Similar content being viewed by others
Data Availability
The nucleotide sequence of the SAP11 gene from the AlmWB phytoplasma strains, PEAB and AlKI, identified in this study were deposited in the GenBank under accession numbers ON186619 and ON186620.
References
Abbasian M, Salehi M (2010) Reaction of stone fruit cultivars to almond witches’ broom phytoplasma. Iran J Plant Pathol 46:153–160
Abou-Jawdah Y, Dakhil H, El-Mehtar S, Lee IM (2003) Almond witches’-broom phytoplasma, a potential threat to almond, peach and nectarine. Can J Plant Pathol 25:28–32
Abou-Jawdah Y, Karakashian A, Sobh H, Martini M, Lee IM (2002) An epidemic of almond witches’-broom in Lebanon: classification and phylogenetic relationship of the associated phytoplasma. Plant Dis 86:477–484
Anabestani A, Izadpanah K, Abbà S, Galetto L, Ghorbani A, Palmano S, Siampour M, Veratti F, Marzachì C (2017) Identification of putative effector genes and their transcripts in three strains related to ‘Candidatus Phytoplasma aurantifolia’. Microbiol Res 199:57–66
Babaei G, Esmaeilzadeh-Hosseini SA, Shirmardi HA, Bertaccini A (2021) Identification of a 16SrIX‐B Phytoplasma strain associated with Daphne mucronata phyllody in Iran. For Pathol 51:e12703
Bertaccini A, Arocha-Rosete Y, Contaldo N, Duduk B, Fiore N, Montano HG, Kube M, Kuo CH, Martini M, Oshima K, Quaglino F(2022) Revision of the ‘Candidatus Phytoplasma’species description guidelines. IJSEM 72:005353
Bertaccini A, Lee IM (2018) Phytoplasmas: an update. In: Rao GP, Beratccini A, Fiore N, Liefting LW (eds) Phytoplasmas: plant pathogenic Bacteria-I. Springer, Singapore, pp 1–29
Daire X, Clair D, Reinert W, Boudon-Padieu E (1997) Detection and differentiation of grapevine yellows phytoplasmas belonging to the elm yellows group and to the stolbur subgroup by PCR amplification of non-ribosomal DNA. Eur J Plant Pathol 103:507–514
Deng S, Hiruki C (1991) Amplification of 16S rRNA genes from culturable and nonculturable mollicutes. J Microbiol Methods 14:53–61
EPPO (2017) Pest risk analysis for ‘Candidatus Phytoplasma phoenicium’. EPPO, Paris. Available at https://pra.eppo.int/pra/3dc3b492-546f-4ae8-952a-10f02a0fdf8f. Accessed 2 May 2022
Gundersen DE, Lee IM (1996) Ultrasensitive detection of phytoplasmas by nested-PCR assays using two universal primer pairs. Phytopathol Mediterr 35:144–151
Hogenhout SA, Oshima K, Ammar ED, Kakizawa S, Kingdom HN, Namba S (2008) Phytoplasmas: bacteria that manipulate plants and insects. Mol Plant Pathol 9:403–423
Hoshi A, Oshima K, Kakizawa S, Ishii Y, Ozeki J, Hashimoto M, Komatsu K, Kagiwada S, Yamaji Y, Namba S (2009) A unique virulence factor for proliferation and dwarfism in plants identified from a phytopathogenic bacterium. PNAS 106:6416–6421
Iwabuchi N, Kitazawa Y, Maejima K, Koinuma H, Miyazaki A, Matsumoto O, Suzuki T, Nijo T, Oshima K, Namba S, Yamaji Y (2020) Functional variation in phyllogen, a phyllody-inducing phytoplasma effector family, attributable to a single amino acid polymorphism. Mol Plant Pathol 21:1322–1336
Janik K, Mithöfer A, Raffeiner M, Stellmach H, Hause B, Schlink K (2017) An effector of apple proliferation phytoplasma targets TCP transcription factors—a generalized virulence strategy of phytoplasma? Mol Plant Pathol 18:435–442
Kison H, Seemüller E (2001) Differences in strain virulence of the European stone fruit yellows phytoplasma and susceptibility of stone fruit trees on various rootstocks to this pathogen. J Phytopathol 149:533–541
Lee IM, Bottner-Parker KD, Zhao Y, Bertaccini A, Davis RE (2012) Differentiation and classification of phytoplasmas in the pigeon pea witches’-broom group (16SrIX): an update based on multiple gene sequence analysis. IJSEM 62:2279–2285
Ludwiczak J, Winski A, Szczepaniak K, Alva V, Dunin-Horkawicz S (2019) DeepCoil—a fast and accurate prediction of coiled-coil domains in protein sequences. Bioinformatics 35:2790–2795
Marcone C (2014) Molecular biology and pathogenicity of phytoplasmas. Ann Appl Biol 165:199–221
Lova MM, Quaglino F, Abou-Jawdah Y, Choueiri E, Sobh H, Casati P, Tedeschi R, Alma A, Bianco PA (2011) Identification of new 16SrIX subgroups,-F and-G, among ‘Candidatus Phytoplasma phoenicium’ strains infecting almond, peach and nectarine in Lebanon. Phytopathol Mediterr 50:273–282
MacLean AM, Sugio A, Makarova OV, Findlay KC, Grieve VM, Tóth R, Nicolaisen M, Hogenhout SA (2011) Phytoplasma effector SAP54 induces indeterminate leaf-like flower development in Arabidopsis plants. Plant Physiol 157:831–841
Minato N, Himeno M, Hoshi A, Maejima K, Komatsu K, Takebayashi Y, Kasahara H, Yusa A, Yamaji Y, Oshima K, Kamiya Y (2014) The phytoplasmal virulence factor TENGU causes plant sterility by downregulating of the jasmonic acid and auxin pathways. Sci Rep 4:7399
Mosayyebi N, Mehraban Z, Siampour M, Babaei G, Quaglino F (2021) Multilocus sequence typing and phylogenetic analysis revealed two distinct almond witches’-broom phytoplasma subpopulations in Iran. Ann Appl Biol 179:48–59
Pastore M, Piccirillo P, Simeone AM, Tian J, Paltrinieri S, Bertaccini A (2001) Transmission by patch grafting of ESFY phytoplasma to apricot (Prunus armeniaca L.) and Japanese plum (Prunus salicina Lindl). Acta Hortic 550:339–344
Petersen TN, Brunak S, Von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat methods 8:785–786
Quaglino F, Kube M, Jawhari M, Abou-Jawdah Y, Siewert C, Choueiri E, Sobh H, Casati P, Tedeschi R, Lova MM, Alma A (2015) ‘Candidatus Phytoplasma phoenicium ’associated with almond witches’-broom disease: from draft genome to genetic diversity among strain populations. BMC microbiol 15:148
Salehi M, Hosseini SA, Salehi E, Quaglino F, Bianco PA (2020) Peach witches’-broom, an emerging disease associated with ‘Candidatus Phytoplasma phoenicium’and ‘Candidatus Phytoplasma aurantifolia’in Iran. Crop Protec 127:104946
Salehi M, Izadpanah K(1995) Almond brooming. In: Proceedings of the 12th Iranian Plant Protection Congress, Karaj, Iran. p 238
Salehi M, Izadpanah K, Heydarnejad J (2006) Characterization of a new almond witches’ broom phytoplasma in Iran. J Phytopathol 154:386–391
Salehi M, Salehi E, Siampour M, Quaglino F, Bianco PA (2018) Apricot yellows associated with ‘Candidatus Phytoplasma phoenicium’ in Iran. Phytopathol Mediterr 57:269–283
Seemüller E, Schneider B (2007) Differences in virulence and genomic features of strains of ‘Candidatus Phytoplasma mali’, the apple proliferation agent. Phytopathology 97:964–970
Smart CD, Schneider B, Blomquist CL, Guerra LJ, Harrison NA, Ahrens U, Lorenz KH, Seemüller E, Kirkpatrick BC (1996) Phytoplasma-specific PCR primers based on sequences of the 16S-23S rRNA spacer region. Appl Environ Microbiol 62:2988–2993
Sugio A, Kingdom HN, MacLean AM, Grieve VM, Hogenhout SA (2011) Phytoplasma protein effector SAP11 enhances insect vector reproduction by manipulating plant development and defense hormone biosynthesis. PNAS 108:E1254–E1263
Sugio A, MacLean AM, Hogenhout SA (2014) The small phytoplasma virulence effector SAP 11 contains distinct domains required for nuclear targeting and CIN-TCP binding and destabilization. New Phytol 202:838–848
Tawidian P, Jawhari M, Sobh H, Bianco P, Abou-Jawdah Y (2018) The potential of grafting with selected stone fruit varieties for management of almond witches’ broom. Phytopathol Mediterr 56:458–469
Tedeschi R, Picciau L, Quaglino F, Abou-Jawdah Y, Molino Lova M, Jawhari M, Casati P, Cominetti A, Choueiri E, Abdul‐Nour H, Bianco PA (2015) A cixiid survey for natural potential vectors of ‘Candidatus Phytoplasma phoenicium’ in Lebanon and preliminary transmission trials. Ann Appl Biol 16:372–388
Tomkins M, Kliot A, Maree AF, Hogenhout SA (2018) A multi-layered mechanistic modelling approach to understand how effector genes extend beyond phytoplasma to modulate plant hosts, insect vectors and the environment. Curr Opin Plant Biol 44:39–48
Tsirigos KD, Peters C, Shu N, Käll L, Elofsson A (2015) The TOPCONS web server for consensus prediction of membrane protein topology and signal peptides. Nucleic Acids Res 43:W401–W407
Verdin E, Salar P, Danet JL, Choueiri E, Jreijiri F, El Zammar S, Gelie B, Bove JM, Garnier M (2003) ‘Candidatus Phytoplasma phoenicium’ sp. nov, a novel phytoplasma associated with an emerging lethal disease of almond trees in Lebanon and Iran. IJSEM 53:833–838
Weintraub PG, Beanland L (2006) Insect vectors of phytoplasmas. Annu Rev Entomol 51:91–111
Acknowledgements
The authors would like to thank Dr. H. Noorbakhsh for assistance in grafting and rearing stone fruit seedlings.
Funding
This work was funded by Shahrekord University, Shahrekord, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kabiri, H., Siampour, M. & Babaei, G. Response of Prunus species to graft-inoculation by two Iranian strains of almond witches’-broom phytoplasma. J Plant Pathol 105, 29–37 (2023). https://doi.org/10.1007/s42161-022-01230-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-022-01230-y