Abstract
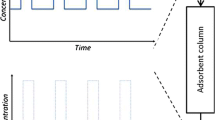
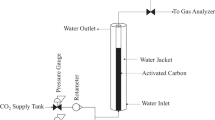
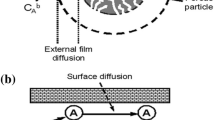
In spite of the adsorption of a solute onto porous material that can be described using the various models, the reliability of these models supported each other needs to be verified. In this work, the adsorption of NH4+ ions from domestic wastewater treatment plant effluent (DWTPE) onto commercial granular activated carbon (CGAC) was processed both in batch and column experiments. The Freundlich and Langmuir isotherm models and the pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models were used for the analysis of data obtained from batch experiments. The bed depth service time (BDST), Thomas, and modified mass transfer factor (MMTF) models were used to describe the behaviors of NH4+ adsorption by the CGAC adsorbent using the data collected from column experiments. The results showed that batch experimental data better matched by the Freundlich isotherm model and by PSO kinetic model indicate the state of multilayer adsorption supported by electrostatic interaction of NH4+ ions with chemical functional groups on the surface of CGAC. The application and limitations of the BDST and Thomas models in the prediction of NH4+ adsorption behaviors require the MMTF models to predict the adsorption kinetics and mass transfer mechanisms of NH4+ ions transported from the bulk water to acceptor sites on the surface of the CGAC adsorbent. The resistance of mass transfer verified for adsorption of NH4+ onto CGAC is dependent on porous diffusion and this may contribute to the development of such porous material for increasing the removal performance of NH4+ ions from DWTPE.
Highlights
-
Adsorption of NH4+ was conducted in batch and coumn experiments.
-
Reliability of different models to describe the adsorption of NH4+ onto CGAC was evaluated.
-
Comprehensive investigation of NH4+ adsorption used the MMTF models was verified.
-
Resistance of mass transfer for adsorption of NH4+ onto CGAC is dependent on porous diffusion.








Similar content being viewed by others
References
American Public Health Association (APHA) (2017) Standard methods for the examination of water and wastewater. 23rd ed. American Public Health Association, Washington, DC
Bimbo N, Smith JP, Aggarwal H, Physick AJ, Pugsley A, Barbour LJ, Ting VP, Mays TJ (2021) Kinetics and enthalpies of methane adsorption in microporous materials AX-21, MIL-101 (Cr) and TE7. Chem Eng Res Des 169:153–164. https://doi.org/10.1016/j.cherd.2021.03.003
Blanchard G, Maunaye M, Martin G (1984) Removal of heavy metals from waters by means of natural zeolites. Water Res 18:1501–1507. https://doi.org/10.1016/0043-1354(84)90124-6
Bohart GS, Adams EQ (1920) Some aspects of the behavior of charcoal with respect to chlorine. J Am Chem Soc 42:523–544. https://doi.org/10.1021/ja01448a018
Cai Y, Qi H, Liu Y, He X (2016) Sorption/desorption behavior and mechanism of NH4+ by biochar as a nitrogen fertilizer sustained-release material. J Agric Food Chem 64:4958–4964. https://doi.org/10.1021/acs.jafc.6b00109
Cruz H, Luckman P, Seviour T, Verstraete W, Laycock B, Pikaar I (2018) Rapid removal of ammonium from domestic wastewater using polymer hydrogels. Sci Rep 8:2912. https://doi.org/10.1038/s41598-018-21204-4
Cui X, Dai X, Khan KY, Li T, Yang X, He Z (2016) Removal of phosphate from aqueous solution using magnesium-alginate/chitosan modified biochar microspheres derived from Thalia dealbata. Bioresour Technol 218:1123–1132. https://doi.org/10.1016/j.biortech.2016.07.072
Deng Y, Huang S, Dong C, Meng Z, Wang X (2020) Competitive adsorption behaviour and mechanisms of cadmium, nickel and ammonium from aqueous solution by fresh and ageing rice straw biochars. Bioresour Technol 303:122853. https://doi.org/10.1016/j.biortech.2020.122853
Dong Y, Lin H, He Y (2017) Correlation between physicochemical properties of modified clinoptilolite and its performance in the removal of ammonia-nitrogen. Environ Monit Assess 189:107. https://doi.org/10.1007/s10661-017-5806-9
Eturki S, Ayari F, Jedidi N, Ben Dhia H (2012) Use of clay mineral to reduce ammonium from wastewater. Effect of various parameters. Surf Eng Appl Electrochem 48:276–283. https://doi.org/10.3103/S1068375512030064
Fan R, Chen C-L, Lin J-Y, Tzeng J-H, Huang C-P, Dong C, Huang CP (2019a) Adsorption characteristics of ammonium ion onto hydrous biochars in dilute aqueous solutions. Bioresour Technol 272:465–472. https://doi.org/10.1016/j.biortech.2018.10.064
Fan Z, Li Z, Sun Q, Cunha E, Kinloch IA, Ren H, Young RJ (2019b) Surface functionality analysis by Boehm titration of graphene nanoplatelets functionalized via a solvent-free cycloaddition reaction. Nanoscale Adv 1:1432–1441. https://doi.org/10.1039/c8na00280k
Fletcher AJ, Uygur Y, Thomas KM (2007) Role of surface functional groups in the adsorption kinetics of water vapor on microporous activated carbons. J Phys Chem C 111:8349–8359. https://doi.org/10.1021/jp070815v
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:384–470
Fulazzaky MA (2011) Determining the resistance of mass transfer for adsorption of the surfactants onto granular activated carbons from hydrodynamic column. Chem Eng J 166:832–840. https://doi.org/10.1016/j.cej.2010.11.052
Fulazzaky MA (2012) Analysis of global and sequential mass transfers for the adsorption of atrazine and simazine onto granular activated carbons from a hydrodynamic column. Anal Methods 4:2396–2403. https://doi.org/10.1039/C2AY05467A
Fulazzaky MA (2019) Study of the dispersion and specific interactions affected by chemical functions of the granular activated carbons. Environ Nanotechnol Monit Manage 12:100230. https://doi.org/10.1016/j.enmm.2019.100230
Fulazzaky MA, Khamidun MH, Omar R (2013) Understanding of mass transfer resistance for the adsorption of solute onto porous material from the modified mass transfer factor models. Chem Eng J 228:1023–1029. https://doi.org/10.1016/j.cej.2013.05.100
Fulazzaky MA, Khamidun MH, Din MFM, Yusoff ARM (2014) Adsorption of phosphate from domestic wastewater treatment plant effluent onto the laterites in a hydrodynamic column. Chem Eng J 258:10–17. https://doi.org/10.1016/j.cej.2014.07.092
Fulazzaky MA, Abdullah NH, Yusoff ARM, Paul E (2015) Conditioning the alternating aerobic–anoxic process to enhance the removal of inorganic nitrogen pollution from a municipal wastewater in France. J Clean Prod 100:195–201. https://doi.org/10.1016/j.jclepro.2015.03.043
Fulazzaky MA, Majidnia Z, Idris A (2017a) Mass transfer kinetics of Cd(II) ions adsorption by titania polyvinylalcohol-alginate beads from aqueous solution. Chem Eng J 308:700–709. https://doi.org/10.1016/j.cej.2016.09.106
Fulazzaky MA, Nuid M, Aris A, Muda K (2017b) Kinetics and mass transfer studies on the biosorption of organic matter from palm oil mill effluent by aerobic granules before and after the addition of Serratia marcescens SA30 in a sequencing batch reactor. Process Saf Environ Protect 107:259–268. https://doi.org/10.1016/j.psep.2017.02.016
Fulazzaky MA, Nuid M, Aris A, Muda K (2018) Mass transfer kinetics of biosorption of nitrogenous matter from palm oil mill effluent by aerobic granules in sequencing batch reactor. Environ Technol 39:2151–2161. https://doi.org/10.1080/09593330.2017.1351494
Fulazzaky MA, Nuid M, Aris A, Fulazzaky M, Sumeru K, Muda K (2019) Mass transfer kinetics of phosphorus biosorption by aerobic granules. J Water Process Eng 31:100889. https://doi.org/10.1016/j.jwpe.2019.100889
Fulazzaky MA, Salim NAA, Khamidun MH, Puteh MH, Yusoff ARM, Abdullah NH, Syafiuddin A, Zaini MAA (2021) The mechanisms and kinetics of phosphate adsorption onto iron-coated waste mussel shell observed from hydrodynamic column. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-021-03563-0
Galamini G, Ferretti G, Medoro V, Tescaro N, Faccini B, Coltorti M (2020) Isotherms, kinetics, and thermodynamics of NH4+ adsorption in raw liquid manure by using natural chabazite zeolite-rich tuff. Water 12:2944. https://doi.org/10.3390/w12102944
Hendriks ATWM, Langeveld JG (2017) Rethinking wastewater treatment plant effluent standards: nutrient reduction or nutrient control? Environ Sci Technol 51:4735–4737. https://doi.org/10.1021/acs.est.7b01186
Hu X, Zhang X, Ngo HH, Guo W, Wen H, Li C, Zhang Y, Ma C (2020) Comparison study on the ammonium adsorption of the biochars derived from different kinds of fruit peel. Sci Total Environ 707:135544. https://doi.org/10.1016/j.scitotenv.2019.135544
Huang WY, Zhu RH, He F, Li D, Zhu Y, Zhang YM (2013) Enhanced phosphate removal from aqueous solution by ferric-modified laterites: equilibrium, kinetics and thermodynamic studies. Chem Eng J 228:679–687. https://doi.org/10.1016/j.cej.2013.05.036
Huang J, Kankanamge NR, Chow C, Welsh DT, Li T, Teasdale PR (2018) Removing ammonium from water and wastewater using cost-effective adsorbents: a review. J Environ Sci 63:174–197. https://doi.org/10.1016/j.jes.2017.09.009
Hutchins RA (1973) New method simplifies design of activated carbon systems. Chem Eng 80:133–138
Juela D, Vera M, Cruzat C, Astudillo A, Vanegas E (2022) A new approach for scaling up fixed-bed adsorption columns for aqueous systems: a case of antibiotic removal on natural adsorbent. Process Saf Environ Protect 159:953–963. https://doi.org/10.1016/j.psep.2022.01.046
Karri RR, Sahu JN, Chimmiri V (2018) Critical review of abatement of ammonia from wastewater. J Mol Liq 261:21–31. https://doi.org/10.1016/j.molliq.2018.03.120
Kausar A, Bhatti HN, Iqbal M, Ashraf A (2017) Batch versus column modes for the adsorption of radioactive metal onto rice husk waste: conditions optimization through response surface methodology. Water Sci Technol 76:1035–1043. https://doi.org/10.2166/wst.2017.220
Kaya EMÖ, Özcan AS, Gök Ö, Özcan A (2013) Adsorption kinetics and isotherm parameters of naphthalene onto natural- and chemically modified bentonite from aqueous solutions. Adsorption 19:879–888. https://doi.org/10.1007/s10450-013-9542-3
Khadhri N, El Khames Saad M, Ben Mosbah M, Moussaoui Y (2019) Batch and continuous column adsorption of indigo carmine onto activated carbon derived from date palm petiole. J Environ Chem Eng 7:102775. https://doi.org/10.1016/j.jece.2018.11.020
Khalil A, Sergeevich N, Borisova V (2018) Removal of ammonium from fish farms by biochar obtained from rice straw: isotherm and kinetic studies for ammonium adsorption. Adsorpt Sci Technol 36:1294–1309. https://doi.org/10.1177/0263617418768944
Khamidun MH, Abdullah S, Ali UFM, Fulazzaky MA (2018) Analysis of ammonium adsorption behavior and mass transfer resistance from aerated lagoon effluent onto activated carbon. Malay J Anal Sci 22:723–734. https://doi.org/10.17576/mjas-2018-2204-19
Kizito S, Wu S, Wandera SM, Guo L, Dong R (2016) Evaluation of ammonium adsorption in biochar-fixed beds for treatment of anaerobically digested swine slurry: experimental optimization and modeling. Sci Total Environ 563–564:1095–1104. https://doi.org/10.1016/j.scitotenv.2016.05.149
Kober R, Kunzler DDF, Schwaab M, Steffani E, Barbosa-Coutinho E, Pinto JC (2021) Sequential experimental designs for discrimination among adsorption equilibrium models. Chem Eng Res Des 170:434–443. https://doi.org/10.1016/j.cherd.2021.04.025
Köse TE, Kıvanç B (2011) Adsorption of phosphate from aqueous solutions using calcined waste eggshell. Chem Eng J 178:34–39. https://doi.org/10.1016/j.cej.2011.09.129
Krsmanovic M, Biswas D, Ali H, Kumar A, Ghosh R, Dickerson AK (2021) Hydrodynamics and surface properties influence biofilm proliferation. Adv Colloid Interface Sci 288:102336. https://doi.org/10.1016/j.cis.2020.102336
Kuang Y, Zhang X, Zhou S (2020) Adsorption of methylene blue in water onto activated carbon by surfactant modification. Water 12:587. https://doi.org/10.3390/w12020587
Kumar A, Jena HM (2016) Preparation and characterization of high surface area activated carbon from Fox nut (Euryale ferox) shell by chemical activation with H3PO4. Result Phys 6(2016):651–658. https://doi.org/10.1016/j.rinp.2016.09.012
Kumari U, Mishra A, Siddiqi H, Meikap BC (2021) Effective defluoridation of industrial wastewater by using acid modified alumina in fixed-bed adsorption column: Experimental and breakthrough curves analysis. J Clean Prod 279:123645. https://doi.org/10.1016/j.jclepro.2020.123645
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403. https://doi.org/10.1021/ja02242a004
Le Moal M, Gascuel-Odoux C, Ménesguen A, Souchon Y, Étrillard C, Levain A, Moatar F, Pannard A, Souchu P, Lefebvre A, Pinay G (2019) Eutrophication: a new wine in an old bottle? Sci Total Environ 651:1–11. https://doi.org/10.1016/j.scitotenv.2018.09.139
Leszczynski J (2021) Removal of ammonium ions from aqueous solutions using weathered halloysite. Materials 14:4359. https://doi.org/10.3390/ma14164359
Li ZQ, Lu CJ, Xia ZP, Zhou Y, Luo Z (2007) X-ray diffraction patterns of graphite and turbostratic carbon. Carbon 45:1686–1695. https://doi.org/10.1016/j.carbon.2007.03.038
Liu Y, Shen L (2008) From Langmuir kinetics to first- and second-order rate equations for adsorption. Langmuir 24:11625–11630. https://doi.org/10.1021/la801839b
Liu J, Jiang J, Aihemaiti A, Meng Y, Yang M, Xu Y, Gao Y, Zou Q, Chen X (2019) Removal of phosphate from aqueous solution using MgO-modified magnetic biochar derived from anaerobic digestion residue. J Environ Manage 250:109438. https://doi.org/10.1016/j.jenvman.2019.109438
Mashifana T, Sithole T (2020) Recovery of silicon dioxide from waste foundry sand and alkaline activation of desilicated foundry sand. J Sustain Metall 6:700–714. https://doi.org/10.1007/s40831-020-00303-5
Mazloomi F, Jalali M (2017) Adsorption of ammonium from simulated wastewater by montmorillonite nanoclay and natural vermiculite: experimental study and simulation. Environ Monit Assess 189:415. https://doi.org/10.1007/s10661-017-6080-6
Mthombeni NH, Mbakop S, Ray SC, Leswifi T, Ochieng A, Onyango MS (2018) Highly efficient removal of chromium (VI) through adsorption and reduction: a column dynamic study using magnetized natural zeolite-polypyrrole composite. J Environ Chem Eng 6:4008–4017. https://doi.org/10.1016/j.jece.2018.05.038
Öztürk N, Kavak D (2005) Adsorption of boron from aqueous solutions using fly ash: batch and column studies. J Hazard Mater 127:81–88. https://doi.org/10.1016/j.jhazmat.2005.06.026
Pember N, Millar GJ, Couperthwaite SJ, De Bruyn M, Nuttall K (2016) BDST modelling of sodium ion exchange column behaviour with strong acid cation resin in relation to coal seam water treatment. J Environ Chem Eng 4:2216–2224. https://doi.org/10.1016/j.jece.2016.04.002
Radhika R, Jayalatha T, Rekha Krishnan G, Jacob S, Rajeev R, George BK (2018) Adsorption performance of packed bed column for the removal of perchlorate using modified activated carbon. Process Saf Environ Prot 117:350–362. https://doi.org/10.1016/j.psep.2018.04.026
Rashidi Nodeh H, Sereshti H, Zamiri Afsharian E, Nouri N (2017) Enhanced removal of phosphate and nitrate ions from aqueous media using nanosized lanthanum hydrous doped on magnetic graphene nanocomposite. J Environ Manage 197:265–274. https://doi.org/10.1016/j.jenvman.2017.04.004
Ren Z, Jia B, Zhang G, Fu X, Wang Z, Wang P, Lv L (2021) Study on adsorption of ammonia nitrogen by iron-loaded activated carbon from low temperature wastewater. Chemosphere 262:127895. https://doi.org/10.1016/j.chemosphere.2020.127895
Ribas MC, Adebayo MA, Prola LDT, Lima EC, Cataluña R, Feris LA, Puchana-Rosero MJ, Machado FM, Pavan FA, Calvete T (2014) Comparison of a homemade cocoa shell activated carbon with commercial activated carbon for the removal of reactive violet 5 dye from aqueous solutions. Chem Eng J 248:315–326. https://doi.org/10.1016/j.cej.2014.03.054
Rout PR, Bhunia P, Dash RR (2017) Evaluation of kinetic and statistical models for predicting breakthrough curves of phosphate removal using dolochar-packed columns. J Water Process Eng 17:168–180. https://doi.org/10.1016/j.jwpe.2017.04.003
Sabzehmeidani MM, Mahnaee S, Ghaedi M, Heidari H, Roy ALVAL (2021) Carbon based materials: a review of adsorbents for inorganic and organic compounds. Mater Adv 2:598–627. https://doi.org/10.1039/D0MA00087F
Seruga P, Krzywonos M, Pyzanowska J, Urbanowska A, Pawlak-Kruczek H, Niedzwiecki Ł (2019) Removal of ammonia from the municipal waste treatment effluents using natural minerals. Molecules 24:3633. https://doi.org/10.3390/molecules24203633
Shi M, Wang Z, Zheng Z (2013) Effect of Na+ impregnated activated carbon on the adsorption of NH4+-N from aqueous solution. J Environ Sci 25:1501–1510. https://doi.org/10.1016/S1001-0742(8)60227-7
Singh NB, Nagpal G, Agrawal S, Rachna D (2018) Water purification by using adsorbents: a review. Environ Technol Innov 11:187–240. https://doi.org/10.1016/j.eti.2018.05.006
Suchetana B, Rajagopalan B, Silverstein JA (2019) Investigating regime shifts and the factors controlling Total Inorganic Nitrogen concentrations in treated wastewater using non-homogeneous Hidden Markov and multinomial logistic regression models. Sci Total Environ 646:625–633. https://doi.org/10.1016/j.scitotenv.2018.07.194
Sumaraj XZ, Sarmah AK, Padhye LP (2020) Acidic surface functional groups control chemisorption of ammonium onto carbon materials in aqueous media. Sci Total Environ 698:134193. https://doi.org/10.1016/j.scitotenv.2019.134193
Syafiuddin A, Fulazzaky MA (2021) Decolorization kinetics and mass transfer mechanisms of Remazol Brilliant Blue R dye mediated by different fungi. Biotechnol Rep 29:e00573. https://doi.org/10.1016/j.btre.2020.e00573
Syafiuddin A, Salmiati S, Jonbi J, Fulazzaky MA (2018) Application of the kinetic and isotherm models for better understanding of the behaviors of silver nanoparticles adsorption onto different adsorbents. J Environ Manage 218:59–70. https://doi.org/10.1016/j.jenvman.2018.03.066
Tang SH, Ahmad Zaini MA (2020) Development of activated carbon pellets using a facile low-cost binder for effective malachite green dye removal. J Clean Prod 253:119970. https://doi.org/10.1016/j.jclepro.2020.119970
Torrik E, Soleimani M, Ravanchi MT (2019) Application of kinetic models for heavy metal adsorption in the single and multicomponent adsorption system. Int J Environ Res 13:813–828. https://doi.org/10.1007/s41742-019-00219-3
Wang L, Chen Z, Wen H, Cai Z, He C, Wang Z, Yan W (2018) Microwave assisted modification of activated carbons by organic acid ammoniums activation for enhanced adsorption of acid red 18. Powder Technol 323:230–237. https://doi.org/10.1016/j.powtec.2017.10.021
Yin Q, Zhang B, Wang R, Zhao Z (2017) Biochar as an adsorbent for inorganic nitrogen and phosphorus removal from water: a review. Environ Sci Pollut Res 24:26297–26309. https://doi.org/10.1007/s11356-017-0338-y
Yin Q, Liu M, Ren H (2019) Biochar produced from the co-pyrolysis of sewage sludge and walnut shell for ammonium and phosphate adsorption from water. J Environ Manage 249:109410. https://doi.org/10.1016/j.jenvman.2019.109410
Yuan MH, Chen YH, Tsai JY, Chang CY (2016) Ammonia removal from ammonia-rich wastewater by air stripping using a rotating packed bed. Process Saf Environ Prot 102:777–785. https://doi.org/10.1016/j.psep.2016.06.021
Yusuf M, Song K, Li L (2020) Fixed bed column and artificial neural network model to predict heavy metals adsorption dynamic on surfactant decorated graphene. Coll Surf a: Physicochem Eng Aspects. https://doi.org/10.1016/j.colsurfa.2019.124076
Zhai L, Bai Z, Zhu Y, Wang B, Luo W (2018) Fabrication of chitosan microspheres for efficient adsorption of methyl orange. Chin J Chem Eng 26:657–666. https://doi.org/10.1016/j.cjche.2017.08.015
Zhou Y, Wang L, Zhou Y, Mao XZ (2020) Eutrophication control strategies for highly anthropogenic influenced coastal waters. Sci Total Environ 705:135760. https://doi.org/10.1016/j.scitotenv.2019.135760
Zhu K, Fu H, Zhang J, Lv X, Tang J, Xu X (2012) Studies on removal of NH4+-N from aqueous solution by using the activated carbons derived from rice husk. Biomass Bioenergy 43:18–25. https://doi.org/10.1016/j.biombioe.2012.04.005
Zhu Y, Kolar P, Shah SB, Cheng JJ, Lim PK (2016) Avocado seed-derived activated carbon for mitigation of aqueous ammonium. Indus Crops Prod 92:34–41. https://doi.org/10.1016/j.indcrop.2016.07.016
Acknowledgements
This research was supported by the Ministry of Education (MOE) through Fundamental Research Grant Scheme (FRGS/1/2017/TK10/UTM/02/9). We also want to thank the Government of Malaysia, which provide MyBrain 15 program for sponsoring this work under the self-funded research grant and L00022 from the Ministry of Science, Technology and Innovation (MOSTI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Fulazzaky, M.A., Salim, N.A.A., Puteh, M.H. et al. Reliability of the Mass Transfer Factor Models to Describe the Adsorption of NH4+ by Granular Activated Carbon. Int J Environ Res 16, 30 (2022). https://doi.org/10.1007/s41742-022-00408-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41742-022-00408-7