Abstract
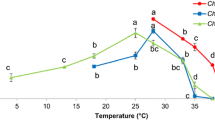
Red macroalga Pyropia spiralis thrives in the upper intertidal zone and demonstrates remarkable resilience to environmental fluctuations, attributed to protective mechanisms against factors like solar radiation, temperature, and desiccation. The aim of this is study was assess the thermosensitivity and tolerance of P. spiralis, subjecting to five temperatures (15, 20, 25, 30, and 35 °C) evaluating growth rate, photosynthetic performance, and chemical composition. The hypothesis states that higher temperatures will reduce growth and photosynthesis, overstimulate metabolism, degrade pigment composition, and trigger the synthesis of protective metabolites for stabilizing cellular structures under elevated temperature conditions. The study revealed thermotolerance between 15 and 25 °C, characterized by stable energy dissipation and minimal pigment changes. Yet, at 30 and 35 °C, there was significant thermosensitivity, leading to a notable drop in photosynthesis and pigments. Carbon-to-nitrogen (C:N) ratio, notably below 20 (9.58 at 15 °C and 7.86 at 25 °C), indicated a balanced growth characterized by optimal proportions of minerals, carbohydrates, and proteins. P. spiralis ' nutritional traits make it a promising candidate for use in seaweed-based products. Analysis of specific amino acids highlighted an upward trend at 25 °C, while higher temperatures induced a thermal-response profile associated with osmoprotectants, primary stress signals, and glutathione biosynthesis. Additionally, lower temperatures were linked to increased soluble carbohydrates, whereas higher temperatures inversely impacted protein accumulation. It is crucial to note that, being just one variable, the study provides a partial perspective, highlighting the importance of future, multifactorial analyses.





Similar content being viewed by others
Data availability
The original contributions presented in the study are include in the article. Further inquiries can be directed to the corresponding author.
References
Adams J, Ross A, Anastasakis K, Hodgson E, Gallagher J, Jones J, Donnison I (2011) Seasonal variation in the chemical composition of the bioenergy feedstock Laminaria digitata for thermochemical conversion. Bioresour Technol 102:226–234. https://doi.org/10.1016/j.biortech.2010.06.152n
AOAC Official Methods of Analysis (2001). Total dietary fiber in foods and food products with ≤ 2% starch: non-enzimatic-gravimetric method. In: The Association of Official Analytical Chemists, 17th edition, Gaithersburg, USA, Methods 993.21.
Bradford M (1976) A rapid sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brown M, Frazer A, Brasch D, Melton L (1990) Growth and reproduction of Porphyra columbina Mont. (Bangiales, Rhodophyceae) from southern New Zealand. J Appl Phycol 2:35–44. https://doi.org/10.1007/BF02179767
Che Y, Fu A, Hou X, McDonald K, Buchanan B, Huang W, Luanb S (2013) C-terminal processing of reaction center protein D1 is essential for the function and assembly of photosystem II in Arabidopsis. Proc Natl Acad Sci 40:16247–16252. https://doi.org/10.1073/pnas.1313894110
Contreras-Porcia L, Thomas D, Flores V, Correa J (2011) Tolerance to oxidative stress induced by desiccation in Porphyra columbina (Bangiales, Rhodophyta). J Exp Bot 62:1815–1829. https://doi.org/10.1093/jxb/erq364
Corzo A, Niell F (1991) C/N ratio in response to nitrogen supply and light quality in Ulva rigida C. Agardh (Chlorophyta: Ulvophyceae). Sci Mar 55:405–411
Dawczynski C, Schubert R, Jahreis G (2007) Amino acids, fatty acids, and dietary fiber in edible seaweed product. Food Chem 103:891–899. https://doi.org/10.1016/j.foodchem.2006.09.041
Diniz G, Barbarino E, Oiano-Neto J, Pacheco S, Lourenço S (2011) Gross chemical profile and calculation of nitrogen-to-protein conversion factors for five tropical seaweeds. Am J Plant Sci 2:287–296. https://doi.org/10.4236/ajps.2011.23032
Duarte C (1992) Nutrient concentration of aquatic plants: patterns across species. Limnol Oceanogr 37:882–889. https://doi.org/10.4319/lo.1992.37.4.0882
Dudgeon S, Davison I, Vadas R (1989) Effect of freezing on photosynthesis of intertidal macroalgae: relative tolerance of Chondrus crispus and Mastocarpus stellatus (Rhodophyta). Mar Biol 101:107–114. https://doi.org/10.1007/BF00393483
Edwards P (1970) Illustrated guide to the seaweeds and sea grasses in the vicinity of Porto Aransas, Texas
Egydio A, Santa Catarina C, Floh E, Santos D (2013) Free amino acid composition of Annona (Annonaceae) fruit species of economic interest. Ind Crops Prod 45:373–376. https://doi.org/10.1016/j.indcrop.2012.12.033
Figueroa F, Bonomi J, Malta E, Conde-Álvarez R, Nitschke U, Arenas F, Mata M, Connan S, Abreu M, Marquardt R, Vaz-Pinto F, Konotchick T, Celis-Plá P, Ordoñez M, Ruiz E, Flores P, Los De Ríos J, Kirke D, Chow F, Nassar C, Robledo D, Pérez-Ruzafa A, Bañares-España E, Altamirano M, Jiménez C, Korbee N, Bischof K, Stengel D (2014) Short-term effects of increasing CO2, nitrate and temperature on three mediterranean macroalgae: biochemical composition. Aquatic Biol 22:177–193. https://doi.org/10.3354/ab00610
Grzymski J, Johnsen G, Sakshug E (1997) The significance of intracellular self-shading on the bio-optical properties of brown, red and green macroalgae. J Phycol 33:408–414. https://doi.org/10.1111/j.0022-3646.1997.00408.x
Guidi L, Degl’Innocenti E (2012) Chlorophyll a fluorescence in abiotic stress. In: Venkateswarlu B, Shanker A, Shanker C, Maheswari M (eds) Crop stress and its management: perspectives and strategies. Springer, Berlin, pp 359–398
Hernandez I, Corzo A, Gordillo J, Saez E, Fernandez J, Niell R (1993) Seasonal cycle of the gametophytic form of Porphyra umbilicalis: nitrogen and carbon. Mar Ecol Progr Ser 3:301–331. https://doi.org/10.3354/meps099301
Hobday A, Lisa V, Alexander S, Perkins D, Smale S, Straub E, Oliver J, Benthuysen JA et al (2016) A hierarchical approach to defining marine heatwaves. Prog Oceanogr 141:227–238. https://doi.org/10.1016/j.pocean.2015.12.014
Houvinen P, Gómez I, Orostegui M (2007) Patterns and UV sensitivity of carbon anhydrase and nitrate reductase activities in south pacific macroalgae. Mar Biol 151:1813–1821. https://doi.org/10.1007/s00227-007-0613-4
Jahns P, Holzwarth A (2012) The role of the xanthophyll cycle and lutein in photoprotection of photosystem II. Biochim Biophys Acta 1817:182–193. https://doi.org/10.1016/j.bbabio.2011.04.012
Jaleel K, Parameswaran U, Gopal A, Manokaran S, Joydas T (2022) Spatio-temporal variations of benthic communities along the coast. In: Ecology and biodiversity of benthos. pp 287–313. https://doi.org/10.1016/B978-0-12-821161-8.00003-9
Jassby A, Platt T (1976) Mathematical formulation of the relationship between photosynthesis and light for phytoplankton. Limnol Oceanogr 21:540–547. https://doi.org/10.4319/lo.1976.21.4.0540
Joly A, Yamaguishi N (1963) The life history of Porphyra atropurpurea (Olivi) de Toni. Bol Fac Filos Ciênc Let Univ São Paulo. 19:1–17
Kim J, Kraemer G, Neefus C, Chung K, Yarish C (2007) Effects of temperature and ammonium on growth, pigment production and nitrogen uptake by four species of Porphyra (Bangiales, Rhodophyta) native to the New England coast. J Appl Phycol 19:431–440. https://doi.org/10.1007/s10811-006-9150-7
Kramer D, Johnson G, Kiirats O, Edwards G (2004) New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth Res 79:209–218. https://doi.org/10.1023/B:PRES.0000015391.99477.0d
Krishnaiah D, Sarbatly R, Prasad D, Bono A (2008) Mineral content of some seaweeds from Sabah’s South China Sea. Asian J Sci Res 1:166–170. https://doi.org/10.3923/ajsr.2008.166.170
Kumar S, Sahoo D, Levine I (2015) Assessment of nutritional value in a brown seaweed Sargassum wightii and their seasonal variations. Algal Res 9:117–125. https://doi.org/10.1016/j.algal.2015.02.024
Kumar V (1993) Biochemical constituents of marine algae from Tuticorin coast. Indian J Mar Sci 22:138–140
Kursar T, Van Der Meer J, Albert R (1983) Light-harvesting system of the red alga Gracilaria tikvahiae. I. Biochemical analyses of pigments mutation. Plant Physiol 73:353–360. https://doi.org/10.1104/pp.73.2.353
Lage-Yusty M, Caramés-Adán P, López-Hernández J (2013) Determination of phycobiliproteins by constant-wavelength synchronous spectrofluorometric method in red algae. J Food 11:243–247. https://doi.org/10.1080/19476337.2012.728629
Lalegerie F, Lajili S, Taupin L, Stiger-Pouvreaua V, Connan S (2019) Photo-protective compounds in red macroalgae from brittany: considerable diversity in mycosporine-like amino acids (MAAs). Mar Envir Res 147:37–48. https://doi.org/10.1016/j.marenvres.2019.04.001
Lichtenthaler H, Buschmann C (2001) Chlorophylls and carotenoids: measurement and characterization by UV-VIS spectroscopy. Curr Protocol Food Anal Chem F4:1. https://doi.org/10.1002/0471142913.faf0403s01
Louda J, Li L, Liu L, Winfree N, Baker E (1998) Chlorophyll-a degradation during cellular senescence and death. Org Geochem 29:1233–1251. https://doi.org/10.1016/S0146-6380(98)00186-7
Marinho-Soriano E, Fonseca P, Carneiro M, Moreira W (2006) Seasonal variation in the chemical composition of two tropical seaweeds. Biores Technol 97:2402–2406. https://doi.org/10.1016/j.biortech.2005.10.014
Matsuko T, Minami A, Iwasaki N, Majima T, Nishimura S, Lee Y (2005) Carbohydrate analysis by a phenol-sulfuric acid method in microplate format. Anal Biochem 339:69–72. https://doi.org/10.1016/j.ab.2004.12.001
Mercado J, Jiménez C, Niell F, Figueroa F (1996) Comparison of methods for measuring light absorption by algae and their application to the estimation of package effect. Science Mar 60:39–45
Monselise E, Parola A, Kose D (2005) Low-frequency electromagnetic fields induce a stress effect upon higher plants as evident by the universal stress signal alanine. Biochem Biophys Res Commun 302:427–434. https://doi.org/10.1016/S0006-291X(03)00194-3
Mourandi-Givernaud A, Givernaud T, Morvan H, Cosson J (1993) Annual variations of the biochemical composition of Gelidium latifolium (greville) thuret et Bornet. Hydrobiol 260:607–612. https://doi.org/10.1007/BF00049078
Navarro N, Houvinen P, Gómez I (2016) Stress tolerance of antarctic macroalgae in the early life stages. Rev Chil Hist Nat 89:5–14. https://doi.org/10.1186/s40693-016-0051-0
Nissim I, Hardy M, Pleasure J, Nisim I, States B (1992) A mechanism of glycine and alanine cytoprotective action: stimulation of stress-induced HSP70 mRNA. Kidney Int 42:775–782. https://doi.org/10.1038/ki.1992.347
Patarra R, Paiva L, Neto A, Lima E, Baptista J (2011) Nutritional value of selected macroalgae. J Appl Phycol 23:205–208. https://doi.org/10.1007/s10811-010-9556-0
Pedersen A, Kraemer G, Yarish C (2004) The effects of temperature and nutrient concentrations on nitrate and phosphate uptake in different species of Porphyra from Long Island Sound (USA). J Exp Mar Biol Ecol 312:235–252. https://doi.org/10.1016/j.jembe.2004.05.021
Peñalver R, Lorenzo J, Ros G, Amarowicz R, Pateiro M, Nieto G (2020) Seaweeds as a functional ingredient for a healthy diet. Mar Drugs 18:301–328. https://doi.org/10.3390/md18060301
Penniman C, Mathieson A, Penniman C (1986) Reproductive phenology and growth of Gracilaria tikvahiae McLachlan (Gigartinales, Rhodophyta) in the Great Bay Estuary. Bot Mar 29:147–154. https://doi.org/10.1515/botm.1986.29.2.147
Ramus J, Rosenberg G (1980) Diurnal photosynthetic performance of seaweeds measured under natural conditions. Mar Biol 56:21–28. https://doi.org/10.1007/BF00390590
Renaud S, Luong-Van J (2006) Seasonal variation in the chemical composition of tropical Australian marine macroalgae. J Appl Phycol 18:381–387. https://doi.org/10.1007/s10811-006-9034-x
Rioux L, Beaulieu L, Turgeon S (2017) Seaweeds: a traditional ingredients for new gastronomic sensation. Food Hydrocoll 68:255–265. https://doi.org/10.1016/j.foodhyd.2017.02.005
Roleda M, Hurd C (2019) Seaweed nutrient physiology: application of concepts to aquaculture and bioremediation. Phycologia 58:552–562. https://doi.org/10.1080/00318884.2019.1622920
Rosenberg C, Ramus J (1982) Ecological growth strategies in the seaweeds Gracilaria foliifera (Rhodophyceae) and Ulva sp (Chlorophyceae): soluble nitrogen and reserve carbohydrates. Mar Biol 66:251–259. https://doi.org/10.1007/BF00397030
Ruangchuay R, Masahiro M (2003) Physiological responses of blade and conchocelis of Porphyra vietnamensis Tanaka et Pham-Hoang Ho (Bangiales, Rhodophyta) from Thailand in culture. Algae 18:21–28. https://doi.org/10.4490/ALGAE.2003.18.1.021
Ruperez P (2002) Mineral content of edible marine seaweed. Food Chem 79:23–26. https://doi.org/10.1016/S0308-8146(02)00171-1
Salehi B, Sharifi-Rad J, Seca A, Pinto D, Michalak I, Trincone A, Mishra A, Nigam M, Zam W, Martins N (2019) Current trends on seaweeds: looking at chemical composition, phytopharmacology, and cosmetic applications. Molecules 24:4182–4321. https://doi.org/10.3390/molecules24224182
Santa-Catarina C, Silveira V, Balbuena T, Viana A, Estelita M, Handro W, Floh E (2006) IAA, ABA, polyamines and free amino acids associated with zygotic embryo development of Ocotea catharinensis. Plant Growth Regul 49:237–247. https://doi.org/10.1007/s10725-006-9129-z
Schmidt É, Felix M, Kreusch M, Pereira D, Costa G, Simioni C, Ouriques L, Steiner N, Chow F, Floh E, Ramlov F, Maraschin M, Bouzon Z (2016) Profiles of carotenoids and amino acids and total phenolic compounds of the red alga Pterocladiella capillacea exposed to cadmium and different salinities. J Appl Phycol 28:1955–1963. https://doi.org/10.1007/s10811-015-0737-8
Schreiber U, Neubaer C (1990) O2-dependent electron flow, membrane energization and the mechanism of non-photochemical quenching of chlorophyll fluorescence. Photosynth Res 25:279–293. https://doi.org/10.1007/BF00033169
Schubert N, Garcia-Mendoza E, Pacheco-Ruiz I (2006) Carotenoid composition of marine algae. J Phycol 42:1208–1216. https://doi.org/10.1111/j.1529-8817.2006
Sudatti D, Fujii M, Rodrigues S, Turra A, Pereira R (2011) Effects of abiotic factors on growth and chemical defenses in cultivated clones of Laurencia dendroidea J. Agardh (Ceramiales, Rhodophyta). Mar Biol 158:1439–1446. https://doi.org/10.1007/s00227-011-1660-4
Terada R, Yuge T, Watanabe Y, Mine T, Morikawa T, Nishihara G (2020) Chronic effects of three different stressors, irradiance, temperature, and desiccation, on the PSII photochemical efficiency in the heteromorphic life-history stages of cultivated Pyropia yezoensis f. narawaensis (Bangiales) from Japan. J Appl Phycol. https://doi.org/10.1007/s10811-020-02152-x
Urrea-Victoria V, Nardelli A, Floh E, Chow F (2020) Sargassum stenophyllum (Fucales, Ochrophyta) responses to temperature short-term exposure: photosynthesis and chemical composition. Braz J Bot 43:733–745. https://doi.org/10.1007/s40415-020-00639-y
Ursi S, Plastino E (2001) Crescimento in vitro de linhagens de coloração vermelha e marrom-esverdeada clara de Gracilaria sp. (Gracilariales, Rhodophyta) em dois meios de cultura: análise de diferentes estádios reprodutivos. Braz J Bot 24:587–594. https://doi.org/10.1590/S0100-84042001000500014
Watanabe Y, Morikama T, Mine T, Kawamura Y, Nishihara G, Terada R (2017) Chronological change and the potential of recovery on the photosynthetic efficiency of Pyropia yezoensis f. narawaensis (Bangiales) during the sporelings frozen storage treatment in the Japanese Nori cultivation. Phycol Res 65:265–271. https://doi.org/10.1111/pre.12185
Watanabe Y, Nishihara G, Tokunaga S, Terada R (2014) Effect of irradiance and temperature on the photosynthesis of a cultivated red alga, Pyropia tenera (=Porphyra tenera), at the southern limit of distribution in Japan. Phycol Res 62:187–196. https://doi.org/10.1111/pre.12053
Watanabe Y, Yamada H, Mine T, Kawamura Y, Nishihara G, Terada R (2016) Photosynthetic responses of Pyropia yezoensis f. narawaensis (Bangiales, Rhodophyta) to a thermal and PAR gradient vary with the life-history stage. Phycologia 55:665–672. https://doi.org/10.2216/16-25.1
Ye Y, Zhang L, Yang R, Luo Q, Chen H, Yan X, Tang H (2013) Metabolic phenotypes associated with high-temperature tolerance of Porphyra haitanensis strains. J Agric Food Chem 61:8356–8363. https://doi.org/10.1021/jf402749a
Acknowledgements
VUV acknowledges PhD fellowships financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES; Finance Code 001) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP; 2014/25073-3). FC acknowledges the financial support of FAPESP for the BIOTA-FAPESP Grant (2013/50731-1) and CNPq Research Productivity Grant (303937/2015-7). We also thanks Dra. Eny I.S. Floh who kindly provided equipment and infrastructure for amino acids analysis.
Author information
Authors and Affiliations
Contributions
VUV conceived the conceptualization, methodologies, formal analysis, writing original draft, review and editing versions. FC conceived the idea, conceptualization, collection, methodologies, formal analysis, funding acquisition, supervision, data discussion and interpretation, and writing, review and editing versions.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Urrea-Victoria, V., Chow, F. Short-term thermal exposure effects on growth rate, photosynthetic performance, and chemical composition in Pyropia spiralis (Rhodophyta) from the brazilian coast: insights and limitations. Braz. J. Bot (2024). https://doi.org/10.1007/s40415-024-01010-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40415-024-01010-1