Abstract
Background
Grapevine trunk diseases (GTDs) are an economically serious threat, affecting grapevine long-term sustainability and productivity. The use of antagonistic endophytic bacteria can be a promissing approach for GTDs’ management.
Results
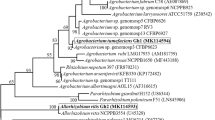
253 bacterial endophytes were isolated from the stems and roots of grapevines from 68 geographic locations in eight provinces of Iran. Based on biochemical properties, 22 strains were selected and identified using the 16S rRNA gene. The representative strains belonged to 13 genera of Agrobacterium, Arthrobacter, Bacillus, Chryseobacterium, Klebsiella, Kocuria, Pantoea, Pseudomonas, Rahnella, Rothia, Serratia, Staphylococcus and Variovorax. Using the dual culture method, antagonistic activity of 27 strains from 50 selected ones was shown against Fomitiporia mediterranea, a pathogen of GTDs; in which its hyphal growth was inhibited from 10 to 55%. Among the tested bacteria, Serratia plymuthica GI154, Bacillus sp. GI298 and Pseudomonas fluorescens GI310 showed the highest fungal growth inhibition zones, respectively. Co-treatment of GI154, GI298, and GI310 with F. mediterranea, in planta, could reduce the length of its internal canker by 54.41, 60.16 and 49.42% in compare with contol, respectively.
Conclusions
To the best of our knowledge, this is the first report of isolation of Arthrobacter agilis, Chryseobacterium daecheongense, Rahnella aquatilis, Serratia plymuthica, Staphylococcus epidermidis, Staphylococcus warneri and Variovorax paradoxus from grapevine in Iran. Moreover, this is the first report of Rothia amarae as a bacterial endophyte of the grapevine for the world. Grapevine bacterial endophytes exhibit auspicious biocontrol activities that can be utilized in sustainable and organic agriculture.






Similar content being viewed by others
Availability of data and materials
Almost all data generated or analyzed related to this topic, are included in this published article. The datasets which was not used directly in this article are available from the corresponding author on reasonable request.
Abbreviations
- GTDs:
-
Grapevine trunk diseases
- BCAs:
-
Biological control agents
- NA:
-
Nutrient agar
- HR:
-
Hypersensitive response
- NB:
-
Nutrient broth
- CTAB:
-
Cetyl trimethyl ammonium bromide
- RH:
-
Relative humidity
References
Aballay E, Prodan S, Mårtensson A, Persson P (2012) Assessment of rhizobacteria from grapevine for their suppressive effect on the parasitic nematode Xiphinema index. Crop Prot 42:36–41
Abed-Ashtiani F, Narmani A, Arzanlou M (2019) Analysis of Kalmusia variispora associated with grapevine decline in Iran. Eur J Plant Pathol 154:787–799
Aftab Uddin M, Akter S, Ferdous M, Haidar B, Amin A, Shofiul Islam Molla AHM, Khan H, Islam MR (2021) A plant endophyte Staphylococcus hominis strain MBL_AB63 produces a novel lantibiotic, homicorcin and a position one variant. Sci Rep 11:11211
Akbari E, Rasekh B, Beheshti Maal K, Karbasiun F, Yazdian F, Emami-Karvani Z, Peighami R (2021) A novel biosurfactant producing Kocuria rosea ABR6 as potential strain in oil sludge recovery and lubrication. AMB Expr 11:131
Akram W, Anjum T (2011) Quantitative changes in defense system of tomato induced by two strains of Bacillus against Fusarium wilt. Indian J Fundam 1(3):7–13
Alizadeh M (2017) Bacterial wetwood disease. Plant Chem Ecophysiol 2(1):1015
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215(3):403–410
Álvarez-Pérez JM, González-García S, Cobos R, Olego MÁ, Ibañez A, Díez-Galán A, Garzón-Jimeno E, Coque JJR (2017) Use of endophytic and rhizosphere actinobacteria from grapevine plants to reduce nursery fungal graft infections that lead to young grapevine decline. Appl Environ Microbiol 83(24):e01564-e1617
Amarloo OA, Mohammadi H, Mahdian SA, Tajick Ghanbary MA (2020) Identification and pathogenicity of fungal species associated with grapevine trunk diseases in Khorasan-Razavi province. Iran Mycol Iran 7(1):83–94
Arif N, Khullar S, Kumar R, Choudhary SK, Kapil A, Dhawan B (2019) Pleural effusion due to Chryseobacterium indologenes: case report and review of literature. J Lab Physicians 11(3):284–286
Asghari S, Harighi B, Mozafari AA, Esmaeel Q, Barka EA (2019) Screening of endophytic bacteria isolated from domesticated and wild growing grapevines as potential biological control agents against crown gall disease. Biocontrol 64:723–735
Baldan R, Cigana C, Testa F, Bianconi I, De Simone M, Pellin D, Di Serio C, Bragonzi A, Cirillo DM (2014) Adaptation of Pseudomonas aeruginosa in cystic fibrosis airways influences virulence of Staphylococcus aureus in vitro and murine models of co-infection. PLoS ONE 9(3):e89614
Basavand E, Khodaygan P, Ghelardini L, Rahimian H (2022) Isolation and identification of Rahnella victoriana associated with bacterial canker of Eucalyptus in Iran. For Pathol 52(3):e12743
Beck HE, Zimmermann NE, McVicar TR, Vergopolan N, Berg A, Wood EF (2018) Present and future Köppen-Geiger climate classification maps at 1-km resolution. Sci Data 30(5):180214
Brisse S, Grimont F, Grimont PAD (2006) The genus Klebsiella. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The Prokaryotes. Springer, New York, NY, pp 159–196
Bruisson S, Zufferey M, L’Haridon F, Trutmann E, Anand A, Dutartre A, De Vrieze M, Weisskopf L (2019) Endophytes and epiphytes from the grapevine leaf microbiome as potential biocontrol agents against phytopathogens. Front Microbiol 10:2726
Bruno G, Sparapano L, Graniti A (2007) Effects of three esca-associated fungi on Vitis vinifera L.: IV. Diffusion through the xylem of metabolites produced by two tracheiphilous fungi in the woody tissue of grapevine leads to esca-like symptoms on leaves and berries. Physiol Mol Plant Pathol 711(3):106–124
Brunori E, Farina R, Biasi R (2016) Sustainable viticulture: the carbon-sink function of the vineyard agro-ecosystem. Agric Ecosyst Environ 223:10–21
Burr TJ, Otten L (1999) Crown gall of grape: biology and disease management. Annu Rev Phytopathol 37:53–80
Campisano A, Antonielli L, Pancher M, Yousaf S, Pindo M, Pertot I (2014) Bacterial endophytic communities in the grapevine depend on pest management. PLoS ONE 9(11):e112763
Carrero P, Garrote JA, Pacheco S, García AI, Gil R, Carbajosa SG (1995) Report of six cases of human infection by Serratia plymuthica. J Clin Microbiol 33:275–276
Chen F, Guo YB, Wang JH, Li JY, Wang HM (2007) Biological control of grape crown gall by Rahnella aquatilis HX2. Plant Dis 91(8):957–963
Cobos R, Ibañez A, Diez-Galán A, Calvo-Peña C, Ghoreshizadeh S, Coque JJR (2022) The grapevine microbiome to the rescue: implications for the biocontrol of trunk diseases. Plants (basel) 11(7):840
Compant S, Mathieu F (2017) Biocontrol of major grapevine diseases: leading research. CABI, Wallingford, UK, pp 160–170
Compant S, Clément C, Sessitsch A (2010) Plant growth promoting bacteria in the rhizo and endosphere of plants. Their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem 42:669–678
Del Pilar M-D, Díaz-Losada E, Andrés-Sodupe M, Bujanda R, Maldonado-González MM, Ojeda S, Yacoub A, Rey P, Gramaje D (2021) Field evaluation of biocontrol agents against black-foot and Petri diseases of grapevine. Pest Manag Sci 77(2):697–708
FAO (2020). http://data.un.org/Data.aspx?d=FAO&f=itemCode%3A560
Farashiani A, Mousavi Jorf SA, Karimi MR (2012) Study of esca of grapevine in Bojnord. Iran J Plant Pathol 48(2):143–153
Fidan O, Zhan J (2019) Discovery and engineering of an endophytic Pseudomonas strain from Taxus chinensis for efficient production of zeaxanthin diglucoside. J Biol Eng 13:66
Fischer M (2002) A new wood-decaying basidiomycete species associated with esca of grapevine: Fomitiporia mediterranea (Hymenochaetales). Mycol Prog 1:315–324
Fokkema NJ (1978) Fungal antagonism in the phyllosphere. Ann Appl Biol 89(1):115–119
Furuya S, Mochizuki M, Aoki Y, Kobayashi H, Takayanagi T, Shimizu M, Suzuki S (2011) Isolation and characterization of Bacillus subtilis KS1 for the biocontrol of grapevine fungal diseases. Biocontrol Sci Technol 21(6):705–720
Galland D, Paul B (2001) Pythium perplexum isolated from soil in France: Morphology, molecular characterisation and biological control. Microbiol Res 156(2):185–189
Gerin D, Cariddi C, de Miccolis ARM, Rotolo C, Dongiovanni C, Faretra F, Pollastro S (2019) First report of Pseudomonas grapevine bunch rot caused by Pseudomonas syringae pv. syringae. Plant Dis 103(8):1954–1960
Grimont PAD, Grimont F, Richard C, Davis BR, Steigerwalt AG, Brenner DJ (1978) Deoxyribonucleic acid relatedness between Serratia plymuthica and other Serratia species, with a description of Serratia odorifera sp. nov. (Type strain: ICPB 3995). Int J Syst Bacteriol 28(4):453–463
Haidar R, Roudet J, Bonnard O, Cécile Dufour M, France Corio-Costet M, Fert M, Gautier T, Deschamps A, Fermaud M (2016) Screening and modes of action of antagonistic bacteria to control the fungal pathogen Phaeomoniella chlamydospora involved in grapevine trunk diseases. Microbiol Res 192:172–184
Haidar R, Amira Y, Roudet J, Marc F, Patrice R (2021) Application methods and modes of action of Pantoea agglomerans and Paenibacillus sp. to control the grapevine trunk disease-pathogen. Neofusicoccum Parvum. OENO One 55(3):1–16
Hall BH, McMahon RL, Noble D, Cottier EJ, McLintock D (2002) First report of Pseudomonas syringae on grapevines (Vitis vinifera) in South Australia. Australas Plant Pathol 31:421–422
Hallmann J, Berg G (2006) Spectrum and population dynamics of bacterial root endophytes. In: Schulz B, Boyle C, Sieber T (eds) Microbial root endophytes. Springer-Verlag, Berlin Heidelberg, pp 15–31
Hallmann J, Quadt-Hallmann A, Mahaffee WF, Kloepper JW (1997) Bacterial endophytes in agricultural crops. Can J Microbiol 43:895–914
Han JI, Choi HK, Lee SW, Orwin PM, Kim J, Laroe SL, Kim T, O’Neil J, Leadbetter JR, Lee SY, Hur CG, Spain JC, Ovchinnikova G, Goodwin L, Han C (2011) Complete genome sequence of the metabolically versatile plant growth-promoting endophyte Variovorax paradoxus S110. J Bacteriol 193(5):1183–1190
Kamath U, Singer C, Isenberg HD (1992) Clinical significance of Staphylococcus warneri bacteremia. J Clin Microbiol 30(2):261–264
Kim KK, Bae HS, Schumann P, Lee ST (2005) Chryseobacterium daecheongense sp. nov., isolated from freshwater lake sediment. Int J Syst Evol Microbiol 55(1):133–138
Klingner AE, Palleroni NJ, Pontis RE (1976) Isolation of Pseudomonas syringae from lesions on Vitis vinifera. J Phytopathol 86:107–116
Kobayashi DY, Palumbo JD (2000) Bacterial endophytes and their effects on plants and uses in agriculture. In: Bacon CW, White JF (eds) Microbial endophytes, 1st edn. Marcel Dekker Inc., New York, pp 199–233
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23(21):2947–2948
Leal C, Richet N, Guise JF, Gramaje D, Armengol J, Fontaine F, Trotel-Aziz P (2021) Cultivar contributes to the beneficial effects of Bacillus subtilis PTA-271 and Trichoderma atroviride SC1 to protect grapevine against Neofusicoccum parvum. Front Microbiol 12:726132
Malfanova N, Lugtenberg B, Berg G (2013) Bacterial endophytes: who and where, and what are they doing there. In: de Bruijn FJ (ed) Molecular microbial ecology of the rhizosphere. Wiley-Blackwell Hoboken, NJ, USA, pp 391–403
Mažeikien I, Frercks B, Burokien D, Maˇcionien I, Šalaševiˇcien A (2021) Endophytic community composition and genetic-enzymatic features of cultivable bacteria in Vaccinium myrtillus L. in forests of the Baltic–Nordic region. Forests 12:1647
Moore LW, Canfield M (1996) Biology of Agrobacterium and management of crown gall disease. In: Hall R (ed) Principles and practice of managing soil borne plant pathogens. APS Press, St. Paul, MN, USA, pp 151–191
Moradi-Amirabad Y, Rahimian H, Babaeizad V, Denman S (2019) Brenneria spp. and Rahnella victoriana associated with acute oak decline symptoms on oak and hornbeam in Iran. For Pathol 49:e12535
Niem JM, Billones-Baaijens R, Stodart B, Savocchia S (2020) Diversity profiling of grapevine microbial endosphere and antagonistic potential of endophytic Pseudomonas against grapevine trunk diseases. Front Microbiol 11:477
Nunes C, Usall N, Teixidó N, Viñas I (2001) Biological control of postharvest pear disease using a bacterium, Pantoea agglomerans CPA-2. Int J Food Microbiol 70(1–2):53–61
Nunes C, Usall J, Teixidó N, Fons E, Vinas I (2002) Post-harvest biological control by Pantoea agglomerans (CPA-2) on Golden Delicious apples. J Appl Microbiol 92(2):247–255
Pal KK, McSpadden Gardener B (2006) Biological control of plant pathogens. Plant Heal Instr. https://doi.org/10.1094/PHI-A-2006-1117-02
Pancher M, Ceol M, Corneo PE, Longa CM, Yousaf S, Pertot I, Campisano A (2012) Fungal endophytic communities in grapevines (Vitis vinifera L.) respond to crop management. Appl Environ Microbiol 78(12):4308–4317
Phukon M, Sahu P, Srinath R, Nithya A, Babu S (2013) Unusual occurrence of Staphylococcus warneri as endophyte in fresh fruits along with usual Bacillus spp. J Food Saf 33:102–106
Pinto C, Gomes AC (2016) Vitis vinifera microbiome: from basic research to technological development. Biocontrol 61:243–256
Platzer V, Schweigkofler W (2009) In vitro-efficacy of fungicides on F.mediterranea and Phaeomoniella chlamydospora, the causative pathogens of the esca-disease of grapevines. Mitt Klosterneuburg 59(2):74–83
Rhouma A, Ferchichi A, Hafsa M, Boubaker A (2004) Efficacy of the non pathogenic Agrobacterium strains K84 and K1026 against crown gall in Tunisia. Phytopathol Mediterr 43(2):167–176
Román-Ponce B, Wang D, Soledad Vásquez-Murrieta M, Feng Chen W, Estrada-de Los Santos P, Hua Sui X, Tao Wang E (2016) Kocuria arsenatis sp. nov., an arsenic-resistant endophytic actinobacterium associated with Prosopis laegivata grown on high-arsenic-polluted mine tailing. Int J Syst Evol Microbiol 66(2):1027–1033
Russi A, Almança MAK, Grohs DS, Schwambach J (2020) Biocontrol of black foot disease on grapevine rootstocks using Bacillus subtilis strain F62. Trop Plant Pathol 45:103–111
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425
SAS (2013) Statistical analysis software. Users’ guide statistics version 9.4. SAS Institute Inc., Cary
Schaad NW, Jones JB, Chun W (2001) Laboratory guide identification pathogenic bacteria: American Phytopathological Society. APS Press, Eagan, USA, p 373
Sherafati F, Khodaygan P, Azadvar M, Sedaghati E, Saberi-Riseh R, Baghaee-Ravari S (2014) Association of Pantoea agglomerans with the citrus bacterial canker disease in Iran. JCP 3(3):345–355
Siala R, Chobba IB, Vallaeys T, Triki MA, Jrad M, Cheffi M, Ayedi I, Elleuch A, Nemsi A, Cerqueira F, Gdoura R, Drira N, Gharsallah N (2016) Analysis of the cultivable endophytic bacterial diversity in the date palm (Phoenix dactylifera L.) and evaluation of its antagonistic potential against pathogenic Fusarium species that cause date palm bayound disease. J Appl Environ Microbiol 4(5):93–104
Syed Ab Rahman SF, Singh E, Pieterse CMJ, Schenk PM (2018) Emerging microbial biocontrol strategies for plant pathogens. Plant Sci 267:102–111
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30(12):2725–2729
Vaghari Souran SE, Shekariesfahlan A, Ashrafi F, Naeimi S, Ghasemi A (2021) Isolation and identification of grapevine endophytic bacteria in west Azarbaijan province. BCPP 9(1):131–143
Velázquez-Becerra C, Macías-Rodríguez LI, López-Bucio J, Flores-Cortez I, Santoyo G, Hernández-Soberano C, Valencia-Cantero E (2013) The rhizobacterium Arthrobacter agilis produces dimethylhexadecylamine, a compound that inhibits growth of phytopathogenic fungi in vitro. Protoplasma 250(6):1251–1262
Vicent JM (1947) Distortion of fungal hyphae in the presence of certain inhibitors. Nature 159:850
Vivas J, González JA, Barbeyto L, Rodríguez LA (2000) Identification of environmental Serratia plymuthica strains with the new Combo Panels Type 1S. Diagnosis 95(2):227–229
William S, Helene Feil A, Copeland A (2012) Bacterial genomic DNA isolation using CTAB. DOE Joint Genome Institute US Department of Energy, Walnut Creek, California, p 4
Wright SA, Zumoff CH, Schneider L, Beer SV (2001) Pantoea agglomerans strain EH318 produces two antibiotics that inhibit Erwinia amylovora in vitro. Appl Environ Microbiol 67(1):284–292
Yacoub S, Lam PK, le Vu HM, Le TL, Ha NT, Toan TT, Van NT, Quyen NT, Le Duyen HT, Van Kinh N, Fox A, Mongkolspaya J, Wolbers M, Simmons CP, Screaton GR, Wertheim H, Wills B (2016) Association of microvascular function and endothelial biomarkers with clinical outcome in dengue: an observational study. J Infect Dis 214(5):697–706
Yoon JH, Kang SJ, Oh TK (2007) Chryseobacterium daeguense sp. nov., isolated from wastewater of a textile dye works. Int J Syst Evol Microbiol 57:1355–1359
Zabalgogeazcoa I (2008) Fungal endophytes and their interaction with plant pathogens. Span J Agric Res 6:138–146
Zarraonaindia I, Owens SM, Weisenhorn P, West K, Hampton-Marcell J, Lax S, Bokulich NA, Mills DA, Martin G, Taghavi S, van der Lelie D, Gilbert JA (2015) The soil microbiome influences grapevine-associated microbiota. Mbio 6(2):02527–02614
Zhao X, Zhou ZJ, Han Y, Wang ZZ, Fan J, Xiao HZ (2013) Isolation and identification of antifungal peptides from Bacillus BH072, a novel bacterium isolated from honey. Microbiol Res 168(9):598–606
Acknowledgements
We would like to thank Masoud Amir Maafi (AREEO, Iran) and Shahram Shahrokhi (AREEO, Iran) for providing the phytotron facility.
Funding
This research project was sponsored by Iranian Research institute of Plant Protection and Islamic Azad University (North Tehran Branch).
Author information
Authors and Affiliations
Contributions
Conceptualization, AS, SEVS, SN and AG; methodology, SEVS, AS and AG; software, AS, SEVS, and AG; validation, AS, SEVS, SN, FA and AG; data analysis, SEVS, AS and AG; investigation, SEVS and AS; resources, AS, SEVS, and AG; data curation, AS; writing-original draft preparation, SEVS; writing-review and editing, AS and SN; supervision, AS and FA; funding acquisition, AS and SEVS. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vaghari Souran, S.E., Shekariesfahlan, A., Ashrafi, F. et al. Isolation and identification of grapevine endophytic bacteria with antagonistic potential against Fomitiporia mediterranea, a pathogen involved in grapevine trunk disease. J Plant Dis Prot 130, 1371–1384 (2023). https://doi.org/10.1007/s41348-023-00788-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-023-00788-8