Abstract
Background and Objective
Registrational trials for ciltacabtagene autoleucel [cilta-cel]) and idecabtagene vicleucel [ide-cel] chimeric antigen receptor T-cell (CAR-T) therapies were single-arm studies conducted with relapse refractory multiple myeloma (MM) patients who were triple-class-exposed (TCE) or triple-class-refractory (TCR). It is critical for researchers conducting comparative effectiveness research (CER) to carefully consider the most appropriate data sources and comparable patient populations. The aim of this study was to identify potential data sources and populations for comparing to single-arm CAR-T trials CARTITUDE-1 (cilta-cel) and KarMMa (ide-cel).
Methods
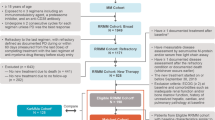
A 2-part global systematic literature search produced a review of (1) clinical trials of National Comprehensive Cancer Network (NCCN) guideline preferred regimens in previously treated MM, and (2) real-world data cohorts of TCE or TCR populations, published between 1/1/2015 and 12/10/2020, with sample sizes of > 50 patients and reporting survival-related outcomes. Implications on CER and accepted best practices are discussed.
Results
Nine clinical trials of NCCN preferred regimens were identified along with five real-world data-based publications. No clinical trials evaluated patients with TCE or TCR MM. Among the real-world data-based publications, two evaluated patients exclusively with TCR MM, two analyzed a mixed population of patients with TCE or TCR MM, and one publication assessed patients exclusively with TCE MM. Real-world data treatment patterns were heterogeneous.
Conclusion
Current NCCN preferred regimens were not specifically studied in TCE or TCR MM patients, although some studies do include a proportion of these types of patients. Therefore, appropriate matching of populations using either real-world data or patient level clinical trial data is critical to putting trials of novel CAR-Ts (i.e., CARTITUDE-1 or KarMMa) into appropriate comparative context.

Similar content being viewed by others
References
Food & Drug Administration. YESCARTA prescribing information. 2020. https://www.fda.gov/media/108377/download. Accessed 18 Dec 2020.
Food & Drug Administration. TECARTUS prescribing information. 2020. https://www.fda.gov/media/140409/download. Accessed 18 Dec 2020.
Food & Drug Administration. KYMRIAH prescribing information. 2018.https://www.fda.gov/files/vaccines%2C%20blood%20%26%20biologics/published/Package-Insert---KYMRIAH.pdf. Accessed 18 Dec 2020.
Janssen Research & Development, LLC. A study of JNJ-68284528, a chimeric antigen receptor T cell (CAR-T) therapy directed against B-cell maturation antigen (BCMA) in participants with relapsed or refractory multiple myeloma (CARTITUDE-1). 2020. https://clinicaltrials.gov/ct2/show/NCT03548207. Accessed 17 Dec 2020.
Celgene. Efficacy and safety study of bb2121 in subjects with relapsed and refractory multiple myeloma (KarMMa). 2020. https://clinicaltrials.gov/ct2/show/NCT03361748. Accessed 17 Dec 2020.
Madduri D, Berdeja JG, Usmani SZ, et al. CARTITUDE-1: phase 1b/2 study of ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T cell therapy, in relapsed/refractory multiple myeloma. Blood. 2020;136(Supplement 1):22–5. https://doi.org/10.1182/blood-2020-136307.
Lin Y, Raje NS, Berdeja JG, et al. Idecabtagene Vicleucel (ide-cel, bb2121), a BCMA-directed CAR T cell therapy, in patients with relapsed and refractory multiple myeloma: updated results from phase 1 CRB-401 study. Blood. 2020;136(Supplement 1):26–7. https://doi.org/10.1182/blood-2020-134324.
Munshi NC, Anderson LD Jr, Shah N, et al. Idecabtagene vicleucel (ide-cel; bb2121), a BCMA-targeted CAR T-cell therapy, in patients with relapsed and refractory multiple myeloma (RRMM): initial KarMMa results. J Clin Oncol. 2020;38(15_suppl):8503–8503. https://doi.org/10.1200/JCO.2020.38.15_suppl.8503.
Gandhi UH, Cornell RF, Lakshman A, et al. Outcomes of patients with multiple myeloma refractory to CD38-targeted monoclonal antibody therapy. Leukemia. 2019;33(9):2266–75. https://doi.org/10.1038/s41375-019-0435-7.
Mehra M, Vogel M, Valluri S, Nair S, Schecter J. Patient characteristics, treatment patterns, and outcomes in patients with triple-class-refractory multiple myeloma. Presented at the: European Hematology Association annual congress; June 11, 2020. Virtual.
Jagannath S, Lin Y, Goldschmidt H, et al. KarMMa-RW: a study of real-world treatment patterns in heavily pretreated patients with relapsed and refractory multiple myeloma (RRMM) and comparison of outcomes to KarMMa. J Clin Oncol. 2020;38(15_suppl):8525–8525. https://doi.org/10.1200/JCO.2020.38.15_suppl.8525.
Kumar SK, Callander NS, Adekola K, Anderson, Jr LD, Baljevic M, Campagnaro E. Multiple myeloma NCCN guidelines. National Comprehensive Cancer Network. 2020. https://www.nccn.org/patients/guidelines/content/PDF/myeloma-patient.pdf. Accessed 17 Dec 2020.
Setoguchi S, Gerhard T. Chapter 5. comparator selection. Presented at the: developing a protocol for observational comparative effectiveness research: a user’s guide. Vol AHRQ Publication No. 12(13) EHC099. Agency for Healthcare Research and Quality; January 2013. Rockville, MD. http://www.effectivehealthcare.ahrq.gov/Methods-OCER.cfm. Accessed 17 Dec 2020.
Federal Coordinating Council for Comparative Effectiveness Research. Report to the President and Congress. Published online June 30, 2009. https://osp.od.nih.gov/wp-content/uploads/FCCCER-Report-to-the-President-and-Congress-2009.pdf. Accessed 17 Dec 2020.
Sox HC, Goodman SN. The methods of comparative effectiveness research. Annu Rev Public Health. 2012;33(1):425–45. https://doi.org/10.1146/annurev-publhealth-031811-124610.
Schaumberg DA, McDonald L, Shah S, Stokes M, Nordstrom BL, Ramagopalan SV. Evaluation of comparative effectiveness research: a practical tool. J Comp Eff Res. 2018;7(5):503–15. https://doi.org/10.2217/cer-2018-0007.
Guidance for researchers on optimal methods for conducting comparative effectiveness research with observational data. 2014. https://www.pcori.org/research-results/2013/guidance-researchers-optimal-methods-conducting-comparative-effectiveness. Accessed 18 Dec 2020.
Berger ML, Sox H, Willke RJ, et al. Good practices for real-world data studies of treatment and/or comparative effectiveness: recommendations from the joint ISPOR-ISPE special task force on real-world evidence in health care decision making. Value Health. 2017;20(8):1003–8. https://doi.org/10.1016/j.jval.2017.08.3019.
European Network of Centres for Pharmacoepidemiology and Pharmacovigilance. ENCePP guide on methodological standards in pharmacoepidemiology. 2020. http://www.encepp.eu/standards_and_guidances/methodologicalGuide.shtml. Accessed 18 Dec 2020.
Vandenbroucke JP, von Elm E, Altman DG, et al. Strengthening the reporting of observational studies in epidemiology (STROBE): explanation and elaboration. PLOS Med. 2007;4(10):e297. https://doi.org/10.1371/journal.pmed.0040297.
Dreyer NA, Bryant A, Velentgas P. The GRACE checklist: a validated assessment tool for high quality observational studies of comparative effectiveness. J Manag Care Spec Pharm. 2016;22(10):1107–13. https://doi.org/10.18553/jmcp.2016.22.10.1107.
Benchimol EI, Smeeth L, Guttmann A, et al. The REporting of studies conducted using observational routinely collected health data (RECORD) statement. PLoS Med. 2015;12(10):e1001885. https://doi.org/10.1371/journal.pmed.1001885.
Janssen Biotech, Inc. DARZALEX® (daratumumab) Approved by U.S. FDA: first human anti-CD38 monoclonal antibody available for the treatment of multiple myeloma. 2015. https://www.jnj.com/media-center/press-releases/darzalex-daratumumab-approved-by-us-fda-first-human-anti-cd38-monoclonal-antibody-available-for-the-treatment-of-multiple-myeloma. Accessed 15 Jan 2021.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group TP. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLOS Med. 2009;6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097.
Wang B-Y, Zhao W-H, Liu J, Chen Y-X, Cao X-M. Long-term follow-up of a phase 1, first-in-human open-label study of LCAR-B38M, a structurally differentiated chimeric antigen receptor T (CAR-T) cell therapy targeting B-cell maturation antigen (BCMA), in patients (pts) with relapsed/refractory multiple myeloma (RRMM). Presented at the: 61st ASH annual meeting; December 7, 2019. Orlando, FL.
Chen L, Xu J, Fu W, Jin S, Yang S. Updated phase 1 results of a first-in-human open-label study of LCAR-B38M, a structurally differentiated chimeric antigen receptor T (CAR-T) cell therapy targeting B-cell maturation antigen (BCMA). Presented at the: 61st ASH annual meeting; December 7, 2019. Orlando, FL.
Wang B-Y, Liu J, Zhao W-H, Chen Y-X, Cao X-M, Yang Y. Chimeric antigen receptor T cell therapy in relapsed or refractory multiple myeloma with extramedullary disease—a single institution observation in China. Presented at the: 62nd ASH Annual Meeting 2020; December 5, 2020. Virtual.
Zhao W-H, Liu J, Wang B-Y, Chen Y-X, Cao X-M, Yang Y. Updated analysis of a phase 1, open-label study of LCAR-B38M, a chimeric antigen receptor T cell therapy directed against B-cell maturation antigen, in patients with relapsed/refractory multiple myeloma. Presented at the: 60th ASH annual meeting; December 1, 2018. San Diego, CA, USA.
Mori Y, Choi I, Yoshimoto G, et al. Phase I/II study of bortezomib, lenalidomide, and dexamethasone treatment for relapsed and refractory multiple myeloma. Int J Hematol. 2020;111(5):673–80. https://doi.org/10.1007/s12185-020-02833-w.
Stewart AK, Rajkumar SV, Dimopoulos MA, et al. Carfilzomib, lenalidomide, and dexamethasone for relapsed multiple myeloma. N Engl J Med. 2015;372(2):142–52. https://doi.org/10.1056/NEJMoa1411321.
Palumbo A, Chanan-Khan A, Weisel K, et al. Daratumumab, bortezomib, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375(8):754–66. https://doi.org/10.1056/NEJMoa1606038.
Dimopoulos MA, Oriol A, Nahi H, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375(14):1319–31. https://doi.org/10.1056/NEJMoa1607751.
Dimopoulos M, Quach H, Mateos M-V, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, Phase 3 study. Lancet Lond Engl. 2020;396(10245):186–97. https://doi.org/10.1016/S0140-6736(20)30734-0.
Attal M, Richardson PG, Rajkumar SV, et al. Isatuximab plus pomalidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): a randomised, multicentre, open-label, Phase 3 study. Lancet Lond Engl. 2019;394(10214):2096–107. https://doi.org/10.1016/S0140-6736(19)32556-5.
Moreau P, Masszi T, Grzasko N, et al. Oral ixazomib, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;374(17):1621–34. https://doi.org/10.1056/NEJMoa1516282.
Krishnan A, Kapoor P, Palmer JM, et al. Phase I/II trial of the oral regimen ixazomib, pomalidomide, and dexamethasone in relapsed/refractory multiple myeloma. Leukemia. 2018;32(7):1567–74. https://doi.org/10.1038/s41375-018-0038-8.
Richardson PG, Oriol A, Beksac M, et al. Pomalidomide, bortezomib, and dexamethasone for patients with relapsed or refractory multiple myeloma previously treated with lenalidomide (OPTIMISMM): a randomised, open-label, Phase 3 trial. Lancet Oncol. 2019;20(6):781–94. https://doi.org/10.1016/S1470-2045(19)30152-4.
Zhou X, Steinhardt M, Grathwohl D, Meckel K, Nickel K. Multi-agent therapy with pomalidomide, bortezomib, doxorubicin, dexamethasone and daratumumab (Pom-PAD-Dara) in relapsed/ refractory multiple myeloma. Cancer Med. 2020;9(16):5819–26. https://doi.org/10.1002/cam4.3209.
Kim C, Braunlin M, Belani R, Wheeling JT. Progression free survival of relapsed or refractory multiple myeloma (RRMM) after failure of three prior lines of therapy in United States (US) real-world practice. Presented at the: 62nd ASH annual meeting and exposition; December 6, 2020. Virtual. https://ash.confex.com/ash/2020/webprogram/Paper140549.html. Accessed 18 Dec 2020.
Richardson PG, Jagannath S, Chari A, et al. Overall survival (OS) with oral selinexor plus low dose dexamethasone (Sd) in patients with triple class refractory-multiple myeloma (TCR-MM). J Clin Oncol. 2019;37(15_suppl):8014–8014. https://doi.org/10.1200/JCO.2019.37.15_suppl.8014.
Cornell R, Parameswaran H, Tang S, Barnstead A, Biran N. Real world vs. clinical trial outcomes of triple class refractory penta-exposed multiple myeloma (MM). Presented at the: 17th international myeloma workshop; September 12, 2019. Hynes Convention Center, Boston, MA, USA.
Shah N, Ayers D, Davies F, Cope S, Mojebi A. Matching-adjusted indirect comparison of efficacy outcomes for idecabtagene vicleucel (ide-cel, bb2121), a BCMA-directed CAR T cell therapy versus conventional care in triple-class exposed relapsed and refractory multiple myeloma. Presented at the: 62nd ASH annual meeting & exposition; December 5, 2020. Virtual.
Suvannasankha A, Kapetanakis V, Prawitz T, et al. MM-209: assessing efficacy via indirect comparison of single-agent belantamab mafodotin (Belamaf; GSK2857916) in DREAMM-2 versus STORM or MAMMOTH studies in relapsed/refractory multiple myeloma (RRMM). Clin Lymphoma Myeloma Leuk. 2020;20:S300–1. https://doi.org/10.1016/S2152-2650(20)30942-3.
Costa LJ, Hari P, Kumar SK, et al. Overall survival of triple class refractory, penta-exposed multiple myeloma (MM) patients treated with selinexor plus dexamethasone or conventional care: a combined analysis of the STORM and mammoth studies. Blood. 2019;134(Supplement_1):3125–3125. https://doi.org/10.1182/blood-2019-124991.
Caro JJ, Ishak KJ. No head-to-head trial? Simulate the missing arms. PharmacoEconomics. 2010;28(10):957–67. https://doi.org/10.2165/11537420-000000000-00000.
Signorovitch JE, Sikirica V, Erder MH, et al. Matching-adjusted indirect comparisons: a new tool for timely comparative effectiveness research. Value Health. 2012;15(6):940–7. https://doi.org/10.1016/j.jval.2012.05.004.
Ishak KJ, Proskorovsky I, Benedict A. Simulation and matching-based approaches for indirect comparison of treatments. PharmacoEconomics. 2015;33(6):537–49. https://doi.org/10.1007/s40273-015-0271-1.
Chari A, Vogl DT, Gavriatopoulou M, et al. Oral selinexor-dexamethasone for triple-class refractory multiple myeloma. N Engl J Med. 2019;381(8):727–38. https://doi.org/10.1056/NEJMoa1903455.
Lonial S, Lee HC, Badros A, et al. Belantamab mafodotin for relapsed or refractory multiple myeloma (DREAMM-2): a two-arm, randomised, open-label, phase 2 study. Lancet Oncol. 2020;21(2):207–21. https://doi.org/10.1016/S1470-2045(19)30788-0.
Acknowledgements
The authors thank Mr. Nicholas Adair (Panalgo LLC) for his assistance with manuscript preparation and literature searches.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research study was funded by Janssen Pharmaceuticals, Inc.
Conflict of interest
Nina Shah has received research funding from Bristol-Myers Squibb, Janssen, Bluebird Bio, Sutro Biopharma, Teneobio, Poseida, and Nektar, and has undertaken an advisory role with GSK, Amgen, Indapta Therapeutics, Sanofi, CareDx, Kite, Karyopharm, and Oncopeptides. Matthew Sussman and Jennifer Benner are employees of Panalgo LLC and were paid consultants in connection with the study. Concetta Crivera and Satish Valluri are employees of Johnson & Johnson (J & J) and have in excess of $10,000 of J & J stock. Sundar Jagannath is a consultant for Bristol-Myers Squibb, Karyopharm Therapeutics, Legend Biotech, Sanofi and Takeda.
Ethics approval
This study involved a systematic review of the literature and therefore did not require ethics approval.
Consent to participate
This study involved a systematic review of the literature and therefore did not require patient consent.
Consent for publication
Not applicable.
Availability of data and material
This study involved a systematic review of the literature; therefore, all data and materials are publicly available.
Code availability
Not applicable.
Appendices
Appendix 1: Search Criteria for Off-label Regimens
Bortezomib + lenalidomide + dexamethasone: ((bortezomib[mh] OR "bortezomib") AND ("lenalidomide" OR lenalidomide[mh]) AND ("dexamethasone" OR dexamethasone[mh]) AND (("relapsed" AND "refractory") AND ("multiple myeloma" OR multiple myeloma[mh]))
Ixazomib + pomalidomide + dexamethasone: ((ixazomib[Supplementary Concept] OR "ixazomib") AND ("pomalidomide" OR pomalidomide[Supplementary Concept]) AND ("dexamethasone" OR dexamethasone[mh]) AND (("relapsed" AND "refractory") AND ("multiple myeloma" OR multiple myeloma[mh]))
Pomalidomide + bortezomib + dexamethasone: ((bortezomib[mh] OR "bortezomib") AND ("pomalidomide" OR pomalidomide[Supplementary Concept]) AND ("dexamethasone" OR dexamethasone[mh]) AND (("relapsed" AND "refractory") AND ("multiple myeloma" OR multiple myeloma[mh]))
Appendix 2: Search Criteria for Full-text Real-world Data Evaluations of TCE or TCR Populations
Search criteria included: ("relapsed" OR "refractory" OR "triple-class" OR "quad-refractory" OR "penta-refractory") AND ("multiple myeloma" OR multiple myeloma[mh]) AND ("Progression-free survival" OR progression free survival[mh] OR "overall survival" OR "prognosis" OR "treatment patterns" OR "outcome" OR prognosis[mh] OR treatment outcome[mh]) AND (Observational study[mh] OR cohort studies[mh] OR "retrospective")
Rights and permissions
About this article
Cite this article
Shah, N., Sussman, M., Crivera, C. et al. Comparative Effectiveness Research for CAR-T Therapies in Multiple Myeloma: Appropriate Comparisons Require Careful Considerations of Data Sources and Patient Populations. Clin Drug Investig 41, 201–210 (2021). https://doi.org/10.1007/s40261-021-01012-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-021-01012-x