Abstract
Background
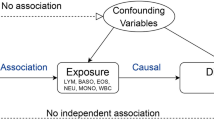
Observational studies have suggested an association between white blood cells (WBCs) and frailty, but considering the susceptibility to reverse causality and confounding, the causal direction and magnitude of this association remain ambiguous. Our aim was to investigate the causal effect of WBCs on frailty by means of a Mendelian randomization (MR) analysis.
Methods
Based on the genome-wide association study (GWAS) summary statistics data provided by the European Bioinformatics Institute (EBI), we carried out a two-sample MR study. We applied the genetically predicted independent WBCs from GWAS as a measure of exposure data. The Rockwood Frailty Index (FI) was used as outcome measure, which was derived from a meta-analysis from GWAS in UK Biobank European ancestry participants and Swedish TwinGene participants. Our study applied inverse variance weighted (IVW), weighted median, Mendelian randomization-Egger (MR-Egger) and outlier test (MR-PRESSO) methods to explore relationships between various WBCs and frailty.
Results
In our study, a possible causal relationship between eosinophil levels and frailty was demonstrated by two-sample MR analysis. Eosinophils were associated with FI (beta:0.0609; 95% CI 0.0382, 0.0836; P = 1.38E–07). Our results suggest that as the level of eosinophils increases, so does the risk of frailty. No meaningful causal relationship between neutrophils, lymphocytes, monocytes or basophils and FI was found in the MR results (P > 0.05).
Conclusions
According to this MR study, higher eosinophil counts are related to an increased risk of frailty. To validate these findings and investigate the mechanisms underlying these connections, future studies are warranted.



Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Dent E, Martin FC, Bergman H et al (2019) Management of frailty: opportunities, challenges, and future directions. Lancet 394:1376–1386. https://doi.org/10.1016/S0140-6736(19)31785-4
Hoogendijk EO, Afilalo J, Ensrud KE et al (2019) Frailty: implications for clinical practice and public health. Lancet 394:1365–1375. https://doi.org/10.1016/S0140-6736(19)31786-6
Kojima G, Iliffe S, Walters K (2018) Frailty index as a predictor of mortality: a systematic review and meta-analysis. Age Ageing 47:193–200. https://doi.org/10.1093/ageing/afx162
Mitnitski AB, Mogilner AJ, Rockwood K (2001) Accumulation of deficits as a proxy measure of aging. ScientificWorldJournal 1:323–336. https://doi.org/10.1100/tsw.2001.58
Ferrucci L, Fabbri E (2018) Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol 15:505–522. https://doi.org/10.1038/s41569-018-0064-2
Yao X, Li H, Leng SX (2011) Inflammation and immune system alterations in frailty. Clin Geriatr Med 27:79–87. https://doi.org/10.1016/j.cger.2010.08.002
Li H, Manwani B, Leng SX (2011) Frailty, inflammation, and immunity. Aging Dis 2:466–473
Rea IM, Gibson DS, McGilligan V et al (2018) Age and age-related diseases: role of inflammation triggers and cytokines. Front Immunol 9:586. https://doi.org/10.3389/fimmu.2018.00586
Honda T, Uehara T, Matsumoto G et al (2016) Neutrophil left shift and white blood cell count as markers of bacterial infection. Clin Chim Acta 457:46–53. https://doi.org/10.1016/j.cca.2016.03.017
Fernandez-Garrido J, Ruiz-Ros V, Navarro-Martínez R et al (2018) Frailty and leucocyte count are predictors of all-cause mortality and hospitalization length in non-demented institutionalized older women. Exp Gerontol 103:80–86. https://doi.org/10.1016/j.exger.2018.01.007
Leng SX, Xue Q-L, Tian J et al (2007) Inflammation and frailty in older women. J Am Geriatr Soc 55:864–871. https://doi.org/10.1111/j.1532-5415.2007.01186.x
Sleiman PMA, Grant SFA (2010) Mendelian randomization in the era of genomewide association studies. Clin Chem 56:723–728. https://doi.org/10.1373/clinchem.2009.141564
Sekula P, Del Greco MF, Pattaro C et al (2016) Mendelian randomization as an approach to assess causality using observational data. J Am Soc Nephrol 27:3253–3265. https://doi.org/10.1681/ASN.2016010098
Boehm FJ, Zhou X (2022) Statistical methods for Mendelian randomization in genome-wide association studies: a review. Comput Struct Biotechnol J 20:2338–2351. https://doi.org/10.1016/j.csbj.2022.05.015
Smith GD, Ebrahim S (2003) “Mendelian randomization”: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol 32:1–22. https://doi.org/10.1093/ije/dyg070
Lv J, Wu L, Sun S et al (2023) Smoking, alcohol consumption, and frailty: a Mendelian randomization study. Front Genet 14:1092410. https://doi.org/10.3389/fgene.2023.1092410
Mourtzi N, Georgakis M, Ntanasi E et al (2023) Genetically downregulated Interleukin-6 signalling is associated with a lower risk of frailty. Age Ageing. https://doi.org/10.1093/ageing/afac318
Tomata Y, Wang Y, Hägg S et al (2022) Protein nutritional status and frailty: a Mendelian randomization study. J Nutr 152:269–275. https://doi.org/10.1093/jn/nxab348
Tomata Y, Wang Y, Hägg S et al (2021) Fatty acids and frailty: a mendelian randomization study. Nutrients 13:3539. https://doi.org/10.3390/nu13103539
Astle WJ, Elding H, Jiang T et al (2016) The allelic landscape of human blood cell trait variation and links to common complex disease. Cell 167:1415-1429.e19. https://doi.org/10.1016/j.cell.2016.10.042
Atkins JL, Jylhävä J, Pedersen NL et al (2021) A genome-wide association study of the frailty index highlights brain pathways in ageing. Aging Cell 20:e13459. https://doi.org/10.1111/acel.13459
Burgess S, Thompson SG, CRP CHD Genetics Collaboration (2011) Avoiding bias from weak instruments in Mendelian randomization studies. Int J Epidemiol 40:755–764. https://doi.org/10.1093/ije/dyr036
Blodgett J, Theou O, Kirkland S et al (2015) Frailty in NHANES: comparing the frailty index and phenotype. Arch Gerontol Geriatr 60:464–470. https://doi.org/10.1016/j.archger.2015.01.016
Rockwood K, Mitnitski A (2007) Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci 62:722–727. https://doi.org/10.1093/gerona/62.7.722
Burgess S, Thompson SG (2017) Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol 32:377–389. https://doi.org/10.1007/s10654-017-0255-x
Brion M-JA, Shakhbazov K, Visscher PM (2013) Calculating statistical power in Mendelian randomization studies. Int J Epidemiol 42:1497–1501. https://doi.org/10.1093/ije/dyt179
Burgess S, Thompson SG (2015) Multivariable Mendelian randomization: the use of pleiotropic genetic variants to estimate causal effects. Am J Epidemiol 181:251–260. https://doi.org/10.1093/aje/kwu283
Franceschi C, Bonafè M, Valensin S et al (2000) Inflamm-aging. An evolutionary perspective on immunosenescence. Ann N Y Acad Sci 908:244–254. https://doi.org/10.1111/j.1749-6632.2000.tb06651.x
Soysal P, Stubbs B, Lucato P et al (2016) Inflammation and frailty in the elderly: a systematic review and meta-analysis. Ageing Res Rev 31:1–8. https://doi.org/10.1016/j.arr.2016.08.006
Cheng Z, He D, Li J et al (2022) C-reactive protein and white blood cell are associated with frailty progression: a longitudinal study. Immun Ageing 19:29. https://doi.org/10.1186/s12979-022-00280-1
Immune System Dysfunction in the Elderly-PubMed. https://pubmed.ncbi.nlm.nih.gov/28423084/. Accessed 1 May 2023
Compté N, Bailly B, De Breucker S et al (2015) Study of the association of total and differential white blood cell counts with geriatric conditions, cardio-vascular diseases, seric IL-6 levels and telomere length. Exp Gerontol 61:105–112. https://doi.org/10.1016/j.exger.2014.11.016
Leng SX, Hung W, Cappola AR et al (2009) White blood cell counts, insulin-like growth factor-1 levels, and frailty in community-dwelling older women. J Gerontol A Biol Sci Med Sci 64:499–502. https://doi.org/10.1093/gerona/gln047
Samson LD, Engelfriet P, Verschuren WMM et al (2022) Impaired JAK-STAT pathway signaling in leukocytes of the frail elderly. Immun Ageing 19:5. https://doi.org/10.1186/s12979-021-00261-w
Rothenberg ME, Hogan SP (2006) The eosinophil. Annu Rev Immunol 24:147–174. https://doi.org/10.1146/annurev.immunol.24.021605.090720
Fernández-Garrido J, Navarro-Martínez R, Buigues-González C et al (2014) The value of neutrophil and lymphocyte count in frail older women. Exp Gerontol 54:35–41. https://doi.org/10.1016/j.exger.2013.11.019
Valdiglesias V, Sánchez-Flores M, Maseda A et al (2015) Lymphocyte subsets in a population of nonfrail elderly individuals. J Toxicol Environ Health A 78:790–804. https://doi.org/10.1080/15287394.2015.1051170
Marcos-Pérez D, Sánchez-Flores M, Maseda A et al (2018) Frailty in older adults is associated with plasma concentrations of inflammatory mediators but not with lymphocyte subpopulations. Front Immunol 9:1056. https://doi.org/10.3389/fimmu.2018.01056
Admission neutrophil-to-lymphocyte ratio and monocyte-to-lymphocyte ratio to predict 30-day and 1-year mortality in geriatric hip fractures-PubMed. https://pubmed.ncbi.nlm.nih.gov/32739153/. Accessed 1 May 2023
Cybularz M, Wydra S, Berndt K et al (2021) Frailty is associated with chronic inflammation and pro-inflammatory monocyte subpopulations. Exp Gerontol 149:111317. https://doi.org/10.1016/j.exger.2021.111317
Acknowledgements
Our data, analytic methods or materials are available to other researchers. We used summary statistics from a genome-wide association study (GWAS) and then performed two-sample MR analyses. Details are in the section of the original article. Since the data can be available to everyone, the studies reported in the manuscript were not pre-registered.This study was funded by the National Key R&D Program of China (2020YFC2008900), the National Natural Science Foundation of China (81701366), Zhejiang Public Welfare Technology Application Program Project (LGF22H250001), the Medical Health Science and Technology Project of Zhejiang Province(2022RC149).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Ethics for this research was obtained from Ethics Review Committee of the First Affiliated Hospital of Zhejiang University School of Medicine.
Informed consent
All participants signed written informed consent forms.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, L., Zhao, Xh., Zhou, Sx. et al. Genetic predisposition to white blood cells in relation to the risk of frailty. Aging Clin Exp Res 35, 3023–3031 (2023). https://doi.org/10.1007/s40520-023-02609-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-023-02609-2