Abstract
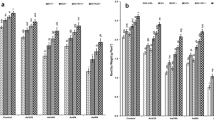
Charcoal rot (CR), caused by Macrophomina phaseolina, is a major disease of sunflowers in semi-arid regions with poor soil conditions. There are fungicide-based strategies to mange this disease; however, alternative eco-friendly control approachs are needed to prevent further deterioration for the environment. This study describes CR control in sunflower grown in calcareous soil using a talc-based bioformulation containing a mixture of rhizobacterial strains (Azotobacter chroococcum, Azospirillum brasilense, and Klebsiella pneumoniae) integrated with silicon nutrition. The study was conducted under greenhouse conditions; M. phaseolina was inoculated in the soil, and rhizobacterial strains were applied as seed treatment, as well as soil drenching at 7, 30, and 45 days after planting (DAP). As the silicon source (Si), potassium silicate was added to nutrient solution at a concentration of 2 mM and applied weekly starting from 7 DAP. The plants, collected at 85 DAP, showed that applying rhizobacteria and Si individually and in combination was effective in controlling CR disease and improving plant growth. The treatment that included rhizobacterial mixture and Si nutrition had a synergistic effect and resulted in the highest reduction (P < 0.05) in the disease incidence (65.3%), root-rot index (57.8%), and stem tissue colonization (63.6%) compared to non-treated inoculated control. Also, the decrease in the growth and nutrient uptake of sunflower plants due to the pathogen-induced stress was significantly reduced. Notably, rhizobacteria + Si treated-plants exhibited less lipid peroxidation and more phenolic and lignin contents with greater defence enzyme activities due to reduced root damage during M. phaseolina infection. Therefore, the integration between rhizobacterial inoculation and Si fertilization can be a good candidate for managing CR in sunflowers in semi-arid regions.






Similar content being viewed by others
References
Abbas HK, Bellaloui N, Accinelli C, Smith JR, Shier WT (2019) Toxin production in soybean (Glycine max L.) plants with charcoal rot disease and by Macrophomina phaseolina, the fungus that causes the disease. Toxins 11:645. https://doi.org/10.3390/toxins11110645
Abo-Elyousr KA, Hashem M, Ali EH (2009) Integrated control of cotton root rot disease by mixing fungal biocontrol agents and resistance inducers. Crop Prot 28:295–301. https://doi.org/10.1016/j.cropro.2008.11.004
Ahemad M, Kibret M (2014) Mechanisms and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Univ Sci 26:1–20. https://doi.org/10.1016/j.jksus.2013.05.001
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein- dye binding. Anal. Biochem 72:1151–1154. https://doi.org/10.1016/0003-2697(76)90527-3
Bruce RJ, West CA (1989) Elicitation of lignin biosynthesis and isoperoxidase activity by pectic fragments in suspension cultures of castor bean. Plant Physiol 91:889–897. https://doi.org/10.1104/pp.91.3.889
Bueno ACSO, Castro GLS, Silva Junior DD, Pinheiro HA, Filippi MCC, Silva GB (2017) Response of photosynthesis and chlorophyll a fluorescence in leaf scald-infected rice under influence of rhizobacteria and silicon fertilizer. Plant Pathol 66:1487–1495. https://doi.org/10.1111/ppa.12690
Campbell MM, Sederoff RR (1996) Variation in lignin content and composition. Plant Physiol 110:3–13. https://doi.org/10.1104/pp.110.1.3
Chamorro M, Seijo TE, Noling JC, De los Santos B, Peres NA (2016) Efficacy of fumigant treatments and inoculum placement on control of Macrophomina phaseolina in strawberry beds. Crop Prot 90: 163–169. https://doi.org/10.1016/j.cropro.2016.08.020
Chance B, Maehley AC (1955) The assay of catalases and peroxidases. Methods Enzymol 2:764–775. https://doi.org/10.1002/9780470110171.ch14
Dallagnol LJ, Rodrigues FA, DaMatta FM, Mielli MVB, Pereira SC (2011) Deficiency in silicon uptake affects cytological, physiological, and biochemical events in the rice-Bipolaris oryzae interaction. Phytopathology 101:92–104. https://doi.org/10.1094/PHYTO-04-10-0105
El_Komy MH, Hassouna MG, Abou-Taleb EM, Al-Sarar AS, Abobakr Y (2020a) A mixture of Azotobacter, Azospirillum, and Klebsiella strains improves root-rot disease complex management and promotes growth in sunflowers in calcareous soil. Eur J Plant Pathol:1–14. https://doi.org/10.1007/s10658-019-01921-w
El_Komy MH, Saleh AA, Ibrahim YE, Molan YY (2020b) Early production of reactive oxygen species coupled with an efficient antioxidant system play a role in potato resistance to late blight. Trop Plant Pathol 1–12. https://doi.org/10.1007/s40858-019-00318-8
Fan XY, Lin WP, Rui LIU, Jiang NH, Cai KZ (2018) Physiological response and phenolic metabolism in tomato (Solanum lycopersicum) mediated by silicon under Ralstonia solanacearum infection. J Integr Agric 17:2160–2171. https://doi.org/10.1016/S2095-3119(18)62036-2
Fortunato AA, Rodrigues FA, Nascimento KJT (2012) Physiological and biochemical aspects of the resistance of banana plants to Fusarium wilt potentiated by silicon. Phytopathology 102:957–966. https://doi.org/10.1094/PHYTO-02-12-0037-R
Gadaga SJC, Abreu MS, Resende MLV, Ribeiro Júnior PM (2017) Phosphites for the control of anthracnose in common bean. Pesqui Agropecu Bras 52:36–44. https://doi.org/10.1590/s0100-204x2017000100005
Gomez KA, Gomez AA (1984) Statistical procedures for agricultural research. Wiley, New York. https://doi.org/10.1017/S0014479700014496
Greger M, Landberg T, Vaculík M (2018) Silicon influences soil availability and accumulation of mineral nutrients in various plant species. Plants 7:41. https://doi.org/10.3390/plants7020041
Harveson RM, Markell SG, Block CC, Gulya TJ (2016) Compendium of sunflower diseases and pests. APS press, Minnesota https://doi.org/10.1094/9780890545096
Hassouna MG (1973) Biological curing of nitrogen deficiency in Gramineae. I. Inoculation of barley grown in pots with non-symbiotic N2-fixers. Proceedings of the First Congress of the Egyptian Phytopathological Society, Nov. 12–15, 1973, Cairo, Egypt. Academy of Scientific Research and Technology
Hassouna MG, El-Saedy MAM, Saleh HM (1998) Biocontrol of soil-borne plant pathogens attacking cucumber (Cucumis sativus) by Rhizobacteria in a semiarid environment. Arid Land Res Manag 12:345–357. https://doi.org/10.1080/15324989809381523
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Kar M, Mishra D (1976) Catalase, peroxidase, and polyphenoloxidase activities during rice leaf senescence. Plant Physiol 57:315–319. https://doi.org/10.1104/pp.57.2.315
Khan MA, Ungar IA (1996) Influence of salinity and temperature on the germination of Haloxylon recurvum bunge ex. boiss. Ann Bot 78:547–551. https://doi.org/10.1006/anbo.1996.0159
Kilmer VJ, Madison WI (1965) Silicon. Pages 959–962 in: Methods of Soil Analysis. American Society of Agronomy: Part 2. https://doi.org/10.2134/agronmonogr9.2.c13
Kumari S, Khanna V (2020) Induction of systemic resistance in chickpea (Cicer arietinum L.) against Fusarium oxysporum f. sp. ciceris by antagonistic rhizobacteria in assistance with native mesorhizobium. Curr Microbiol 77:85–98. https://doi.org/10.1007/s00284-019-01805-6
Lebeda A, Sedlářová M, Petřivalský M, Prokopová J (2008) Diversity of defence mechanisms in plant– Oomycete interactions: a case study of Lactuca spp. and Bremia lactucae. Eur J Plant Pathol 122:71–89. https://doi.org/10.1007/s10658-008-9292-3
Mahmood S, Daur I, Al-Solaimani SG, Ahmad S, Madkour MH, Yasir M, Ali Z (2016) Plant growth promoting rhizobacteria and silicon synergistically enhance salinity tolerance of mung bean. Front Plant Sci 7:876. https://doi.org/10.3389/fpls.2016.00876
Malavolta E, Vitti GC, Oliveira SAD (1997) Avaliação do estado nutricional das plantas: princípios e aplicações
Mengistu A, Ray JD, Smith JR, Paris RL (2007) Charcoal rot disease assessment of soybean genotypes using a colony-forming unit index. Crop Sci 47:2453–2461. https://doi.org/10.2135/cropsci2007.04.0186
Mohaghegh P, Khoshgoftarmanesh AH, Shirvani M, Sharifnabi B, Nili N (2011) Effect of silicon nutrition on oxidative stress induced by Phytophthora melonis infection in cucumber. Plant Dis 95:455–460. https://doi.org/10.1094/PDIS-05-10-0379
Morrison IM, Stewart D (1995) Determination of lignin in the presence of ester-bound substituted cinnamic acids by a modified acetyl bromide procedure. J Sci Food Agric 69:151–157. https://doi.org/10.1002/jsfa.2740690204
Mufti R, Bano A (2019) PGPR-induced defense responses in the soybean plant against charcoal rot disease. Eur J Plant Pathol 155:983–1000. https://doi.org/10.1007/s10658-019-01828-6
Nafady NA, Hashem M, Hassan EA, Ahmed HA, Alamri SA (2019) The combined effect of arbuscular mycorrhizae and plant-growth-promoting yeast improves sunflower defense against Macrophomina phaseolina diseases. Biol Control 138:104049. https://doi.org/10.1016/j.biocontrol.2019.104049
Nascimento KJT, Debona D, Rezende D, DaMatta FM, Rodrigues FÁ (2018) Changes in leaf gas exchange and chlorophyll a fluorescence on soybean plants supplied with silicon and infected by Cercospora sojina. J Phytopathol 166:747–760. https://doi.org/10.1111/jph.12757
Ouda S (2015) Major crops and water scarcity in Egypt: irrigation water management under changing climate. Springer. https://doi.org/10.1007/978-3-319-21771-0
Peters FR, Datnoff L, Korndörfer G, Seebold K, Rush M (2001) Effect of silicon and host resistance on sheath blight development in rice. Plant Dis 85:827–832. https://doi.org/10.1094/PDIS.2001.85.8.827
Prasad M, Srinivasan R, Chaudhary M, Choudhary M, Jat LK (2019) Chapter seven - plant growth promoting Rhizobacteria (PGPR) for sustainable agriculture: perspectives and challenges. In: Singh AK, Kumar A, Singh PK (eds) PGPR amelioration in sustainable agriculture. Woodhead Publishing, Oxford, pp 129–157. https://doi.org/10.1016/B978-0-12-815879-1.00007-0
Rasoolizadeh A, Labbé C, Sonah H, Deshmukh RK, Belzile F, Menzies JG, Bélanger RR (2018) Silicon protects soybean plants against Phytophthora sojae by interfering with effector-receptor expression. BMC Plant Biol 18:97. https://doi.org/10.1186/s12870-018-1312-7
Rodrigues FÁ, Rios JA, Debona D, Aucique-Pérez CE (2017) Pyricularia oryzae-wheat interaction: physiological changes and disease management using mineral nutrition and fungicides. Trop Plant Pathol 42:223–229. https://doi.org/10.1007/s40858-017-0130-z
Saleh AA, Sharafaddin AH, El_Komy MH, Ibrahim YE, Hamad YK, Molan YY (2017) Fusarium species associated with date palm in Saudi Arabia. Eur J Plant Pathol 148:367–377. https://doi.org/10.1007/s10658-016-1095-3
Singh K (2011) Organic amendments to soil inoculated arbuscular mycorrhizal fungi and Pseudomonas fluorescens treatments reduce the development of root-rot disease and enhance the yield of Phaseolus vulgaris L. Eur J Soil Biol 47:288–295. https://doi.org/10.1016/j.ejsobi.2011.07.002
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic phosphotungstic acid reagents. Am J Enol Viticult 16:144–158
Sundaramoorthy S, Raguchander T, Ragupathi N, Samiyappan R (2012) Combinatorial effect of endophytic and plant growth promoting rhizobacteria against wilt disease of Capsicum annum L. caused by Fusarium solani. Biol Control 60:59–67. https://doi.org/10.1016/j.biocontrol.2011.10.002
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain treated bean plants: protective role of exogenous polyamines. Plant Sci 151:59–66. https://doi.org/10.1016/S0168-9452(99)00197-1
Wang M, Gao L, Dong S, Sun Y, Shen Q, Guo S (2017) Role of silicon on plant–pathogen interactions. Front Plant Sci 8:701. https://doi.org/10.3389/fpls.2017.00701
Zhou X, Shen Y, Fu X, Wu F (2018) Application of sodium silicate enhances cucumber resistance to Fusarium wilt and alters soil microbial communities. Front Plant Sci 9:624. https://doi.org/10.3389/fpls.2018.00624
Zucker M (1965) Induction of phenylalanine deaminase by light and its relation to chlorogenic acid synthesis in potato tuber tissue. Plant Physiol 40:779–784. https://doi.org/10.1104/pp.40.5.779
Zveibil A, Mor N, Gnayem N, Freeman S (2012) Survival, hostepathogen interaction, and management of Macrophomina phaseolina on strawberry in Israel. Plant Dis 96:265–272. https://doi.org/10.1094/PDIS-04-11-0299
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through research group No. RG-1440-029.
Author information
Authors and Affiliations
Contributions
MHK designed and performed the experiments, collected the data and wrote the manuscript. YEI, AAS, and YYM analyzed the experimental data and helped in writing the manuscript. All authors reviewed the manuscript critically.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
Authors declared that this manuscript have not published elsewhere. All authors read and approved the final version of this manuscript. The authors declare that the present work was developed without any potential conflict of interest, with no human or animal participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El_Komy, M.H., Ibrahim, Y.E., Saleh, A.A. et al. Integration of rhizobacterial mixture and silicon nutrition shows potential for the management of charcoal rot of sunflowers caused by Macrophomina phaseolina in semi-arid regions. J Plant Pathol 102, 1227–1239 (2020). https://doi.org/10.1007/s42161-020-00652-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-020-00652-w