Abstract
Background
Heart failure affects over 1 million people in Germany and contributes to morbidity, mortality, and high healthcare costs. A recent large randomized controlled trial compared the novel compound sacubitril/valsartan (LCZ696) with the angiotensin-converting enzyme (ACE) inhibitor enalapril and found a 16% reduction in mortality hazard. In Germany, sacubitril/valsartan was launched at the beginning of 2016.
Objective
The purpose of this study was to conduct a post hoc analysis of the cost effectiveness, budget impact, and disease burden reduction of sacubitril/valsartan compared with ACE inhibitors for patients with heart failure from the perspective of the German social health insurance (SHI), based on the results of this trial.
Methods
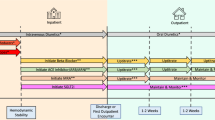
A Markov (cohort) state transition model was constructed to simulate treatment over a remaining lifetime. Based on the Markov model, a dynamic population model was developed that projects the incidence, prevalence, mortality, and healthcare costs of heart failure in the SHI population from 2017 to 2060. The population model follows prevalent and incident cohorts over time. Each year a new cohort is added, while the existing cohorts age by 1 year or die. To test for sensitivity of results, a Monte Carlo simulation was run.
Results
Based on the price negotiated between manufacturer and representatives of the SHI, the base-case incremental cost-effectiveness ratio (ICER) of sacubitril/valsartan versus ACE inhibitors is €23,401 per life-year gained (in 2018 Euros). At a price of zero, the cost-effectiveness ratio is already €9594 per life-year gained due to high background costs of heart failure. Annual budget impact and reduction of disease burden reach a maximum at 4–8 years after launch (€221 million and 2.9%, respectively, in the base case).
Conclusions
The ICER of sacubitril/valsartan is projected to be at or below the level of other accepted interventions for the treatment of asymptomatic to severe heart failure in Germany. Projected budget impact leads to an increase in SHI expenditures by < 0.04% per year.




Similar content being viewed by others
References
Ohlmeier C, Mikolajczyk R, Frick J, Prütz F, Haverkamp W, Garbe E. Incidence, prevalence and 1-year all-cause mortality of heart failure in Germany: a study based on electronic healthcare data of more than six million persons. Clin Res Cardiol. 2015;104(8):688–96.
Störk S, Handrock R, Jacob J, Walker J, Calado F, Lahoz R, Hupfer S, Klebs S. Epidemiology of heart failure in Germany: a retrospective database study. Clin Res Cardiol. 2017;106(11):913–22.
Pocock SJ, Ariti CA, McMurray JJ, Maggioni A, Køber L, Squire IB, Swedberg K, Dobson J, Poppe KK, Whalley GA, Doughty RN, Meta-Analysis Global Group in Chronic Heart Failure. Predicting survival in heart failure: a risk score based on 39 372 patients from 30 studies. Eur Heart J. 2013;34(19):1404–13.
Bundesamt Statistisches. Krankheitskostenrechnung. Wiesbaden: Statistisches Bundesamt; 2015.
McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR, Rouleau JL, Shi VC, Solomon SD, Swedberg K. Zile MR, PARADIGM-HF Investigators and Committees. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371(11):993–1004.
Packer M, McMurray JJ, Desai AS, Gong J, Lefkowitz MP, Rizkala AR, Rouleau JL, Shi VC, Solomon SD, Swedberg K, Zile M, Andersen K, Arango JL, Arnold JM, Bělohlávek J, Böhm M, Boytsov S, Burgess LJ, Cabrera W, Calvo C, Chen CH, Dukat A, Duarte YC, Erglis A, Fu M, Gomez E, Gonzàlez-Medina A, Hagège AA, Huang J, Katova T, Kiatchoosakun S, Kim KS, Kozan Ö, Llamas EB, Martinez F, Merkely B, Mendoza I, Mosterd A, Negrusz-Kawecka M, Peuhkurinen K, Ramires FJ, Refsgaard J, Rosenthal A, Senni M, Sibulo AS Jr, Silva-Cardoso J, Squire IB, Starling RC, Teerlink JR, Vanhaecke J, Vinereanu D. Wong RC, PARADIGM-HF Investigators and Coordinators. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation. 2015;131(1):54–61.
Lillyblad MP. Dual angiotensin receptor and neprilysin inhibition with sacubitril/valsartan in chronic systolic heart failure: understanding the New PARADIGM. Ann Pharmacother. 2015;49(11):1237–51.
Simpson J, Jhund PS, Silva Cardoso J, Martinez F, Mosterd A, Ramires F, Rizkala AR, Senni M, Squire I, Gong J, Lefkowitz MP, Shi VC, Desai AS, Rouleau JL, Swedberg K, Zile MR, McMurray JJ, Packer M. Solomon SD, PARADIGM-HF Investigators and Committees. Comparing LCZ696 with enalapril according to baseline risk using the MAGGIC and EMPHASIS-HF Risk Scores: an analysis of mortality and morbidity in PARADIGM-HF. J Am Coll Cardiol. 2015;66(19):2059–71.
Novartis Pharma GmbH. Sacubitril/Valsartan (Entresto®): Dossier zur Nutzenbewertung gemäß § 35a SGB V (Modul 4 A). https://www.g-ba.de/downloads/92-975-1312/2015-12-21_Modul4A_Sacubitril-Valsartan.pdf. Accessed 13 June 2018.
Garg R, Yusuf S. Overview of randomized trials of angiotensin-converting enzyme inhibitors on mortality and morbidity in patients with heart failure. Collaborative Group on ACE Inhibitor Trials. JAMA. 1995;273(18):1450–6.
Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen. General methods. Version 5.0. Köln: IQWiG. 2017.
Claggett B, Packer M, McMurray JJ, Swedberg K, Rouleau J, Zile MR, Jhund P, Lefkowitz M, Shi V, Solomon SD, PARADIGM-HF Investigators. Estimating the long-term treatment benefits of sacubitril-valsartan. N Engl J Med. 2015;373(23):2289–90.
Lauer-Fischer GmbH, Lauer-Taxe Arzneimittelpreise. https://www.lauer-fischer.de. Accessed 30 June 2018
Fischer KE, Stargardt T. The diffusion of generics after patent expiry in Germany. Eur J Health Econ. 2016;17(8):1027–40.
Kassenärztliche Bundesvereinigung. Einheitlicher Bewertungsmaßstab (EBM). Stand: 1. Quartal 2015. Berlin: Kassenärztliche Bundesvereinigung; 2018.
Institut für das Entgeltsystem im Krankenhaus. Fallpauschalen-Katalog 2015. Siegburg: InEK GmbH; 2018.
Management Consult Kestermann (2015). Gutachten zur ambulanten Notfallversorgung im Krankenhaus: Fallkostenkalkulation und Strukturanalyse. http://www.dkgev.de/media/file/19401.2015-02-17_Gutachten_zur_ambulanten_Notfallversorgung_im_Krankenhaus_2015.pdf. Accessed 29 May 2018.
Bundesamt Statistisches. Krankheitskostenrechnung. Bonn: Statistisches Bundesamt; 2010.
Trueman D, Kapetanakis V, Briggs A, Lewis E, Rouleau J, Solomon SD, Swedberg K, Zile MR, Packer M, McMurray JJV, Croft DC, Haroun R, Gielen V. Better health-related quality of life in patients treated with sacubitril/valsartan compared with enalapril, irrespective of NYHA class: Analysis of EQ-5D in PARADIGM-HF. Eur Heart J. 2017;38(Suppl 1):P3373.
Novartis Pharma GmbH. Sacubitril/Valsartan (Entresto®): Dossier zur Nutzenbewertung gemäß § 35a SGB V (Modul 3 A). https://www.g-ba.de/downloads/92-975-1311/2015-12-21_Modul3A_Sacubitril-Valsartan.pdf. Accessed 29 May 2018.
Jhund PS, Fu M, Bayram E, Chen CH, Negrusz-Kawecka M, Rosenthal A, Desai AS, Lefkowitz MP, Rizkala AR, Rouleau JL, Shi VC, Solomon SD, Swedberg K, Zile MR, McMurray JJ, Packer M, PARADIGM-HF Investigators and Committees. Efficacy and safety of LCZ696 (sacubitril-valsartan) according to age: insights from PARADIGM-HF. Eur Heart J. 2015;36(38):2576–84.
EuroQol Group. EuroQol–a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199–208.
Barendregt JJ. The half-cycle correction: banish rather than explain it. Med Decis Mak. 2009;29(4):500–2.
Bundesamt Statistisches. Allgemeine Sterbetafel 2010/12. Wiesbaden: Statistisches Bundesamt; 2015.
Kuntz K, Weinstein M. Modelling in economic evaluation. In: Drummond M, McGuire A, editors. Economic evaluation in health care: merging theory with practice. New York: Oxford University Press; 2001.
Bundesamt Statistisches. Bevölkerung: Deutschland, Stichtag, Altersjahre. Wiesbaden: Statistisches Bundesamt; 2015.
Statistisches Bundesamt. Bevölkerung Deutschlands bis 2060 – 13. koordinierte Bevölkerungsvorausberechnung. Wiesbaden: Statistisches Bundesamt; 2010.
Gampe J. Human mortality beyond age 110. In Supercentenarians. Berlin: Springer; 2010. p. 168–230.
Owan TE, Redfield MM. Epidemiology of diastolic heart failure. Prog Cardiovasc Dis. 2005;47(5):320–32.
Lewis EF, Lamas GA, O’Meara E, Granger CB, Dunlap ME, McKelvie RS, Probstfield JL, Young JB, Michelson EL, Halling K, Carlsson J, Olofsson B, McMurray JJ, Yusuf S, Swedberg K, Pfeffer MA, CHARM Investigators. Characterization of health-related quality of life in heart failure patients with preserved versus low ejection fraction in CHARM. Eur J Heart Fail. 2007;9(1):83–91.
Tiller D, Russ M, Greiser KH, Nuding S, Ebelt H, Kluttig A, Kors JA, Thiery J, Bruegel M, Haerting J, Werdan K. Prevalence of symptomatic heart failure with reduced and with normal ejection fraction in an elderly general population-the CARLA study. PLoS One. 2013;8(3):e59225.
Wachter R. Diastolic heart failure and multimorbidity. Dtsch Med Wochenschr. 2015;140(6):402–5.
Cassel D, Ulrich V. AMNOG-Check 2017: Gesundheitsökonomische Analysen der Versorgung mit Arzneimittel-Innovationen. Baden-Baden: Nomos Verlagsgesellschaft; 2017. p. 168.
Cavender MA, Norhammar A, Birkeland KI, Jørgensen ME, Wilding JP, Khunti K, Fu AZ, Bodegård J. Blak BT, Wittbrodt E, Thuresson M, Fenici P, Hammar N, Kosiborod M, CVD-REAL Investigators and StudyGroup. SGLT-2 Inhibitors and Cardiovascular Risk: An Analysis of CVD-REAL. J Am Coll Cardiol. 2018;71(22):2497–506.
Sullivan SD, Mauskopf JA, Augustovski F, Jaime Caro J, Lee KM, Minchin M, Orlewska E, Penna P, Rodriguez Barrios JM, Shau WY. Budget impact analysis-principles of good practice: report of the ISPOR 2012 Budget Impact Analysis Good Practice II Task Force. Value Health. 2014;17(1):5–14.
Aidelsburger P, Grabein K, Klauss V, Wasem J. Cost-effectiveness of cardiac resynchronization therapy in combination with an implantable cardioverter defibrillator (CRT-D) for the treatment of chronic heart failure from a German health care system perspective. Clin Res Cardiol. 2008;97(2):89–97.
Gandjour A, Holler A, Adarkwah CC. Cost-effectiveness of implantable defibrillators after myocardial infarction based on 8-year follow-up data (MADIT II). Value Health. 2011;14(6):812–7.
Sandhu AT, Ollendorf DA, Chapman RH, Pearson SD, Heidenreich PA. Cost-effectiveness of sacubitril-valsartan in patients with heart failure with reduced ejection fraction. Ann Intern Med. 2016 Nov 15;165(10):681–9.
Gaziano TA, Fonarow GC, Claggett B, Chan WW, Deschaseaux-Voinet C, Turner SJ, Rouleau JL, Zile MR, McMurray JJ, Solomon SD. Cost-effectiveness analysis of sacubitril/valsartan vs enalapril in patients with heart failure and reduced ejection fraction. JAMA Cardiol. 2016;1(6):666–72.
King JB, Shah RU, Bress AP, Nelson RE, Bellows BK. Cost-effectiveness of sacubitril-valsartan combination therapy compared with enalapril for the treatment of heart failure with reduced ejection fraction. JACC Heart Fail. 2016;4(5):392–402.
van der Pol S, Degener F, Postma MJ, Vemer P. An economic evaluation of sacubitril/valsartan for heart failure patients in the Netherlands. Value Health. 2017;20(3):388–96.
Corro Ramos I, Versteegh MM, de Boer RA, Koenders JM, Linssen GC, Meeder JG, Rutten-van Mölken MP. Cost-effectiveness of the angiotensin receptor neprilysin inhibitor sacubitril/valsartan for patients with chronic heart failure and reduced ejection fraction in the Netherlands: a country adaptation analysis under the former and current Dutch Pharmacoeconomic Guidelines. Value Health. 2017;20(10):1260–9.
National Institute for Health and Care Excellence. Final appraisal determination: sacubitril valsartan for treating symptomatic chronic heart failure with reduced ejection fraction. Issue date: March 2016.
Neumann PJ, Sanders GD, Russell LB, Siegel JE, Ganiats TG, editors. Cost-effectiveness in health and medicine. 2nd ed. New York: Oxford University Press; 2016.
Grimm SE, Dixon S, Stevens JW. When future change matters: modeling future price and diffusion in health technology assessments of medical devices. Value Health. 2016;19(6):720–6.
Heidenreich PA, Albert NM, Allen LA, Bluemke DA, Butler J, Fonarow GC, Ikonomidis JS, Khavjou O, Konstam MA, Maddox TM, Nichol G, Pham M, Piña IL, Trogdon JG, American Heart Association Advocacy Coordinating Committee; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Radiology and Intervention; Council on Clinical Cardiology; Council on Epidemiology and Prevention; Stroke Council. Forecasting the impact of heart failure in the United States: a policy statement from the American Heart Association. Circ Heart Fail. 2013;6(3):606–19.
Author information
Authors and Affiliations
Contributions
AG was involved in model conceptualization, building, and validation, and wrote the first draft of the manuscript. DO was involved in model conceptualization and commented on the draft version of the manuscript.
Corresponding author
Ethics declarations
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Funding
This study was funded by Novartis Deutschland GmbH.
Appendix 1
Appendix 1
Calculation of the probability of death under enalapril at age 65 years under real-world conditions
where \(p_{\text{trial}}\) is the probability of death under enalapril at age 65 years in the PARADIGM-HF trial, \(i\) is the New York Heart Association (NYHA) class, \({\text{RR}}\) is the ‘rate ratio’ of the MAGGIC risk score (NYHA class II was set to 1.0) [3], and \(f\) is the fraction of patients.
Rights and permissions
About this article
Cite this article
Gandjour, A., Ostwald, D.A. Sacubitril/Valsartan (LCZ696): A Novel Treatment for Heart Failure and its Estimated Cost Effectiveness, Budget Impact, and Disease Burden Reduction in Germany. PharmacoEconomics 36, 1285–1296 (2018). https://doi.org/10.1007/s40273-018-0688-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-018-0688-4