Abstract
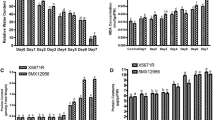
Tomato being sensitive to high temperature experiences mild to high temperature stresses under climate change conditions. To understand the response of tomato genotypes to mild temperature stress, a study was conducted in temperature gradient tunnel facility. The results revealed that across the genotypes studied, specific activity of antioxidant enzymes viz., superoxide dismutase (SOD), peroxidase (POX) and glutathione reductase (GR) increased significantly. Among the genotypes, increase in SOD activity was highest in cv. Arka Vikas, followed by IIHR 2195 and least in Abhinava. The GR activity was highest in Abhinava, followed by IIHR 2195 and least in cv. Arka Vikas. The mild temperature stress caused reduction in catalase (CAT) activity. The decrease in CAT activity and concomitant increase in POX activity was observed in cv. Arka Vikas. Low leaf water potential (Ψleaf) and higher electrolyte leakage indicated that the membrane integrity was affected across the tomato genotypes even under mild temperature stress. Among the genotypes studied, cv. Arka Vikas showed greater activity of SOD and POX, higher membrane stability and least reduction in water potential under mild temperature stress.
Similar content being viewed by others
References
Almeselmani, M., Deshmukh, P. S., Sairam, R. K., Kushwaha, S. R., & Singh, T. P. (2006). Protective role of antioxidant enzymes under high temperature stress. Plant Science, 171, 382–388.
Camejo, D., Alarcon, J. J., Torres, W., Rodriguez, P., & Dell Amico, J. M. (2002). Changes induced by high temperatures in photosynthesis and antioxidant response on two genotypes of tomato (Lycopresicon esculentum M.). Cultivos Tropicales, 23(4), 33–37.
Camejo, D., Rodriguez, P., Morales, M. A., Dell Amico, J. M., Torrecillas, A., & Alarcon, J. J. (2005). High temperature effects on photosynthetic activity of two tomato cultivars with different heat susceptibility. Journal of Plant Physiology, 162(3), 281–289.
Foyer, C. H. (2002). The contribution of photosynthetic oxygen metabolism to oxidative stress in plants. In Dirk Inze & Marc Van Montagu (Eds.), Oxidative stress in plants (pp. 33–66). New York: Taylor and Francis.
Geisenberg, C., & Stewart, K. (1986). Field crop management. In J. G. Atherton & J. Rudich (Eds.), The tomato crop (pp. 511–557). New York: Chapman and Hall.
IPCC. (2012). Summary for policymakers. In C. B. Field, V. Barros, T. F. Stocker, D. Qin, D. J. Dokken, K. L. Ebi, M. D. Mastrandrea, K. J. Mach, G. K. Plattner, S. K. Allen, M. Tignor, & P. M. Midgley (Eds.), Managing the risks of extreme events and disasters to advance climate change adaptation. A special report of working groups i and ii of the intergovernmental panel on climate change (pp. 1–19). Cambridge: Cambridge University Press.
Islam, M. T. (2011). Effect of temperature on photosynthesis, yield attributes and yield of tomato genotypes. International Journal of Experimental Agriculture, 2(1), 8–11.
Masia, A. (1998). Superoxide dismutase and catalase activities in apple during ripening and post harvest and with special reference to ethylene. Physiologia Plantarum, 104, 668–672.
Mavis, R. D., & Stellwagen, E. (1968). Purification and subunit structure of glutathione reductase from bakers’ yeast. Journal of Biological Chemistry, 243, 809–814.
Morales, D., Rodrıguez, P., Dell’amico, J., Nicolas, E., Torrecillas, A., & Sanchez-Blanco, M. J. (2003). High temperature preconditioning and thermal shock imposition affects water relations, gas exchange and root hydraulic conductivity in tomato. Biologia Plantarum, 47, 203–208.
Parvaiz, A., Maryam, S., & Satyawati, S. (2008). Reactive oxygen species, antioxidants and signaling in plants. Journal of Plant Biology, 51(3), 167–173.
Peet, M. M., Willits, D. H., & Gardner, R. (1997). Response of ovule development and post-pollen production processes in male-sterile tomatoes to chronic, sub-acute high temperature stress. Journal of Experimental Botany, 48, 101–111.
Rainwater, D. T., Gossett, D. R., Millhollonm, E. P., Hanna, H. Y., Banks, S. W., & Lucas, M. C. (1996). The relationship between yield and the antioxidant defense system in tomatoes grown under heat stress. Free Radical Research, 25(5), 421–435.
Rivero, R. M., Ruiz, J. M., & Romero, L. (2004). Oxidative metabolism in tomato plants subjected to heat stress. Journal of Horticulture Science and Biotechnology, 79, 560–564.
Sato, S., Kamiyama, M., Iwata, T., Makita, N., Furukawa, H., & Ikeda, H. (2006). Moderate increase of mean daily temperature adversely affects fruit set of Lycopersicon esculentum by disrupting specific physiological processes in male reproductive development. Annals of Botany London, 97, 731–738.
Subhas, C. M. (1990). Enzymatic properties association with resistance to rust and powdery mildew in peas. Indian Journal of Horticulture, 47, 341–345.
Yadav, I. S. (1998). Vegetable varieties released by Indian institute of horticultural research, technical Bulletin 2. Bangalore: IIHR.
Zhanyuan, D., & Bramlage, W. J. (1994). Superoxide dismutase activities in senescing apple fruit (Malus domesticva Borkh.). Journal of Food Science, 59(3), 581–584.
Acknowledgments
This work is a part of Indian Council for Agriculture Research (ICAR), New Delhi, India, funded project National Initiative on Climate Resilient Agriculture (NICRA). We are grateful to the Director, Indian Institute of Horticulture Research (IIHR), Bangalore for the facilities. We would also like to acknowledge Mr. Sridhar, C. for the technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laxman, R.H., Rao, N.K.S., Biradar, G. et al. Antioxidant enzymes activity and physiological response of tomato (Lycopersicon esculentum M.) genotypes under mild temperature stress. Ind J Plant Physiol. 19, 161–164 (2014). https://doi.org/10.1007/s40502-014-0091-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-014-0091-x