Abstract
Introduction
Thyrotropin stimulating hormone (TSH) suppression in patients with differentiated thyroid cancer (DTC) aims to decrease the growth and proliferation of thyroid cancer cells. However, the effect of TSH-suppressive therapy on bone microarchitecture remains undefined.
Methods
Cross-sectional study including 43 women with DTC undergoing TSH-suppressive therapy (sTSH) compared to 20 women also on levothyroxine (LT4) therapy but with TSH in the low-normal range (nTSH) since the thyroid surgery. Bone mineral density (BMD) was measured by dual-energy X-ray absorptiometry (DXA), and trabecular bone score (TBS) was evaluated using the TBS iNsigth software. Fracture risk assessed by FRAX, with and without TBS, was calculated. The relationship between suppressive therapy-related parameters and bone parameters was investigated.
Results
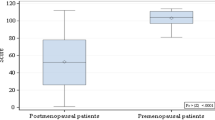
The TBS mean values were not significantly different in the sTSH and nTSH groups (1.273 ± 0.12 vs 1.307 ± 0.14, p = 0.7197). In both groups, postmenopausal women had degraded microarchitecture (TBS 1.216 ± 0.11 vs 1.213 ± 0.09, p = 0.9333), while premenopausal women had normal microarchitecture (1.328 ± 0.11 vs 1.401 ± 0.12, p = 0.195). The percentage of all postmenopausal women with degraded TBS was 54.7%, while the percentage of osteoporosis diagnoses was 16.1%. The TBS-adjusted FRAX-probability of fracture was similar in sTSH and nTSH groups. Body mass index (BMI) and menopausal status were the only variables associated with TBS and BMD.
Conclusion
Trabecular microarchitecture assessed by TBS was similar between women on long-term suppressive therapy in DTC and those on LT4 replacement therapy aiming at a TSH level within the low-normal reference range. Low TBS values were observed in postmenopausal women of both groups, suggesting that not only suppressed TSH levels but also a low-normal TSH is associated with deteriorated bone microarchitecture in postmenopausal women following total thyroidectomy.

Similar content being viewed by others
References
Seib CD, Sosa JA (2019) Evolving understanding of the epidemiology of thyroid cancer. EndocrinolMetabClin North Am 48(1):23–35. https://doi.org/10.1016/j.ecl.2018.10.002
Biondi B, Cooper DS (2019) Thyroid hormone suppression therapy. EndocrinolMetabClin North Am 48(1):227–237. https://doi.org/10.1016/j.ecl.2018.10.008
Grani G, Ramundo V, Verrienti A, Sponziello M, Durante C (2019) Thyroid hormone therapy in differentiated thyroid cancer. Endocrine 66:43–50. https://doi.org/10.1007/s12020-019-02051-3
Bassett JH, Williams GR (2016) Role of thyroid hormones in skeletal development and bone maintenance. Endocr Rev 37:135–187. https://doi.org/10.1210/er.2015-1106
Segna D, Bauer DC, Feller M, Schneider C, Fink HA, Aubert CE et al (2018) Association between subclinical thyroid dysfunction and change in bone mineral density in prospective cohorts. J Intern Med 283(1):56–72. https://doi.org/10.1111/joim.12688
Quan ML, Pasieka JL, Rorstad O (2002) Bone mineral density in well-differentiated thyroid cancer patients treated with suppressive thyroxine: a systematic overview of the literature. J SurgOncol 79:62–69. https://doi.org/10.1002/jso.10043
Heemstra KA, Hamdy NA, Romijn JA, Smit JW (2006) The effects of thyrotropin-suppressive therapy on bone metabolism in patients with well-differentiated thyroid carcinoma. Thyroid 16:583–591. https://doi.org/10.1089/thy.2006.16.583
Uzzan B, Campos J, Cucherat M, Nony P, Boissel JP, Perret GY (1996) Effects on bone mass of long term treatment with thyroid hormones: a meta-analysis. J ClinEndocrinolMetab 81(12):4278–4289. https://doi.org/10.1210/jcem.81.12.8954028
Sugitani I, Fujimoto Y (2011) Effect of postoperative thyrotropin suppressive therapy on bone mineral density in patients with papillary thyroid carcinoma: a prospective controlled study. Surgery 150(6):1250–1257. https://doi.org/10.1016/j.surg.2011.09.013
Wang LY, Smith AW, Palmer FL, Tuttle RM, Mahrous A, Nixon IJ et al (2015) Thyrotropin suppression increases the risk of osteoporosis without decreasing recurrence in ATA low- and intermediate-risk patients with differentiated thyroid carcinoma. Thyroid 25(3):300–307. https://doi.org/10.1089/thy.2014.0287
Mazziotti G, Formenti AM, Frara S, Olivetti R, Banfi G, Memo M et al (2018) High prevalence of radiological vertebral fractures in women on thyroid-stimulating hormone-suppressive therapy for thyroid carcinoma. J ClinEndocrinolMetab 103(3):956–964. https://doi.org/10.1210/jc.2017-01986
Papaleontiou M, Banerjee M, Reyes-Gastelum D, Hawley ST, Haymart MR (2019) Risk of osteoporosis and fractures in patients with thyroid cancer: a case-control study in U.S. Veterans Oncologist 24:1166–1173. https://doi.org/10.1634/theoncologist.2019-0234
Yoon BH, Lee Y, Oh HJ, Kim SH, Lee YK (2019) Influence of thyroid-stimulating hormone suppression therapy on bone mineral density in patients with differentiated thyroid cancer: a meta-analysis. J Bone Metab 26: 51–60. https://doi.org/10.11005/jbm.2019.26.1.51
Brancatella A, Marcocci C (2020) TSH suppressive therapy and bone. Endocr Connect 9:R158–R172. https://doi.org/10.1530/EC-20-0167
Vestergaard P, Rejnmark L, Mosekilde L (2005) Influence of hyper- and hypothyroidism, and the effects of treatment with antithyroid drugs and levothyroxine on fracture risk. Calcif Tissue Int 77(3):139–144. https://doi.org/10.1007/s00223-005-0068-x
Thayakaran R, Adderley NJ, Sainsbury C, Torlinska B, Boelaert K, Šumilo D et al (2019) Thyroid replacement therapy, thyroid stimulating hormone concentrations, and long term health outcomes in patients with hypothyroidism: longitudinal study. BMJ 366:l4892. https://doi.org/10.1136/bmj.l4892
Gonzalez Rodriguez E, Stuber M, Del Giovane C, Feller M, Collet TH, Löwe AL et al (2020) Skeletal effects of levothyroxine for subclinical hypothyroidism in older adults: a trust randomized trial nested study. J ClinEndocrinolMetabol. https://doi.org/10.1210/clinem/dgz058
Compston JE, McClung MR, Leslie WD (2019) Osteoporosis. Lancet 393:364–376. https://doi.org/10.1016/S0140-6736(18)32112-3
NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy (2001) Osteoporosis prevention, diagnosis, and therapy. JAMA 285(6):785–795. https://doi.org/10.1001/jama.285.6.785
Siris ES, Chen YT, Abbott TA, Barrett-Connor E, Miller PD, Wehren LE et al (2004) Bone mineral density thresholds for pharmacological intervention to prevent fractures. Arch Intern Med 164:1108–1112. https://doi.org/10.1001/archinte.164.10.1108
Silva BC, Leslie WD (2017) Trabecular bone score: a new DXA-derived measurement for fracture risk assessment. EndocrinolMetabClin North Am 46:153–180. https://doi.org/10.1016/j.ecl.2016.09.005
McCloskey EV, Odén A, Harvey NC, Leslie WD, Hans D, Johansson H et al (2016) A meta-analysis of trabecular bone score in fracture risk prediction and its relationship to FRAX. J Bone Miner Res 31:940–948. https://doi.org/10.1002/jbmr.2734
Martineau P, Silva BC, Leslie WD (2017) Utility of trabecular bone score in the evaluation of osteoporosis. CurrOpinEndocrinol Diabetes Obes 24:402–410. https://doi.org/10.1097/MED.0000000000000365
Moon JH, Kim KM, Oh TJ, Choi SH, Lim S, Park YJ et al (2017) The Effect of TSH suppression on vertebral trabecular bone scores in patients with differentiated thyroid carcinoma. J ClinEndocrinolMetab 102:78–85. https://doi.org/10.1210/jc.2016-2740
De Mingo Dominguez ML, Guadalix Iglesias S, Martin-Arriscado Arroba C, López Alvarez B, Martínez Diaz-Guerra G, Martinez-Pueyo JI et al (2018) Low trabecular bone score in postmenopausal women with differentiated thyroid carcinoma after long-term TSH suppressive therapy. Endocrine 62:166–173. https://doi.org/10.1007/s12020-018-1671-8
Kim EH, Jeon YK, Pak K, Kim IJ, Kim SJ, Shin S, et al (2019) Effects of thyrotropin suppression on bone health in menopausal women with total thyroidectomy. J Bone Metab 26: 31–8. https://doi.org/10.11005/jbm.2019.26.1.31
Iglesias SG, Dominguez MLM, Herrero EF, Martinez-Pueyo JI, Arroba CM, Diaz-Guerra GM et al (2019) Trabecular bone score and bone mineral density in patients with postsurgical hypoparathyroidism after total thyroidectomy for differentiated thyroid carcinoma. Surgery 165:814–819. https://doi.org/10.1016/j.surg.2018.10.034
Silva BC, Broy SB, Boutroy S, Schousboe JT, Shepherd JA, Leslie WD (2015) Fracture risk prediction by Non-BMD DXA measures: the 2015 ISCD official positions Part 2: trabecular bone score. J ClinDensitom 18(3):309–330. https://doi.org/10.1016/j.jocd.2015.06.008
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE et al (2016) 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American thyroid association guidelines task force on thyroid nodules and differentiated Thyroid cancer. Thyroid 26(1):1–133. https://doi.org/10.1089/thy.2015.0020
Assessment of fracture risk and its application to screening for postmenopausal osteoporosis (1994) Report of a WHO Study Group. World Health Organ Tech Rep Ser 843:1–129. https://pubmed.ncbi.nlm.nih.gov/7941614
Hans D, Goertzen AL, Krieg MA, Leslie WD (2011) Bone microarchitecture assessed by TBS predicts osteoporotic fractures independent of bone density: the Manitoba study. J Bone Miner Res 26:2762–2769. https://doi.org/10.1002/jbmr.499
Radominski SC, Bernardo W, Paula AP, Albergaria BH, Moreira C, Fernandes CE et al (2017) Brazilian guidelines for the diagnosis and treatment of postmenopausal osteoporosis. Rev Bras ReumatolEngl Ed 57(Suppl 2):452–466. https://doi.org/10.1016/j.rbre.2017.07.001
Rosner B (2016) Fundamentals of Biostatistics. Cengage Learning, Boston, USA
Hwangbo Y, Kim JH, Kim SW, Park YJ, Park DJ, Kim SY et al (2016) High-normal free thyroxine levels are associated with low trabecular bone scores in euthyroid postmenopausal women. OsteoporosInt 27(2):457–462. https://doi.org/10.1007/s00198-015-3270-3
Savvidis C, Tournis S, Dede AD (2018) Obesity and bone metabolism. Hormones (Athens) 17(2):205–217. https://doi.org/10.1007/s42000-018-0018-4
Langsetmo L, Vo TN, Ensrud KE, Taylor BC, Cawthon PM, Schwartz AV et al (2016) The association between trabecular bone score and lumbar spine volumetric bmd is attenuated among older men with high body mass index. J Bone Miner Res 31(10):1820–1826. https://doi.org/10.1002/jbmr.2867
Romagnoli E, Lubrano C, Carnevale V, Costantini D, Nieddu L, Morano S et al (2016) Assessment of trabecular bone score (TBS) in overweight/obese men: effect of metabolic and anthropometric factors. Endocrine 54(2):342–347. https://doi.org/10.1007/s12020-016-0857-1
Messina C, Buonomenna C, Menon G, Magnani S, Albano D, Gitto S et al (2019) Fat mass does not increase the precision error of trabecular bone score measurements. J ClinDensitom 22(3):359–366. https://doi.org/10.1016/j.jocd.2019.01.001
Kim MK, Yun KJ, Kim MH, Lim DJ, Kwon HS, Song KH et al (2015) The effects of thyrotropin-suppressing therapy on bone metabolism in patients with well-differentiated thyroid carcinoma. Bone 71:101–105. https://doi.org/10.1016/j.bone.2014.10.009
Greendale GA, Huang M, Cauley JA, Liao D, Harlow S, Finkelstein JS et al (2020) Trabecular bone score declines during the menopause transition: the study of women’s health across the nation (SWAN). J ClinEndocrinolMetab. https://doi.org/10.1210/clinem/dgz056
Iki M, Tamaki J, Kadowaki E, Sato Y, Dongmei N, Winzenrieth R et al (2014) Trabecular bone score (TBS) predicts vertebral fractures in Japanese women over 10 years independently of bone density and prevalent vertebral deformity: the Japanese Population-Based Osteoporosis (JPOS) cohort study. J Bone Miner Res 29:399–407. https://doi.org/10.1002/jbmr.2048
Vera L, Gay S, Campomenosi C, Paolino S, Pera G, Monti E et al (2016) Ten-year estimated risk of bone fracture in women with differentiated thyroid cancer under TSH-suppressive levothyroxine therapy. Endokrynol Pol 67(4):350–358. https://doi.org/10.5603/EP.a2016.0046
Vadiveloo T, Donnan PT, Murphy MJ, Leese GP (2013) Age- and gender-specific TSH reference intervals in people with no obvious thyroid disease in Tayside, Scotland: the Thyroid Epidemiology, Audit, and Research Study (TEARS). J ClinEndocrinolMetab 98(3):1147–1153. https://doi.org/10.1210/jc.2012-3191
Rosario PW, Xavier AC, Calsolari MR (2010) TSH reference values for adult Brazilian population. Arq Bras EndocrinolMetabol 54(7):603–606. https://doi.org/10.1590/s0004-27302010000700003
Kim DJ, Khang YH, Koh JM, Shong YK, Kim GS (2006) Low normal TSH levels are associated with low bone mineral density in healthy postmenopausal women. ClinEndocrinol (Oxf) 64(1):86–90. https://doi.org/10.1111/j.1365-2265.2005.02422.x
Kim TH, Joung JY, Kang M, Choi SK, Kim K, Jang JY et al (2015) A modest protective effect of thyrotropin against bone loss is associated with plasma triiodothyronine levels. PLoS ONE 10(12):145292. https://doi.org/10.1371/journal.pone.0145292
Morris MS (2007) The association between serum thyroid-stimulating hormone in its reference range and bone status in postmenopausal American women. Bone 40(4):1128–1134. https://doi.org/10.1016/j.bone.2006.12.001
Leader A, Ayzenfeld RH, Lishner M, Cohen E, Segev D, Hermoni D (2014) Thyrotropin levels within the lower normal range are associated with an increased risk of hip fractures in euthyroid women, but not men, over the age of 65 years. J ClinEndocrinolMetab 99(8):2665–2673. https://doi.org/10.1210/jc.2013-2474
Murphy E, Glüer CC, Reid DM, Felsenberg D, Roux C, Eastell R et al (2010) Thyroid function within the upper normal range is associated with reduced bone mineral density and an increased risk of nonvertebral fractures in healthy euthyroid postmenopausal women. J ClinEndocrinolMetab 95(7):3173–3181. https://doi.org/10.1210/jc.2009-2630
Zhu H, Zhang J, Wang J, Zhao X, Gu M (2020) Association of subclinical thyroid dysfunction with bone mineral density and fracture: a meta-analysis of prospective cohort studies. Endocrine 67(3):685–698. https://doi.org/10.1007/s12020-019-02110-9
Lee K, Lim S, Park H, Woo HY, Chang Y, Sung E et al (2020) Subclinical thyroid dysfunction, bone mineral density, and osteoporosis in a middle-aged Korean population. OsteoporosInt 31(3):547–555. https://doi.org/10.1007/s00198-019-05205-1
Acknowledgements
We thank the densitometry service of the Jenny Faria de Andrade Institute, especially the doctors Olívio Brito Mallheiros and Carlyle Marques Barral and the assistants Maria do Socorro de Souza and Elizabeth Aparecida de Souza.
Funding
This research did not receive any specific grant from any funding agency.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Researches Ethics Committees of Federal University of Minas Gerais – UFMG—and Instituto Mario Penna.
Consent to participate and for publication
Informed consent was obtained from all participants and the document is attached.
Availability of data and material
Contact the author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sousa, B.É.C.A., Silva, B.C., de Oliveira Guidotti, T. et al. Trabecular bone score in women with differentiated thyroid cancer on long-term TSH-suppressive therapy. J Endocrinol Invest 44, 2295–2305 (2021). https://doi.org/10.1007/s40618-021-01537-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-021-01537-0