Abstract
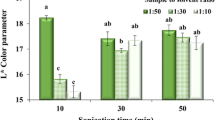
The olive tree (Olea europaea) holds great sociocultural significance in Mediterranean countries, particularly in Morocco, where the olive industry generates a large amount of by-products annually. Our goal is to convert this waste into therapeutic products that are rich in bioactive compounds. This study aimed to determine the impact of thermal activation (ranging from 25 to 100 °C) and the particle size of olive leaf powder (> 125 µm, between 50 and 125 µm, between 25 and 50 µm, or < 25 µm) on its phenolic profile and physical–chemical properties. The results revealed that exposing the powder to a thermal treatment of 80 °C and selecting particle sizes of < 25 µm resulted in the highest polyphenol content, antioxidant activity, and levels of pigments. Identification of phenolic compounds by chromatography (HPLC) showed the presence of hydroxytyrosol, catechin, caffeic acid, vanillin, naringin, oleuropein, quercetin and kaempferol at concentrations that varied according to the heat treatment and particle size. Furthermore, the analysis showed that oleuropein is the primary molecule found in olive leaves, with an average concentration ranging from 6 to 7.7%, depending on the temperature and particle size.





Similar content being viewed by others
Data availability
All the data used to support the findings of this study are included in the article.
Abbreviations
- TPC:
-
Polyphenol content
- TFC:
-
Total flavonoid content
- DPPH:
-
1,1-Diphenyl-2-picrylhydrazyl
- FRAP:
-
Ferric reducing antioxidant power
- ABTS:
-
2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)
- FM:
-
Fresh material
- DW:
-
Dry weight
- PCA:
-
Principal component analysis
- HPLC:
-
High-performance liquid chromatography
- AA:
-
Antioxidant activity
- Ash:
-
Ash content
- pH:
-
Potential of hydrogen
References
Aadesariya MK, Ram VR, Dave PN (2017) Evaluation of antioxidant activities by use of various extracts from Abutilon pannosum and Grewia tenax leaves in the Kachchh region. MOJ Food Process Technol 1:97–112
Abdel-Hameed E-SS, Nagaty MA, Salman MS, Bazaid SA (2014) Phytochemicals, nutritionals and antioxidant properties of two prickly pear cactus cultivars (Opuntia ficus indica Mill.) growing in Taif. KSA Food Chem 160:31–38. https://doi.org/10.1016/j.foodchem.2014.03.060
Altinyay Ç, Levent Altun M (2006) HPLC analysis of oleuropein in Olea europaea L. J Fac Pharm Ankara 35:1–11
Bamba S, Bekro JAM, Virieux D, Kabran GRM, Pirat JL, Bekro YA (2015) Analysis of a Rutaceae fat matter from Côte d’Ivoire. Der Chem Sin 6:47–50
Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239:70–76. https://doi.org/10.1006/abio.1996.0292
Bilgin M, Şahin S (2013) Effects of geographical origin and extraction methods on total phenolic yield of olive tree (Olea europaea) leaves. J Taiwan Inst Chem Eng 44:8–12. https://doi.org/10.1016/j.jtice.2012.08.008
Borello E, Domenici V (2019) Determination of pigments in virgin and extra-virgin olive oils: a comparison between two near UV-vis spectroscopic techniques. Foods 8(1):18. https://doi.org/10.3390/foods8010018
Borjan D, Leitgeb M, Knez Ž, Hrnčič MK (2020) Microbiological and antioxidant activity of phenolic compounds in olive leaf extract. Molecules 25:5946. https://doi.org/10.3390/molecules25245946
Buchner N, Krumbein A, Rohn S, Kroh LW (2006) Effect of thermal processing on the flavonols rutin and quercetin. Rapid Commun Mass Spectrom 20(21):3229–3235. https://doi.org/10.1002/rcm.2720
Chaaban H, Ioannou I, Chebil L, Slimane M, Gérardin C, Paris C, Charbonnel C, Chekir L, Ghoul M (2017) Effect of heat processing on thermal stability and antioxidant activity of six flavonoids. J Food Process Preserv 41(5):e13203. https://doi.org/10.1111/jfpp.13203
Cowan MM (1999) Plant products as antimicrobial agents. Clin Microbiol Rev 12:564–582. https://doi.org/10.1128/CMR.12.4.564
Criado-Navarro I, López-Bascón MA, Ledesma-Escobar CA, Priego-Capote F (2021) Alteration of the phenolic fraction of extra virgin olive oil subjected to frying conditions. ACS Food Sci Tech 1:884–891. https://doi.org/10.1021/acsfoodscitech.1c00077
Del Mar D-P, Priego-Capote F, de Castro MDL (2017) Selective ultrasound-enhanced enzymatic hydrolysis of oleuropein to its aglycon in olive (Olea europaea L.) leaf extracts. Food Chem 220:282–288. https://doi.org/10.1016/j.foodchem.2016.10.011
Dinu M, Pagliai G, Scavone F, Bellumori M, Cecchi L, Nediani C, Francesco S, Lisa G, Mulinacci N (2021) Effects of an olive by-product called pâté on cardiovascular risk factors. J Am Coll Nutr 40(7):617–623. https://doi.org/10.1080/07315724.2020.1813060
Djeridane A, Yousfi M, Nadjemi B, Boutassouna D, Stocker P, Vidal N (2006) Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem 97(4):654–660. https://doi.org/10.1016/j.foodchem.2005.04.028
El Adnany EM, Elhadiri N, Mourjane A, Ouhammou M, Hidar N, Jaouad A, Bitar K, Mahrouz M (2023) Impact and optimization of the conditions of extraction of phenolic compounds and antioxidant activity of olive leaves (Moroccan picholine) using response surface methodology. Separations 10(6):326. https://doi.org/10.3390/separations10060326
Fki I, Sayadi S, Mahmoudi A, Daoued I, Marrekchi R, Ghorbel H (2020) Comparative study on beneficial effects of hydroxytyrosol- and oleuropein-rich olive leaf extracts on high-fat diet-induced lipid metabolism disturbance and liver injury in rats. Biomed Res Int 2020:1–15. https://doi.org/10.1155/2020/1315202
Giuffrè A, Zappia C, Capocasale M (2017) Effects of high temperatures and duration of heating on olive oil properties for food use and biodiesel production. J Am Oil Chem Soc 94(6):819–830. https://doi.org/10.1007/s11746-017-2988-9
Giuffrè AM, Caracciolo M, Zappia C, Capocasale M, Poiana M (2018) Effect of heating on chemical parameters of extra virgin olive oil, pomace olive oil, soybean oil and palm oil. Ital J Food Sci 30:715–739. https://doi.org/10.14674/IJFS-1269
Harborne A (1998) Phytochemical methods a guide to modern techniques of plant analysis. Springer, Berlin, pp 89–90
Hynstova V, Sterbova D, Klejdus B, Hedbavny J, Huska D, Adam V (2018) Separation, identification and quantification of carotenoids and chlorophylls in dietary supplements containing Chlorella vulgaris and Spirulina platensis using high performance thin layer chromatography. J Pharm Biomed Anal 148:108–118. https://doi.org/10.1016/j.jpba.2017.09.018
Karković Marković A, Torić J, Barbarić M, Jakobušić Brala C (2019) Hydroxytyrosol, tyrosol and derivatives and their potential effects on human health. Molecules 24(10):2001. https://doi.org/10.3390/molecules24102001
Kiani R, Arzani A, Mirmohammady Maibody SAM (2021) Polyphenols, flavonoids, and antioxidant activity involved in salt tolerance in wheat, Aegilops cylindrica and their amphidiploids. Front Plant Sci 12:646221. https://doi.org/10.3389/fpls.2021.646221
Lama-Muñoz A, Del Mar CM, Espínola F, Moya M, Romero I, Castro E (2020) Content of phenolic compounds and mannitol in olive leaves extracts from six Spanish cultivars: extraction with the Soxhlet method and pressurized liquids. Food Chem 320:126626. https://doi.org/10.1016/j.foodchem.2020.126626
Li X, Zhou R, Xu K, Xu J, Jin J, Fang H, He Y (2018) Rapid determination of chlorophyll and pheophytin in green tea using Fourier transform infrared spectroscopy. Molecules 23(5):1010. https://doi.org/10.3390/molecules23051010
Liu Z, Sheng L, Canakapalli SS, Wang L (2022) Evaluation of the microbial control efficacies of commonly used home-drying and storage practices of dried peaches. LWT 165:113734. https://doi.org/10.1016/j.lwt.2022.113734
Makris DP, Rossiter JT (2000) Heat-induced, metal-catalyzed oxidative degradation of quercetin and rutin (quercetin 3-O-rhamnosylglucoside) in aqueous model systems. J Agric Food Chem 48(9):3830–3838. https://doi.org/10.1021/jf0001280
Mathur S, Agrawal D, Jajoo A (2014) Photosynthesis: response to high temperature stress. J Photochem Photobiol B 137:116–126. https://doi.org/10.1016/j.jphotobiol.2014.01.010
Medina E, Romero C, García P, Brenes M (2019) Characterization of bioactive compounds in commercial olive leaf extracts, and olive leaves and their infusions. Food Funct 10(8):4716–4724. https://doi.org/10.1039/C9FO00698B
Mercier S, Mondor M, McCarthy U, Villeneuve S, Alvarez G, Uysal I (2019) Optimized cold chain to save food. Saving Food. https://doi.org/10.1016/B978-0-12-815357-4.00007-9
Mohamed MB, Guasmi F, Ali SB, Radhouani F, Faghim JJ, Triki T, Grati Kammoun N, Baffi C, Lucini L, Benincasa C (2018) The LC-MS/MS characterization of phenolic compounds in leaves allows classifying olive cultivars grown in South Tunisia. Biochem Syst Ecol 78:84–90. https://doi.org/10.1016/j.bse.2018.04.005
Moura JDS, Sousa RPE, Martins LHDS, Costa CEFD, Chisté RC, Lopes AS (2023) Thermal degradation of carotenoids from jambu leaves (Acmella oleracea) during convective drying. Foods 12(7):1452. https://doi.org/10.3390/foods12071452
Mourjane A, Hanine H, El Adnany EM, Ouhammou M, Hidar N, Nabil B, Boumendjel A, Bitar K, Mahrouz M (2022) Energetic bio-activation of some organic molecules and their antioxidant activity in the pulp of the Moroccan argan tree Argania spinosa L. Molecules 27:3329. https://doi.org/10.3390/molecules27103329
Multari S, Marsol-Vall A, Keskitalo M, Yang B, Suomela JP (2018) Effects of different drying temperatures on the content of phenolic compounds and carotenoids in quinoa seeds (Chenopodium quinoa) from Finland. J Food Compost Anal 72:75–82. https://doi.org/10.1016/j.jfca.2018.06.008
Nabil B, Ouaabou R, Ouhammou M, Saadouni L, Mahrouz M (2020) Impact of particle size on functional, physicochemical properties and antioxidant activity of cladode powder (Opuntia ficus-indica). J Food Sci Technol 57:943–954. https://doi.org/10.1007/s13197-019-04127-4
Ouhammou M, Adnany EME, Mourjane A, Hammou HA, Bouchdoug M, Jaouad A, Mahrouz M (2022) Physico-chemical analysis and antioxidant activity of Moroccan caper leaves (Capparis spinosa L.). Euro-Mediterr J Environ Integr 7(3):407–414. https://doi.org/10.1007/s41207-022-00327-1
Oyaizu M (1986) Studies on products of browning reaction antioxidative activities of products of browning reaction prepared from glucosamine. Jpn J Nutr Diet 44:307–315. https://doi.org/10.5264/eiyogakuzashi.44.307
Patrón-Vázquez J, Baas-Dzul L, Medina-Torres N, Ayora-Talavera T, Sánchez-Contreras Á, García-Cruz U, Pacheco N (2019) The effect of drying temperature on the phenolic content and functional behavior of flours obtained from lemon wastes. Agron 9(9):474. https://doi.org/10.3390/agronomy9090474
Puigventós L, Navarro M, Alechaga É, Núñez O, Saurina J, Hernández-Cassou S, Puignou L (2015) Determination of polyphenolic profiles by liquid chromatography-electrospray-tandem mass spectrometry for the authentication of fruit extracts. Anal Bioanal Chem 407:597–608. https://doi.org/10.1007/s00216-014-8298-2
Ramírez EM, Brenes M, Romero C, Medina E (2023) Olive leaf processing for infusion purposes. Foods 12(3):591. https://doi.org/10.3390/foods12030591
Romani A, Mulas S, Heimler D (2017) Polyphenols and secoiridoids in raw material (Olea europaea L. leaves) and commercial food supplements. Eur Food Res Technol 243:429–435. https://doi.org/10.1007/s00217-016-2756-3
Sánchez-Gutiérrez M, Bascón-Villegas I, Rodríguez A, Pérez-Rodríguez F, Fernández-Prior Á, Rosal A, Carrasco E (2021) Valorisation of Olea europaea L. olive leaves through the evaluation of their extracts: antioxidant and antimicrobial activity. Foods 10(5):966. https://doi.org/10.3390/foods10050966
Soltani Y, Ali-Bouzidi M, Toumi F, Benyamina A (2017) Antioxidant activities of the extracts of the three organs of Juniperus phoenicea L. in western Algeria. Phytothérapie. https://doi.org/10.1007/s10298-017-1160-x
Sudjana AN, D’Orazio C, Ryan V, Rasool N, Ng J, Islam N, Riley T, Hammer K (2009) Antimicrobial activity of commercial Olea europaea (olive) leaf extract. Int J Antimicrob 33:461–463. https://doi.org/10.1016/j.ijantimicag.2008.10.026
Talhaoui N, Gómez-Caravaca AM, León L, De la Rosa R, Segura-Carretero A, Fernández-Gutiérrez A (2014) Determination of phenolic compounds of ‘Sikitita’ olive leaves by HPLC-DAD-TOF-MS. Comparison with its parents ‘Arbequina’ and ‘Picual’ olive leaves. LWT Food Sci Technol 58(1):28–34. https://doi.org/10.1016/j.lwt.2014.03.014
Talhaoui N, Gómez-Caravaca AM, Roldan C, Leon L, De la Rosa R, Fernandez-Gutierrez A, Segura-Carretero A (2015) Chemometric analysis for the evaluation of phenolic patterns in olive leaves from six cultivars at different growth stages. J Agric Food Chem 63(6):1722–1729. https://doi.org/10.1021/jf5058205
Tan H, Tuck K, Stupans I, Hayball P (2003) Simultaneous determination of oleuropein and hydroxytyrosol in rat plasma using liquid chromatography with fluorescence detection. J Chromatogr B 785(1):187–191. https://doi.org/10.1016/S1570-0232(02)00855-3
Turkmen N, Sari F, Velioglu Y (2005) The effect of cooking methods on total phenolics and antioxidant activity of selected green vegetables. Food Chem 93(4):713–718. https://doi.org/10.1016/j.foodchem.2004.12.038
Vezza T, Rodriguez-Nogales A, Algieri F, Mesa JG, Romero M, Sánchez M, Toral M, Martín-García B, Gómez-Caravaca AM, Arráez-Román D, Segura-Carretero A, Micol V, García F, Utrilla MP, Duarte J, Elena Rodríguez-Cabezas M, Gálvez J (2019) The metabolic and vascular protective effects of olive (Olea europaea L.) leaf extract in diet-induced obesity in mice are related to the amelioration of gut microbiota dysbiosis and to its immunomodulatory properties. Pharmacol Res 150:104487. https://doi.org/10.1016/j.phrs.2019.104487
Wang Y, Chantreau M, Sibout R, Hawkins S (2013) Plant cell wall lignification and monolignol metabolism. Front Plant Sci 4:220. https://doi.org/10.3389/fpls.2013.00220
Zaiter A, Becker L, Karam M-C, Dicko A (2016) Effect of particle size on antioxidant activity and catechin content of green tea powders. J Food Sci Technol 53:2025–2032. https://doi.org/10.1007/s13197-016-2201-4
Zielinska M, Michalska A (2016) Microwave-assisted drying of blueberry (Vaccinium corymbosum L.) fruits: drying kinetics, polyphenols, anthocyanins, antioxidant capacity, colour and texture. Food Chem 212:671–680. https://doi.org/10.1016/j.foodchem.2016.06.003
Acknowledgements
The authors thank the Ministry of Higher Education, Scientific Research and Vocational Training of Morocco, the National Center for Scientific and Technical Research, and the IRCOS Marrakech laboratories as well as Priority Research Program: PPR-Mahrouz-FS-UCA-Marrakech (R2B INNOVA) for financial support.
Funding
Science and Technology Innovative Research Team in Higher Educational Institutions of Hunan Province; Priority Research Program PPR-Mahrouz-FS-UCA-Marrakech for Mostafa Mahrouz; R2B INNOVA for Mostafa Mahrouz.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible Editor: Walid Elfalleh.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
El Adnany, E.M., Elhadiri, N., Mourjane, A. et al. Impact of heat treatment and granulometry of olive leaf powder (Moroccan picholine) on its phenolic compounds, antioxidant activity and physical–chemical properties. Euro-Mediterr J Environ Integr 9, 721–732 (2024). https://doi.org/10.1007/s41207-023-00455-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41207-023-00455-2