Abstract
Background
Eslicarbazepine acetate (ESL), a novel sodium channel blocker, is approved for mono and adjunctive treatment of partial epileptic seizures with or without secondary generalization. Its efficacy in primary generalized seizures has not yet been evaluated.
Objective
To evaluate the efficacy and safety of ESL in primary generalized tonic-clonic seizures (PGTCS) in an observational study.
Methods
The data were collected from a prospective population-based register. Effectiveness was measured as relative reduction in standardized seizure frequency (SSF), responder rate (≥ 50% reduction in SSF), and seizure freedom rate at 6 and 12 months after initiation of ESL. Safety and tolerability were evaluated using patients’ diaries.
Results
Fifty-six adult patients with PGTCS were treated with ESL as adjunctive therapy. Of these, 30.4% (n = 17) had myoclonic seizures in addition to PGTCS. The retention rate after 12 months was 80.4% (n = 45). After initiating ESL therapy, reduction in SSF for PGTCS on ESL was 56.0% after 6 months and 56.9% after 12 months (p < 0.01), whereas myoclonic seizures did not show any significant improvement in frequency. The responder rate for PGTCS was 64.3% after 6 months and 66.1% after 12 months, and seizure freedom was achieved in 32.1% and 35.7%, respectively. Forty-three patients (73.2%) reported no side effects. Among the reported side effects of ESL therapy, headache (7.1%), dizziness (8.9%), tiredness (7.1%), nausea (5.4%), and hyponatremia (5.4%) were the most prevalent.
Conclusions
Our data suggest that ESL may provide additional benefits in the treatment of patients with PGTCS and motivate randomized controlled trials in this indication.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Eslicarbazepine acetate may be beneficial in the adjunctive therapy of primary generalized tonic-clonic seizures. |
Slow inactivation of voltage-gated sodium channels could provide a new therapeutic option in primary generalized tonic-clonic seizures. |
The results we obtained in this observational study should be confirmed in randomized controlled trials. |
1 Introduction
Epilepsy can be divided into focal and generalized epilepsy syndromes [1]. Idiopathic generalized epilepsies encompass childhood absence epilepsy, juvenile absence epilepsy, juvenile myoclonic epilepsy, and epilepsy only with generalized tonic-clonic seizures [1]. The task force of the International League Against Epilepsy (ILAE) on Classification of the Epilepsies recommended two equal terms for these four epilepsy syndromes: “genetic generalized epilepsies” and “idiopathic generalized epilepsy” [1]. Here, we use the term idiopathic generalized epilepsy (IGE). In IGE, absences, myoclonic, or primary generalized tonic-clonic seizures may occur in combination or separately [2]. These types of seizure are accompanied by generalized, bilateral synchronous (poly-)spike wave discharges on the electroencephalogram [3].
IGE typically occurs in childhood and adolescence, may subside in adulthood, and often responds better to medical treatment than focal epilepsy [4]. Nevertheless, it is not uncommon for pharmacoresistant generalized epilepsy to develop in adulthood in 15–40% of cases [5, 6].
Unlike focal epilepsies, the choice of medications and surgical procedures for primary generalized epilepsies is very limited. Of all the antiseizure medications (ASMs) that have been shown to be effective in treating patients with focal epilepsy, only phenobarbital, carbamazepine (CBZ), phenytoin (PHT), valproate (VPA), lamotrigine (LTG), levetiracetam (LEV), topiramate (TPM), and perampanel (PER) have been approved in Europe for treating patients with primary generalized tonic-clonic seizures (PGTCS) [7,8,9,10,11]. Certain ASMs, especially sodium channel blockers, may aggravate myoclonic seizures and absence seizures up to a status epilepticus [12,13,14,15,16].
Successful treatment of refractory IGE requires the use of anticonvulsants with new mechanisms of action. Achieving seizure freedom with as few side effects as possible is particularly important for affected patients [17], reflected by improved quality of life [18, 19]. Therefore, data on the efficacy and safety of novel ASMs in patients with refractory IGE is urgently needed. New ASMs are often first investigated in partial seizures with or without secondary generalization because they are more prevalent in adult populations [20]. Unfortunately, not all pharmaceutical companies proceed to evaluate novel ASMs in primary generalized seizures after their approval for partial seizures [21].
Eslicarbazepine acetate (ESL) is approved for mono and adjunctive treatment of partial epileptic seizures with or without secondary generalization [22]. Its approval was issued in the EU in 2009 and in the USA in 2013, and it has been generic since 2021. ESL is a carboxamide and dibenzazepine derivative closely structurally related to CBZ and oxcarbazepine (OXC) [23].
CBZ has already been shown to be effective on PGTCS [24]. ESL has a different mechanism of action to CBZ, affecting the slow rather than the fast inactivation of sodium channels [25]. Interestingly, lacosamide (LCS) shares the same mechanism of action and has been already been proved in the treatment of PGTCS [10].
In placebo-controlled trials, adverse effects occurred in 60–70% of subjects treated with ESL (compared with 46–49% of subjects treated with placebo) [26]. Adverse effects were mild to moderate and occurred mainly in the first few weeks. The most common side effects are dizziness, fatigue, headache, and nausea [26]. Hyponatremia as a side effect of ESL therapy should also be considered and monitored.
The aim of this observational study was to evaluate the effectiveness and safety/tolerability of ESL as adjunctive therapy in patients with PGTCS.
2 Methods
2.1 Study Design and Clinical Evaluation
Data collection on adult patients with PGTCS treated with ESL as adjunctive therapy was performed using the Mainz Epilepsy Registry (MAINZ-EPIREG). MAINZ-EPIREG is a population-based register of patients with epilepsy who are treated in the Mainz Comprehensive Epilepsy and Sleep Medicine Center (reference area of 4 million inhabitants). All clinical diagnoses of PGTCS were confirmed by EEG records and all patients in our study showed EEG findings consistent with generalized epilepsy. Patients with absence epilepsies were not included, as none of them were treated with ESL in our center.
Effectiveness was measured as relative reduction in standardized seizure frequency (SSF, based on the number of seizures per month), responder rate (≥ 50% reduction in SSF), and seizure freedom rate at 6 and 12 months after initiation of ESL. The monthly SSF at baseline was calculated considering 6 months preceding the initiation of ESL. Seizure frequency was recorded in a systematic way before the initiation of ESL and during the whole study period by means of standardized patients’ diaries. Safety and tolerability data were documented as side effects reported by patients during the treatment and documented in their seizure diaries. These data were also documented in a systematic way.
All patients underwent blood examinations and sodium plasma levels were documented during the entire duration of the observation. The plasma concentration of ESL and (R)-licarbazepine was obtained in all patients while they were on the stable dosing. The approximate time of plasma concentration quantification was 10–12 h after intake of ESL. Our study was approved by the local ethics committee and all patients signed informed consent for participation in this study.
2.2 Statistics
The statistical analysis was performed using IBM SPSS Statistics Version 23.0 (IBM Corp., Armonk, NY, USA). The data are presented as mean and standard deviation (SD) or median and range depending on normality of data. A t test was used for comparisons of normally distributed variables. If data were not normally distributed, the Mann-Whitney U test (two independent groups), the Kruskal-Wallis test (more than two independent groups), or the Wilcoxon rank test (two dependent groups) was applied. Statistical significance was assumed at a P value of < 0.05. In case of multiple comparisons, we used the Bonferroni correction.
3 Results
In total, 56 patients with PGTCS were treated with ESL as adjunctive therapy during the study period, of whom 33 (58.9%) were female. Patients with PGTCS alone made up the majority of our sample (69.7%). The remaining patients suffered from juvenile myoclonic epilepsy with both PGTCS and myoclonic seizures. The mean age of patients was 34.5 (± 9.0 years). The most commonly used dose of ESL was 800 mg/day (400–1600 mg/day). The mean plasma levels of eslicarbazepine and (R)–licarbazepine were 15.6 ± 3.0 µg/mL and 1.0 ± 0.2 µg/mL, correspondingly.
Data on demographics, clinical parameters of epilepsy, and ASMs are presented in Table 1. The mean duration of epilepsy before starting therapy with ESL was 12.4 (± 7.9) years. The mean seizure frequency before starting ESL therapy, the number of ASMs discontinued in the past, and the numbers of adjunctive ASMs are shown in Table 1. The most frequently administered other ASMs were LEV, LTG, and VPA (Table 1). The retention rate after 12 months of ESL therapy was 80.4% (n = 45).
Considering all seizures, the mean SSF was 2.5 (± 4.8) after 6 months and 3.4 (± 5.7) after 12 months, both significantly lower compared to the SSF before starting ESL (p < 0.01 each). Compared to baseline, the mean SSF of PGTCS was also significantly lower at 0.8 (± 1) after 6 months and 0.8 (± 1.1) after 12 months (p < 0.01 each). The mean SSF of myoclonic seizures did not differ compared to baseline (9.5 (± 7.4) at 6 months, p = 0.27; 8.8 (± 6.5) at 12 months, p = 0.18).
Compared to seizure frequency at the time of initiation of ESL, the relative reduction in SSF for PGTCS was 56.0% after 6 months and 56.9% after 12 months (p < 0.01). The response rate in PGTCS on ESL was 64.3% after 6 months and 66.1% after 12 months, and seizure freedom was achieved in 32.1% and 35.7%, respectively (Fig. 1). Myoclonic seizures did not show a good response to ESL, with a relative reduction in SSF of 13.2% and 12.9% at the above-mentioned respective time points. Only 3.5% of patients with myoclonic seizures became free from this type of seizure after 12 months of ESL treatment.
Freedom of seizures, responder rate, and seizure reduction 6 and 12 months after initiation of eslicarbazepine acetate. *Statistically significant difference of p < 0.05 in group comparison between all seizures and PGTCS with myoclonic seizures at 6 and 12 months, respectively. PGTCS primary generalized tonic-clonic seizure, SSF standardized seizure frequency
The combination of ESL with LTG or VPA showed the responder rate of PGTCS of 73.9% and 72.2% at 12-month follow-ups, respectively (Fig. 2). The responder rate in case of the combination of ESL with LEV was 51.1%. TPM, CLB, and PER were used as second or third adjunctive ASMs. Because of the lower number of patients treated with adjunctive TPM, CLB, and PER, statistical analysis was not performed for these subgroups.
Responder rate in primary generalized tonic-clonic seizures by adjunctive medication 6 and 12 months after initiation of eslicarbazepine acetate. **Statistically significant difference at p < 0.01 in the responder rate of PGTCS in comparison with LEV. 1Responder defined as a patient with a reduction in seizure frequency of ≥ 50% compared to baseline. LEV levetiracetam, LTG lamotrigine, VPA valproate
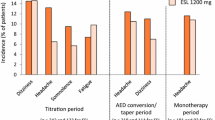
Side effects during ESL therapy are shown in Table 2. Hyponatremia up to a minimum of 122 mmol/L was observed in three patients (5.4%). In total 43 patients (73.2%) reported no side effects (Table 2). Thirteen of 19 reported adverse events (68.4%) occurred in patients using a co-medication with LTG. Among patients with juvenile myoclonic epilepsy, 23.5% (n = 4) had an increase in myoclonic seizures by up to 50% compared to the time before initiation of ESL. Most frequently, side effects were observed in the combination of ESL with LTG.
4 Discussion
The data presented here demonstrate that ESL may be a beneficial treatment for patients with PGTCS. This type of seizure showed a better response to ESL compared to myoclonic seizures. A sub-group of patients even suffered an increase of the frequency of myoclonic seizures.
ESL is a third level derivative of CBZ. CBZ and OXC have already shown a positive effect in the treatment of PGTCS and patients with IGE in previous studies [6, 24, 27]. A recently published study by Cerulli Irelli et al. [6] retrospectively investigated the use of selective sodium channel blockers in 56 patients with IGE. The majority of patients received CBZ and OXC, and only one patient received ESL. For all selective sodium channel blockers combined, that study showed similar responder rates for all seizures (53.5%) at 12 months as in the present paper. In addition, a better responder rate (67.9%) was also observed when only PGTCSs were considered. Similar to our data, a few patients (8.9%) reported worsening of seizures and these were patients with juvenile myoclonus epilepsy. To our knowledge, to date, there is no study that has explicitly investigated ESL in patients with PGTCS. However, the similarity of the mechanisms of action of CBZ, OXC, and ESL suggest a possible overlap in therapeutic effect.
The treatment of IGE can be challenging, and 15–40% of cases are refractory to treatment [5, 6]. The side effects of approved ASM are one of the major reasons for their discontinuation [28]. Especially in patients who do not respond to or do not tolerate standard therapy, a new opportunity appears with each newly introduced ASM [28].
In focal epilepsies, extensive studies have been conducted comparing ESL with other commonly used therapeutic agents [29, 30]. ESL is a well-tolerated ASM that has a slightly different mechanism of action compared to its predecessors, CBZ and OXC. ESL enhances the slow inactivation of voltage-dependent sodium channels [31] similar to LCS [32, 33].
LCS has been shown in previous studies to have good efficacy in patients with generalized epilepsy [10, 11]. A worsening of epileptic seizures described for other sodium channel blockers has also not been shown for LCS [11]. In the present study, worsening was observed only in four patients with myoclonic seizures (23.5%) and in one patient with PGTCS (1.8%). The enhancement of slow inactivation of voltage-gated sodium channels, which is a common mechanism of action in both LCS and ESL, is obviously effective and safe in the treatment of PGTCS. Interestingly, this mechanism of action also shows advantages in the treatment of some definite types of focal epilepsy, such as post-stroke epilepsy [34].
Another interesting finding of this study was the significantly better effectiveness of ESL in combination with LTG or VPA than in combination with other ASMs. LTG has shown good efficacy in generalized epilepsy, partly acting through mechanisms of action other than sodium channel blockade [35]. Nevertheless, this combination with ESL and LTG in the present study showed not only a better responder rate but also more side effects overall. In 13 of the 19 reported cases (68.4%) with side effects, patients used LTG as adjunctive medication. However, specific side effects such as dizziness did not occur more frequently with additional use of LTG. Overall, the rate of adverse events of 26.8% was rather low in our study.
Our study has several limitations. This study was an observational study and its findings are inferior to the result of randomized controlled studies. The investigators were not blinded to the therapy regime. The data are based on a moderate sample size, limiting the statistical power for the analysis of sub-groups and different dosages of ESL. However, it is challenging to recruit patients with generalized epilepsy who are taking off-label therapy with a newer ASM. In other studies investigating new ASMs, the number of patients was often similar or even smaller [6, 11]. We did not perform a precise analysis of pharmacokinetics according to the hours since the intake of ESL as this was beyond the scope of our clinical study. As this was not a placebo-controlled study, a possible placebo effect in some patients could not be calculated. The Hawthorne effect was taken into account by the fact that the patient care during the study was not different from the routine patient care in our center outside of a study setting. The intrinsic variability in seizure frequency, which can potentially increase and decrease the outcome of the study, was addressed and attenuated by the long-term observaton of 12 months.
5 Conclusions
Based on these data, ESL can be considered potentially beneficial in the adjunctive therapy of PGTCS. The mechanism of action of slow inactivation of voltage-gated sodium channels could provide a new therapeutic option in this type of seizure. Concomitant myoclonic seizures do not show a good response to ESL and have a risk of deterioration. Controlled randomized studies are needed to determine whether the results from this observational study can be reproduced at a higher level of evidence.
References
Scheffer IE, Berkovic S, Capovilla G, Connolly MB, French J, Guilhoto L, et al. ILAE classification of the epilepsies: position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017;58(4):512–21.
Beydoun A, D’Souza J. Treatment of idiopathic generalized epilepsy—a review of the evidence. Expert Opin Pharmacother. 2012;13(9):1283–98.
Benbadis SR. Practical management issues for idiopathic generalized epilepsies. Epilepsia. 2005;46(Suppl 9):125–32.
Camfield C, Camfield P. Management guidelines for children with idiopathic generalized epilepsy. Epilepsia. 2005;46(Suppl 9):112–6.
Cerulli Irelli E, Morano A, Barone FA, Fisco G, Fanella M, Orlando B, et al. Persistent treatment resistance in genetic generalized epilepsy: a long-term outcome study in a tertiary epilepsy center. Epilepsia. 2020;61(11):2452–60.
Cerulli Irelli E, Morano A, Fanella M, Orlando B, Salamone EM, Giallonardo AT, et al. Reconsidering the role of selective sodium channel blockers in genetic generalized epilepsy. Acta Neurol Scand. 2021;144(6):647–54.
French JA, Krauss GL, Wechsler RT, Wang XF, DiVentura B, Brandt C, et al. Perampanel for tonic-clonic seizures in idiopathic generalized epilepsy. A randomized trial. Neurology. 2015;85(11):950–7.
Kanner AM, Ashman E, Gloss D, Harden C, Bourgeois B, Bautista JF, et al. Practice guideline update summary: efficacy and tolerability of the new antiepileptic drugs II: treatment-resistant epilepsy: report of the guideline development, dissemination, and implementation Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2018;91(2):82–90.
Kanner AM, Ashman E, Gloss D, Harden C, Bourgeois B, Bautista JF, et al. Practice guideline update summary: Efficacy and tolerability of the new antiepileptic drugs I: Treatment of new-onset epilepsy: report of the guideline development, dissemination, and implementation subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2018;91(2):74–81.
Vossler DG, Knake S, O’Brien TJ, Watanabe M, Brock M, Steiniger-Brach B, et al. Efficacy and safety of adjunctive lacosamide in the treatment of primary generalised tonic-clonic seizures: a double-blind, randomised, placebo-controlled trial. J Neurol Neurosurg Psychiatry. 2020;91(10):1067–75.
Wechsler RT, Yates SL, Messenheimer J, Leroy R, Beller C, Doty P. Lacosamide for uncontrolled primary generalized tonic-clonic seizures: An open-label pilot study with 59-week extension. Epilepsy Res. 2017;130:13–20.
Chaves J, Sander JW. Seizure aggravation in idiopathic generalized epilepsies. Epilepsia. 2005;46(Suppl 9):133–9.
Fanella M, Egeo G, Fattouch J, Casciato S, Lapenta L, Morano A, et al. Oxcarbazepine-induced myoclonic status epilepticus in juvenile myoclonic epilepsy. Epileptic Disord. 2013;15(2):181–7.
Perucca E, Gram L, Avanzini G, Dulac O. Antiepileptic drugs as a cause of worsening seizures. Epilepsia. 1998;39(1):5–17.
Somerville ER. Some treatments cause seizure aggravation in idiopathic epilepsies (especially absence epilepsy). Epilepsia. 2009;50(Suppl 8):31–6.
Thomas P, Valton L, Genton P. Absence and myoclonic status epilepticus precipitated by antiepileptic drugs in idiopathic generalized epilepsy. Brain. 2006;129(Pt 5):1281–92.
Rosenow F, Winter Y, Leunikava I, Brunnert M, Joeres L, Sutphin J, et al. Relative importance of clinical outcomes and safety risks of antiseizure medication monotherapy for patients and physicians: Discrete choice experiment eliciting preferences in real-world study “VOTE.” Epilepsia. 2022;63(2):451–62.
Varesio C, Provenzi L, Donetti Dontin S, Zanaboni MP, Pasca L, Veggiotti P, et al. Pathways to quality of life in adolescents with genetic generalized epilepsy: the role of seizure features and affective symptoms. Epilepsy Behav. 2020;109: 107115.
Winter Y, Daneshkhah N, Galland N, Kotulla I, Krüger A, Groppa S. Health-related quality of life in patients with poststroke epilepsy. Epilepsy Behav. 2018;80:303–6.
Brown C. Pharmacological management of epilepsy. Prog Neurol Psychiatry. 2016;20(2):27–34c.
Chen Z, Brodie MJ, Kwan P. What has been the impact of new drug treatments on epilepsy? Curr Opin Neurol. 2020;33(2):185–90.
Verrotti A, Loiacono G, Rossi A, Zaccara G. Eslicarbazepine acetate: an update on efficacy and safety in epilepsy. Epilepsy Res. 2014;108(1):1–10.
Almeida L, Soares-da-Silva P. Eslicarbazepine acetate (BIA 2–093). Neurotherapeutics. 2007;4(1):88–96.
Tudur Smith C, Marson AG, Williamson PR. Carbamazepine versus phenobarbitone monotherapy for epilepsy. Cochrane Database Syst Rev. 2003(1):Cd001904.
Hebeisen S, Pires N, Loureiro AI, Bonifácio MJ, Palma N, Whyment A, et al. Eslicarbazepine and the enhancement of slow inactivation of voltage-gated sodium channels: a comparison with carbamazepine, oxcarbazepine and lacosamide. Neuropharmacology. 2015;89:122–35.
Gil-Nagel A, Elger C, Ben-Menachem E, Halász P, Lopes-Lima J, Gabbai AA, et al. Efficacy and safety of eslicarbazepine acetate as add-on treatment in patients with focal-onset seizures: integrated analysis of pooled data from double-blind phase III clinical studies. Epilepsia. 2013;54(1):98–107.
Kenyon K, Mintzer S, Nei M. Carbamazepine treatment of generalized tonic-clonic seizures in idiopathic generalized epilepsy. Seizure. 2014;23(3):234–6.
Hao X, Goldberg D, Kelly K, Stephen L, Kwan P, Brodie MJ. Uncontrolled epilepsy is not necessarily the same as drug-resistant epilepsy: differences between populations with newly diagnosed epilepsy and chronic epilepsy. Epilepsy Behav. 2013;29(1):4–6.
Keating GM. Eslicarbazepine acetate: a review of its use as adjunctive therapy in refractory partial-onset seizures. CNS Drugs. 2014;28(7):583–600.
Weissinger F, Losch F, Winter Y, Brecht S, Lendemans D, Kockelmann E. Effectiveness of eslicarbazepine acetate in dependency of baseline anticonvulsant therapy: Results from a German prospective multicenter clinical practice study. Epilepsy Behav. 2019;101(Pt A): 106574.
Holtkamp D, Opitz T, Hebeisen S, Soares-da-Silva P, Beck H. Effects of eslicarbazepine on slow inactivation processes of sodium channels in dentate gyrus granule cells. Epilepsia. 2018;59(8):1492–506.
Beydoun A, DuPont S, Zhou D, Matta M, Nagire V, Lagae L. Current role of carbamazepine and oxcarbazepine in the management of epilepsy. Seizure. 2020;83:251–63.
Beyreuther BK, Freitag J, Heers C, Krebsfänger N, Scharfenecker U, Stöhr T. Lacosamide: a review of preclinical properties. CNS Drug Rev. 2007;13(1):21–42.
Winter Y, Uphaus T, Sandner K, Klimpe S, Stuckrad-Barre SV, Groppa S. Efficacy and safety of antiseizure medication in post-stroke epilepsy. Seizure. 2022;100:109–14.
Mitra-Ghosh T, Callisto SP, Lamba JK, Remmel RP, Birnbaum AK, Barbarino JM, et al. PharmGKB summary: lamotrigine pathway, pharmacokinetics and pharmacodynamics. Pharmacogenet Genomics. 2020;30(4):81–90.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Open Access funding enabled and organized by Projekt DEAL. No finding was provided for this study.
Conflict of interests/competing interests
YW reports honoraria for educational presentations and consultations from Arvelle Therapeutics, Bayer AG, BIAL, Eisai, LivaNova, Novartis, and UCB Pharma. SG received compensation for professional services from Abbott, AbbVie, Bial, Medtronic, UCB, and Zambon; research grants from Abbott, Boston Scientific, MagVenture, German Research Council, Innovations Fund and German Ministry of Education and Health. NM has received honoraria for lecturing and travel expenses for attending meetings from Biogen Idec, GlaxoSmith Kline, Teva, Novartis Pharma, Bayer Healthcare, Genzyme, Alexion Pharamceuticals, Fresenius Medical Care, Diamed, UCB Pharma, and BIAL, has received royalties for consulting from UCB Pharma, and Alexion Pharmaceuticals, and has received financial research support from Euroimmun, Fresenius Medical Care, Diamed, Alexion Pharmaceuticals, and Novartis Pharma. SK has received honoraria for presentations and teaching courses from UCB Pharma and Novartis Pharma, and has been invited to congresses and educational meetings by UCB Pharma and Eisai Pharma. SGM received honoraria for lecturing and travel expenses for attending meetings from Almirall, Amicus Therapeutics Germany, Bayer Health Care, Biogen, Celgene, Diamed, Genzyme, MedDay Pharmaceuticals, Merck Serono, Novartis, Novo Nordisk, ONO Pharma, Roche, Sanofi-Aventis, Chugai Pharma, QuintilesIMS, and Teva. His research is funded by the German Ministry for Education and Research (BMBF), Deutsche Forschungsgemeinschaft (DFG), Else Kröner Fresenius Foundation, German Academic Exchange Service, Hertie Foundation, Interdisciplinary Center for Clinical Studies (IZKF) Muenster, German Foundation Neurology, and by Almirall, Amicus Therapeutics Germany, Biogen, Diamed, Fresenius Medical Care, Genzyme, Merck Serono, Novartis, ONO Pharma, Roche, and Teva. KS and TV declare no conflicts of interest.
Ethics approval
This study was approved by the ethics committee of the Rhineland-Palatinate Chamber of Physicians.
Consent to participate
All patients signed informed consent for participation in this study.
Consent for publication
Not applicable.
Availability of data and material
Data can be received on request from the corresponding author.
Code availability
Not applicable.
Authors’ Contributions
Conceptualization and design: YW, SG. Methodology and data collection: YW, KS, SG. Analysis and interpretation: YW, KS, TV, SG. Writing, original draft preparation: YW, KS, TV, NM, SK, SGM, SG. Writing, reviewing, and editing: YW, KS, TV, NM, SK, SGM, SG. All authors have read and approved the final submitted manuscript and agree to be accountable for the work.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Winter, Y., Sandner, K., Vieth, T.L. et al. Eslicarbazepine Acetate as Adjunctive Therapy for Primary Generalized Tonic-Clonic Seizures in Adults: A Prospective Observational Study. CNS Drugs 36, 1113–1119 (2022). https://doi.org/10.1007/s40263-022-00954-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-022-00954-w