Abstract
Psoriasis is a chronic inflammatory skin disorder, which is associated with a significant negative impact on a patient’s quality of life. Traditional therapies for psoriasis are often not able to meet desired treatment goals, and high-dose and/or long-term use is associated with toxicities that can result in end-organ damage. An improved understanding of the involvement of cytokines in the etiology of psoriasis has led to the development of biologic agents targeting tumor necrosis factor (TNF)-α and interleukins (ILs)-12/23. While biologic agents have improved treatment outcomes, they are not effective in all individuals with psoriasis. The combination of biologic agents with traditional therapies may provide improved therapeutic options for patients who inadequately respond to a single drug or when efficacy may be increased with supplementation of another treatment. In addition, combination therapy may reduce safety concerns and cumulative toxicity, as lower doses of individual agents may be efficacious when used together. This article reviews the current evidence available on the efficacy and safety of combining biologic agents with systemic therapies (methotrexate, cyclosporine, or retinoids) or with phototherapy, and the combination of biologic agents themselves. Guidance is provided to help physicians identify situations and the characteristics of patients who would benefit from combination therapy with a biologic agent. Finally, the potential clinical impact of biologic therapies in development (e.g., those targeting IL-17A, IL-17RA, or IL-23 alone) is analyzed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Accumulating evidence supports the administration of biologic therapies in combination with systemic agents or phototherapy. |
Limited data exist on the co-administration of two biologics. |
Emerging, highly selective biologics may demonstrate the required efficacy to be administered as monotherapy. |
1 Introduction
Psoriasis is a chronic inflammatory skin disease, which affects approximately 3 % of the general population in the USA [1]. The most common form of the disease, plaque psoriasis, is characterized by the development of chronic erythematous plaques covered with silvery white scales, which most commonly appear on the elbows, knees, scalp, umbilicus, and lumbar regions [2]. Psoriasis has been associated with a significant negative impact on the patient’s quality of life, due to the disfiguring effect of the skin lesions and, for some, the functional impairment resulting from joint pain [3]. Additionally, individuals with psoriasis are more susceptible to specific debilitating comorbidities, including cardiometabolic dysfunction, fatigue, and depression [4–6].
The treatment strategy for psoriasis depends on a variety of factors (e.g., the medical history, tolerability of therapies and potential for side effects, and disease severity). Regarding disease severity, there is no commonly accepted definition of mild versus moderate-to-severe psoriasis [7]. Moreover, a patient may have mild disease on the basis of body surface area (BSA) involvement, but localization of lesions in vulnerable areas (e.g., the face, feet, hands, and/or genitals) may warrant systemic therapy. Some guidelines provide specific criteria to help evaluate the severity of a patient’s psoriasis, but all recognize the importance of assessing both the physical and psychosocial burden when considering the best treatment approach [7–10].
The US National Psoriasis Foundation recommends that patients with BSA involvement <5 % should be considered candidates for topical therapy, whereas those with BSA ≥5 % should be considered candidates for systemic therapy alone or in combination with phototherapy [9]. A “rule of tens” has also been proposed, whereby BSA >10 %, Psoriasis Area Severity Index (PASI) >10, or Dermatology Life-Quality Index (DLQI) >10 identify patients with severe disease [10]. More recently, a European consensus meeting defined mild psoriasis as BSA ≤10 %, PASI ≤10, and DLQI ≤10; and moderate-to-severe psoriasis warranting systemic therapy as BSA or PASI >10 and DLQI >10 [7]. The American Academy of Dermatology (AAD) guidelines present a treatment decision tree based on the presence or absence of psoriatic arthritis and categorization of psoriasis as “limited” or “extensive” disease, but specific definitions of these terms are not provided [8].
The ultimate goal of systemic therapy is to eliminate the systemic inflammatory burden of psoriasis and to completely clear the skin [7]. Historically, conventional systemic treatment options for psoriasis have included methotrexate, cyclosporine, and oral retinoids such as acitretin [11]. However, the use of these systemic agents has been limited by insufficient clinical efficacy, safety concerns, or both [7, 12, 13]. Cyclosporine is generally considered the most effective of these agents, providing a rapid response [14]. However, nephrotoxicity, hypertension, and numerous drug interactions may limit its use. Moreover, the duration of cyclosporine use is limited when it is prescribed for psoriasis (1 year in the USA, 2 years in the UK). The hepatotoxic effects of methotrexate necessitate particular caution when it is used in patients with liver problems or in those consuming large amounts of alcohol. Both methotrexate and retinoids are teratogenic [14].
None of these agents fully meets the needs of patients, and many are contraindicated because of the presence of comorbidities. Patient dissatisfaction with conventional systemic therapies has been well documented. Patients have voiced displeasure over inconvenient administration of traditional psoriasis therapies and their related side effects (e.g., hirsutism with cyclosporine, gastrointestinal intolerance with methotrexate, and hair loss and cheilitis with acitretin) [13]. Approximately 40 % of patients on systemic therapy alone have expressed dissatisfaction with their treatment outcomes [15], and overall patient satisfaction has been found to be lower with systemic therapy (cyclosporine, methotrexate, or acitretin) than with biologic agents, biologic/methotrexate combinations, or phototherapy [16]. Dissatisfaction with therapy is a major contributor to diminished adherence among patients with dermatologic disorders; inadequate treatment can therefore add to the already substantial burden of poor health-related quality of life associated with psoriasis [17, 18].
Over the past two decades, our understanding of the etiology of psoriasis has evolved; it is now recognized that both the innate and adaptive immune pathways are involved in its pathogenesis [19, 20]. Consequently, drugs that target specific components of the immune responses involved in the pathogenesis of psoriasis have been developed in an attempt to improve treatment efficacy, safety, and tolerability [21]. These agents include biologics that target cytokines such as tumor necrosis factor (TNF)-α and interleukins (ILs) 12/23 [21]. Despite remarkable improvements in psoriasis treatment outcomes with biologic therapy, however, many patients still do not achieve the desired outcome (Table 1) [22–30], have a prolonged time to response, or fail to maintain efficacy improvements over time. Tolerability may also be an issue (e.g., infections with TNF-α antagonists).
Combination systemic therapy may optimize treatment outcomes because of the potential of additive or synergistic efficacy. In addition, the dose of individual agents may be reduced, thereby decreasing toxicity and improving tolerability and compliance [31]. Up to 30 % of patients receiving a TNF-α antagonist also receive concomitant treatment with a traditional systemic agent such as methotrexate [32, 33]. Data are more limited with other drug combinations, including combinations with biologic agents [21, 31, 34]. In this paper, we review the rationale for the use of combination therapy in the management of psoriasis, along with evidence identified through a nonsystematic review of the literature that is currently available to support this practice. We also discuss new developments in the treatment of psoriasis, which may lessen the need for combination therapy to achieve desired outcomes.
2 Rationale for Combination Therapy
Some of the rationales for combining conventional therapies with biologic agents for psoriasis treatment have a historical basis in the treatment of psoriatic arthritis. In particular, methotrexate has been widely used in combination with biologic agents in clinical trials involving patients with psoriatic arthritis [35–39]. Indeed, the AAD guidelines recommend disease-modifying antirheumatic drugs (e.g., methotrexate), TNF-α antagonists, or a combination of these agents for moderate to severely active psoriatic arthritis [40]. The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) recommends that “a combination of two or more agents could be used in those patients who fail to respond to a single agent, or who present (with) joint damage progression in spite of treatment” [41]. The European League Against Rheumatism (EULAR) has made similar recommendations, but notes that there is a lack of robust evidence in psoriatic arthritis for this type of therapy [42].
The basis for these recommendations is rooted in the potential benefits that combination therapy may offer these patients, and can be extended to patients with psoriasis. These rationales include the potential efficacy synergies that may permit a more complete response and achieve a response more quickly, or both, as well as a potentially diminished risk of specific safety concerns that are caused by broad immunosuppressive therapy or that have been otherwise associated with conventional therapies (e.g., hepatotoxicity, nephrotoxicity, and bone marrow toxicity) [21, 43]. In addition, because the increased cytokine levels associated with psoriasis may also be associated with other inflammatory comorbid conditions, interventions targeting these upregulated cytokines may also provide a broader benefit to the patient. For example, a recent literature review found that methotrexate and TNF-α inhibitors may reduce cardiovascular events in individuals with psoriasis, although additional studies are required in this area [44]. Additionally, data from clinical trials evaluating adalimumab in rheumatoid arthritis demonstrate that patients on methotrexate had significantly higher blood levels of adalimumab than patients treated with adalimumab alone [30]. Thus, methotrexate, through an unknown mechanism, may boost drug levels of adalimumab. It is important to note that exposure to multiple drugs could also increase the risk of certain side effects, depending on the safety profiles of the individual agents being combined.
Combination therapies that include biologic agents may be particularly appropriate for a number of specific groups of patients with psoriasis (Table 2) [45–48]. The AAD guidelines do not provide specific recommendations in this regard [49]. The recent European consensus guidelines recommend that combination therapy should be considered for individuals who are switching to a biologic agent and in whom it may be useful to taper the previous systemic therapy before discontinuation to prevent a disease flare [32, 45, 50]. Additionally, patients with complications or comorbidities may benefit from the use of combination therapy with biologics. Patients with specific safety or toxicity concerns, such as methotrexate-related hepatotoxicity or cyclosporine-related nephrotoxicity, may also benefit from combination therapy with a biologic agent [47]. Additionally, combination therapy may help in the prevention or treatment of adverse events in certain patients (e.g., use of retinoids in a patient at risk of nonmelanoma skin cancer, or anti-TNF therapy in a patient with comorbid Crohn’s disease) [45, 51, 52]. Patients who may benefit from less rigorous treatment regimens with lower doses may also be candidates for combination therapy [53, 54]. In addition, there may be certain situations in which it would be appropriate to initiate combination therapy, such as at times when flares consistently occur (e.g., in the winter) or when a patient changes insurance and there are concerns about possible disruptions in therapy.
3 Clinical Experience with Combination Therapy
Although combination therapy with biologic agents has been widely used in the treatment of rheumatoid arthritis and psoriatic arthritis, only a few small-scale, randomized, controlled trials have been undertaken in patients with psoriasis [21, 31, 34, 48, 55]. In many cases, studies of combination therapy with biologic agents have been conducted against a background of treatment with conventional systemic therapies, such as methotrexate, in individuals without an adequate response to such treatment. Additionally, many of these studies were performed in patients with psoriatic arthritis; the effect of therapy on their psoriasis was also recorded as a secondary consideration [35–39]. Recently, the European consensus meeting developed recommendations for combining biologic and conventional systemic psoriasis therapies to provide some degree of structure for this practice; these recommendations are reported in Table 3 [50].
3.1 Combinations Involving Methotrexate
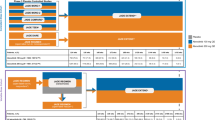
Initial findings of improved skin clearance in individuals with psoriatic arthritis after treatment with methotrexate and adalimumab led to further investigation of combined methotrexate and biologic therapy in psoriasis-specific populations [36]. In an open-label pilot study of 59 patients with active psoriasis (PASI ≥8, BSA >10 %, or both) without an adequate response to long-term (≥3 months) methotrexate therapy, patients were randomized to receive either etanercept and continued methotrexate or etanercept with methotrexate tapered and discontinued [56]. After 24 weeks, the proportion of patients with a Physicians’ Global Assessment (PGA) rating of “clear” or “almost clear” was significantly higher in the combination therapy group than in the etanercept monotherapy group (66.7 versus 37.0 %, respectively; P = 0.025). The same trend was also observed in the improvement of PASI scores by at least 75 % (PASI 75) at weeks 12 and 24. The adverse event rates were 75.0 % in the monotherapy group and 61.3 % in the combination group; adverse infectious events were the most frequent (25.0 and 38.7 %, respectively) [56]. The recent comparison of efficacy between etanercept and etanercept in combination with methotrexate by Gottlieb et al. [57] is one of the most robust combination trials to date in patients with moderate-to-severe psoriasis. Almost 45 % of patients in this trial had received prior systemic therapy, including methotrexate in about 17 % of patients. In this randomized, double-blind trial of 478 patients (BSA ≥10 %, PASI ≥10), PASI 75 response rates at 24 weeks were significantly higher with combination therapy than with etanercept alone (77.3 versus 60.3 %, respectively; P < 0.0001), as were other PASI response rates (Fig. 1) [57]. Combination therapy was also associated with a significant increase in the proportion of patients with a PGA rating of “clear” or “almost clear”, compared with etanercept alone (week 12: 65.5 versus 47.0 %; week 24: 71.8 versus 54.3 %; both P = 0.01). Adverse events were reported in 74.9 % of patients receiving combination therapy and in 59.8 % of those receiving etanercept monotherapy; the adverse event profiles of the two treatments were similar and included nasopharyngitis (9.6 % with combination therapy versus 10.9 % with monotherapy), headache (9.2 versus 9.2 %), and upper respiratory tract infection (8.4 versus 5.0 %).
Proportions of patients with moderate-to-severe plaque psoriasis showing improvements in the Psoriasis Area Severity Index of ≥50 % (PASI 50), ≥75 % (PASI 75), and ≥90 % (PASI 90) at 12 and 24 weeks during treatment with etanercept, alone or combined with methotrexate. Reproduced with permission from Gottlieb et al. [57]. © 2012 The Authors. BJD © 2012 British Association of Dermatologists
The combination of methotrexate and biologic agents also improves psoriasis in individuals without previous methotrexate therapy. The multicenter, randomized, open-label RESPOND trial (N = 115) evaluated the efficacy of methotrexate alone or in combination with infliximab in patients with psoriasis and psoriatic arthritis who had not previously received methotrexate [58]. The mean weekly dose of methotrexate was 15.4 mg in the monotherapy group and 14.6 mg in the combination group; the combination group received infliximab 5 mg/kg at weeks 0, 2, 6, and 14. The baseline PASI scores were relatively low compared with those in most psoriasis trials, with mean scores of approximately 11.6 in the monotherapy group and 8.3 in the combination group. Among patients with baseline PASI measurements of ≥2.5, the PASI 75 response rate at 16 weeks (a secondary outcomes measure) was 97.1 % with combination therapy and 54.3 % with methotrexate alone (P < 0.0001). Treatment-related adverse events occurred in 46 % of patients in the combination group and in 24 % of patients receiving methotrexate alone. The most common treatment-related adverse events were increased levels of hepatic enzymes.
Randomized trials investigating the combination of adalimumab and methotrexate are lacking in psoriasis, although positive results have been reported among patients with rheumatoid arthritis [59, 60]. The findings of the aforementioned randomized trials are further supported by numerous uncontrolled studies and case series that have shown beneficial effects of combinations of biologic therapies with methotrexate in patients with psoriasis or psoriatic arthritis [45, 61–76]. These studies have provided useful insights into the effectiveness and tolerability of combination therapy in routine clinical practice.
Wee et al. [72] retrospectively investigated the safety of infliximab infusions in 59 patients with psoriasis who received infliximab over a 9-year period at a single center in the UK. In this study, 56 % of patients were receiving concomitant systemic therapies; 41 % were receiving methotrexate [72]. Overall, acute infusion reactions were associated with 10 of 858 infliximab infusions (1.2 %), of which three (0.3 %) were severe. The incidence of infusion reactions was significantly lower in patients receiving infliximab with methotrexate than in those receiving infliximab alone (4 versus 27 %, respectively; P = 0.05), potentially as a result of decreased formation of anti-infliximab antibodies due to methotrexate. Another recent study (n = 45) investigated the effectiveness of combination therapy with methotrexate and adalimumab (11 treatment episodes) or adalimumab dose escalation (i.e., weekly dosing; 32 treatment episodes) in patients with psoriasis (a subset of approximately 25 % also had psoriatic arthritis) [74]. Patients were included in this study if they had an inadequate response, determined by the physician’s discretion, to standard adalimumab dosing. Combination therapy resulted in PASI 50 response rates of 9 % after 12 weeks and 18 % after 24 weeks; the corresponding values in patients with increased adalimumab doses were 25 and 35 %, respectively. The mean weekly dose of methotrexate used in this study was 9.5 mg per treatment episode. Adverse event rates were not reported in this trial; no serious adverse events were judged by the investigators to be related to the study medication. The study was too small to draw definitive conclusions but suggests that, at least for some patients, adalimumab dose escalation may be more beneficial than adding methotrexate.
3.2 Combinations Involving Cyclosporine
Cyclosporine therapy allows for regulation of the immune system through a different mechanism of action than current biologic agents, and their combination may improve control of lesion formation. The efficacy of therapy with cyclosporine and adalimumab was investigated in a nonrandomized, open-label study in patients with active psoriatic arthritis that was refractory to methotrexate treatment [77]. After 12 months, PASI 50 response criteria were met by 95 % of patients receiving combination therapy, compared with 85 % of patients receiving adalimumab alone and 65 % of those receiving cyclosporine alone (P = 0.003 versus combination treatment). In a small-scale, open-label study of patients with refractory psoriasis (n = 7), combination therapy with etanercept and low-dose cyclosporine (200 mg/day initially, then 100 mg/day) resulted in a mean reduction in PASI scores of 93.2 % at the end of the maintenance treatment period [78]. In addition, combinations of cyclosporine with biologic therapies have also been studied in a number of nonrandomized trials and case reports [45, 65–67, 79–82]. More rigorous studies are required to validate the safety and efficacy of these treatment regimens. Care must be taken when prescribing cyclosporine, because of concerns about nephrotoxicity, hypertension, and numerous drug interactions. Patients receiving cyclosporine in combination with other agents that suppress the immune system, such as TNF-α inhibitors, should be closely monitored for development of infections. Additionally, cyclosporine is not an option for long-term treatment of psoriasis, because of cumulative toxicity concerns.
3.3 Combinations Involving Retinoids
Individuals with psoriasis, many of whom have had significant phototherapy or excessive sun exposure, are at an increased risk of nonmelanoma skin cancers, such as squamous cell carcinoma and basal cell carcinoma [83]. To help reduce the risk of these types of cancer, oral retinoids may be given in combination with other systemic therapies. This method of treatment decreases the incidence of actinic keratosis and squamous cell carcinoma [31, 45, 84] but may also improve the underlying psoriasis. In a randomized, controlled, investigator-blinded pilot study, 60 patients with moderate-to-severe chronic plaque psoriasis were randomized to receive acitretin (0.4 mg/kg daily), etanercept (25 mg twice weekly), or the two agents in combination with reduced etanercept dosing (25 mg etanercept once weekly plus acitretin 0.4 mg/kg daily) [53]. At 24 weeks, a PASI 75 response was achieved in 30 % of patients receiving acitretin alone, compared with 45 % of those receiving etanercept alone and 44 % of those receiving combination therapy (P = 0.001 for both etanercept groups versus acitretin; Fig. 2) [53]. All treatments were well tolerated, and the only reported adverse event was mild mucosal dryness in two patients in the acitretin group and one patient in the combined group. No malignancies were reported.
Psoriasis Area Severity Index of ≥50 % (PASI 50) and ≥75 % (PASI 75) response rates at 24 weeks in patients with active plaque psoriasis treated with acitretin, etanercept twice weekly, or the two agents in combination with reduced use of etanercept (etanercept once weekly plus daily acitretin) [53]
Efficacious treatment of psoriasis using combinations of biologic therapies with retinoids has also been reported in a number of uncontrolled studies and case reports [45, 64, 66, 67, 85–90].
3.4 Phototherapy–Drug Combinations
Although phototherapy is not a pharmacologic intervention, it is an important treatment modality in the management of psoriasis. The combination of etanercept and narrow-band ultraviolet B (NB-UVB) or etanercept alone were studied in a trial of 75 patients with moderate-to-severe psoriasis that had not reached PASI 90 after 12 weeks of etanercept monotherapy [91]. The investigators observed that there were significant challenges to NB-UVB therapy adherence, with only 21.6 % of patients receiving ≥80 % of NB-UVB treatments. After 24 weeks, there was no significant difference in PASI 75 response rates between patients receiving etanercept monotherapy and those receiving the combination of etanercept and NB-UVB. However, in a small subset of patients with high adherence to NB-UVB therapy, PASI 75 response rates were found to be significantly improved as compared with patients receiving etanercept alone. Poor adherence to NB-UVB therapy was also observed by Park et al. [92] in an etanercept combination trial of obese patients with psoriasis. Interestingly, the combination of etanercept and NB-UVB did not lead to greater clearance of psoriasis than etanercept alone in these patients, and the investigators speculated that the poor adherence to NB-UVB therapy may have been due to patient satisfaction with the degree of psoriasis improvement from etanercept monotherapy. As with the other combination modalities, a number of uncontrolled clinical trials have shown positive results when NB-UVB therapy was combined with etanercept, adalimumab, and ustekinumab [93–99]. Thus, combinations of biologics and phototherapy may increase efficacy but are limited by adherence to therapy and concerns regarding the potential for skin cancer formation.
3.5 Combinations Involving Biologics
The combination of biologic agents has not been studied thoroughly in clinical trials, because of the relatively recent adoption of their use and concerns over blocking two pathways of the immune system. Thus, there are limited data available on the efficacy and safety of this type of therapy. A single case report has described successful treatment of psoriasis with adalimumab and ustekinumab in a patient who had not responded to combination therapy with methotrexate and ustekinumab [45]. However, the combination of biologic therapies did not improve this patient’s psoriatic arthritis. Physicians should proceed cautiously when considering the use of combinations of biologic agents; experience with this approach is very limited at this time and may have unknown consequences.
4 Clinical Implications and Unanswered Questions
Although the studies reviewed above have provided some evidence that therapies combining a conventional agent with a biologic are more effective than those agents used alone, and may be well tolerated in patients with psoriasis, there are still a number of questions that remain regarding the most appropriate use of this strategy [31]. Some of the trials involved patients with psoriatic arthritis and included those with psoriasis disease severity below the criteria for psoriasis trials, or had small sample sizes, or measured efficacy using inadequate or low efficacy endpoints (e.g., PASI 50). It is necessary to identify the safest and most effective combinations to limit potentially dangerous adverse events while achieving higher rates of skin clearance. In addition, the long-term safety of combination therapy is of particular concern; data are not available from controlled trials. Combination therapy with biologics may be beneficial in the management of comorbidities commonly found in patients with psoriasis. For instance, both TNF-α inhibitors and methotrexate reduce the risk of cardiovascular events, but it is unknown if their combination would produce additional benefits [44]. To fully understand the potential positive or negative influence of combination therapy on comorbidities, additional studies are required. Patients with psoriasis are also more likely to develop other health issues such as metabolic syndrome, which may be affected by combination therapy. TNF-α inhibitors may improve insulin resistance and fasting glucose levels, suggesting a possible beneficial role in managing metabolic syndrome, but other studies have reported increases in total cholesterol and high-density lipoprotein cholesterol following treatment [44]. Likewise, further studies are needed to elucidate the effect of combination therapy on metabolic syndrome. Combination therapy may be a more cost-effective method of managing psoriasis, but economic evaluations are needed to determine potential savings in healthcare costs. Disease management for patients with severe psoriasis could be improved, particularly if the findings to date with short-term use of phototherapy or traditional systemic agents in combination with biologic therapy are confirmed in the long term. Finally, it remains to be determined if biologic therapies can be used in combination without an increased safety risk.
5 Emerging Biologics
A number of new biologic therapies are currently in development for the treatment of psoriasis (Table 4) [100–103], including those that target IL-17A or its receptor, IL-23, and T cells [21]. It is not yet known how these therapies, many of which are in phase III clinical trials, will fit into the psoriasis treatment paradigm. These agents offer the potential for selective targeting of key processes in the pathogenesis of psoriasis. For example, compared with TNF-α, IL-17A is a cytokine that is downstream in the psoriasis pathogenesis pathway. As such, inhibiting this cytokine or its receptor may theoretically block psoriatic plaque formation without disrupting upstream cytokines that may be involved in other processes. Downstream blockade therefore has the potential to lessen the unwanted off-target effects associated with more upstream blockade. This selective targeting may also translate into a high level of efficacy, potentially reducing the need for combination therapy and offering patients a more convenient method to meet their treatment goals.
6 Conclusions
There is evidence demonstrating that combination therapy with biologic agents and conventional systemic therapies or phototherapy is effective and well tolerated in the management of moderate-to-severe psoriasis, although there are several limitations with respect to existing data (e.g., many psoriatic arthritis trials; patients often had less severe psoriasis; small studies; assessment of PASI 50 and not PASI 75 as the primary efficacy measure). Combination therapy offers the potential for improved treatment of patient subgroups in which currently available therapies may be of only limited benefit, such as patients with joint involvement or those at risk of end-organ toxicity with methotrexate, or for whom monotherapy has not yielded a desired benefit. Although combination therapy may improve treatment outcomes compared with individual monotherapy, the efficacy benefit from combination treatment can still remain below the desired target. The potential importance of higher treatment goals being achieved by monotherapy is highlighted by the emergence of new biologic therapies, such as IL-17A inhibitors. These agents selectively target key processes in the pathogenesis of psoriasis and thus may offer better efficacy than current biologic and systemic therapies. Therefore, these agents may allow more patients and prescribers to meet their psoriasis management goals without the need to augment treatment regimens with additional agents.
References
Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512–6.
Schön MP, Boehncke WH. Psoriasis. N Engl J Med. 2005;352:1899–912.
Kimball AB, Jacobson C, Weiss S, Vreeland MG, Wu Y. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383–92.
Kurd SK, Troxel AB, Crits-Christoph P, Gelfand JM. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891–5.
Evers AW, Lu Y, Duller P, van der Valk PG, Kraaimaat FW, van de Kerkhof PC. Common burden of chronic skin diseases? Contributors to psychological distress in adults with psoriasis and atopic dermatitis. Br J Dermatol. 2005;152:1275–81.
Onumah N, Kircik LH. Psoriasis and its comorbidities. J Drugs Dermatol. 2012;11:s5–10.
Mrowietz U, Kragballe K, Reich K, Spuls P, Griffiths CE, Nast A, et al. Definition of treatment goals for moderate to severe psoriasis: a European consensus. Arch Dermatol Res. 2011;303:1–10.
Menter A, Gottlieb A, Feldman SR, Van Voorhees AS, Leonardi CL, Gordon KB, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 1. Overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826–50.
Pariser DM, Bagel J, Gelfand JM, Korman NJ, Ritchlin CT, Strober BE, et al. National Psoriasis Foundation clinical consensus on disease severity. Arch Dermatol. 2007;143:239–42.
Finlay AY. Current severe psoriasis and the rule of tens. Br J Dermatol. 2005;152:861–7.
Hsu S, Papp KA, Lebwohl MG, Bagel J, Blauvelt A, Duffin KC, et al. Consensus guidelines for the management of plaque psoriasis. Arch Dermatol. 2012;148:95–102.
Krueger G, Koo J, Lebwohl M, Menter A, Stern RS, Rolstad T. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation patient-membership survey. Arch Dermatol. 2001;137:280–4.
Levy LL, Solomon SM, Emer JJ. Biologics in the treatment of psoriasis and emerging new therapies in the pipeline. Psoriasis Targets Ther. 2012;2:29–43.
Ladizinski B, Lee KC, Wilmer E, Alavi A, Mistry N, Sibbald RG. A review of the clinical variants and the management of psoriasis. Adv Skin Wound Care. 2013;26:271–84.
Nijsten T, Margolis DJ, Feldman SR, Rolstad T, Stern RS. Traditional systemic treatments have not fully met the needs of psoriasis patients: results from a national survey. J Am Acad Dermatol. 2005;52:434–44.
Callis Duffin K, Yeung H, Takeshita J, Krueger GG, Robertson AD, Troxel AB, et al. Patient satisfaction with treatments for moderate-to-severe plaque psoriasis in clinical practice. Br J Dermatol. 2014;170:672–80.
Renzi C, Picardi A, Abeni D, Agostini E, Baliva G, Pasquini P, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337–42.
Parrish L. Psoriasis: symptoms, treatments and its impact on quality of life. Br J Community Nurs. 2012;17:524–8.
Girolomoni G, Mrowietz U, Paul C. Psoriasis: rationale for targeting interleukin-17. Br J Dermatol. 2012;167:717–24.
Chiricozzi A, Krueger JG. IL-17 targeted therapies for psoriasis. Expert Opin Investig Drugs. 2013;22:993–1005.
Sivamani RK, Goodarzi H, Garcia MS, Raychaudhuri SP, Wehrli LN, Ono Y, et al. Biologic therapies in the treatment of psoriasis: a comprehensive evidence-based basic science and clinical review and a practical guide to tuberculosis monitoring. Clin Rev Allergy Immunol. 2013;44:121–40.
Reich K, Nestle FO, Papp K, Ortonne JP, Evans R, Guzzo C, et al. Infliximab induction and maintenance therapy for moderate-to-severe psoriasis: a phase III, multicentre, double-blind trial. Lancet. 2005;366:1367–74.
Menter A, Feldman SR, Weinstein GD, Papp K, Evans R, Guzzo C, et al. A randomized comparison of continuous vs. intermittent infliximab maintenance regimens over 1 year in the treatment of moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2007;56:31.e1–15.
Gottlieb AB, Evans R, Li S, Dooley LT, Guzzo CA, Baker D, et al. Infliximab induction therapy for patients with severe plaque-type psoriasis: a randomized, double-blind, placebo-controlled trial. J Am Acad Dermatol. 2004;51:534–42.
Leonardi CL, Powers JL, Matheson RT, Goffe BS, Zitnik R, Wang A, et al. Etanercept as monotherapy in patients with psoriasis. N Engl J Med. 2003;349:2014–22.
Papp KA, Tyring S, Lahfa M, Prinz J, Griffiths CE, Nakanishi AM, et al. A global phase III randomized controlled trial of etanercept in psoriasis: safety, efficacy, and effect of dose reduction. Br J Dermatol. 2005;152:1304–12.
Leonardi CL, Kimball AB, Papp KA, Yeilding N, Guzzo C, Wang Y, et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1). Lancet. 2008;371:1665–74.
Papp KA, Langley RG, Lebwohl M, Krueger GG, Szapary P, Yeilding N, et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 52-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 2). Lancet. 2008;371:1675–84.
Menter A, Tyring SK, Gordon K, Kimball AB, Leonardi CL, Langley RG, et al. Adalimumab therapy for moderate to severe psoriasis: a randomized, controlled phase III trial. J Am Acad Dermatol. 2008;58:106–15.
Humira (adalimumab) injection [prescribing information]. North Chicago: AbbVie Inc.; 2013.
Jensen P, Skov L, Zachariae C. Systemic combination treatment for psoriasis: a review. Acta Derm Venereol. 2010;90:341–9.
Warren RB, Brown BC, Carmichael AJ, Griffiths CE. Long-term control of recalcitrant psoriasis with combination infliximab and methotrexate. Clin Exp Dermatol. 2009;34:415–6.
Driessen RJ, Boezeman JB, van de Kerkhof PC, de Jong EM. Three-year registry data on biological treatment for psoriasis: the influence of patient characteristics on treatment outcome. Br J Dermatol. 2009;160:670–5.
Gustafson CJ, Watkins C, Hix E, Feldman SR. Combination therapy in psoriasis: an evidence-based review. Am J Clin Dermatol. 2013;14:9–25.
Daly M, Alikhan A, Armstrong AW. Combination systemic therapies in psoriatic arthritis. J Dermatolog Treat. 2011;22:276–84.
Mease PJ, Gladman DD, Ritchlin CT, Ruderman EM, Steinfeld SD, Choy EH, et al. Adalimumab for the treatment of patients with moderately to severely active psoriatic arthritis: results of a double-blind, randomized, placebo-controlled trial. Arthritis Rheum. 2005;52:3279–89.
Mease PJ, Gladman DD, Keystone EC. Alefacept in Psoriatic Arthritis Study Group. Alefacept in combination with methotrexate for the treatment of psoriatic arthritis: results of a randomized, double-blind, placebo-controlled study. Arthritis Rheum. 2006;54:1638–45.
McInnes IB, Kavanaugh A, Gottlieb AB, Puig L, Rahman P, Ritchlin C, et al. Efficacy and safety of ustekinumab in patients with active psoriatic arthritis: 1 year results of the phase 3, multicentre, double-blind, placebo-controlled PSUMMIT 1 trial. Lancet. 2013;382:780–9.
Mease PJ, Reich K. Alefacept in Psoriatic Arthritis Study Group. Alefacept with methotrexate for treatment of psoriatic arthritis: open-label extension of a randomized, double-blind, placebo-controlled study. J Am Acad Dermatol. 2009;60:402–11.
Gottlieb A, Korman NJ, Gordon KB, Feldman SR, Lebwohl M, Koo JY, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 2. Psoriatic arthritis: overview and guidelines of care for treatment with an emphasis on the biologics. J Am Acad Dermatol. 2008;58:851–64.
Ritchlin CT, Kavanaugh A, Gladman DD, Mease PJ, Helliwell P, Boehncke WH, et al. Treatment recommendations for psoriatic arthritis. Ann Rheum Dis. 2009;68:1387–94.
Gossec L, Smolen JS, Gaujoux-Viala C, Ash Z, Marzo-Ortega H, van der Heijde D, et al. European League Against Rheumatism recommendations for the management of psoriatic arthritis with pharmacological therapies. Ann Rheum Dis. 2012;71:4–12.
Lebwohl M. Combining the new biologic agents with our current psoriasis armamentarium. J Am Acad Dermatol. 2003;49:S118–24.
Hugh J, Van Voorhees AS, Nijhawan RI, Bagel J, Lebwohl M, Blauvelt A, et al. From the Medical Board of the National Psoriasis Foundation: The risk of cardiovascular disease in individuals with psoriasis and the potential impact of current therapies. J Am Acad Dermatol. 2014;70:168–77.
Heinecke GM, Luber AJ, Levitt JO, Lebwohl MG. Combination use of ustekinumab with other systemic therapies: a retrospective study in a tertiary referral center. J Drugs Dermatol. 2013;12:1098–102.
Daudén E, Castañeda S, Suárez C, García-Campayo J, Blasco AJ, Aguilar MD, et al. Clinical practice guideline for an integrated approach to comorbidity in patients with psoriasis. J Eur Acad Dermatol Venereol. 2013;27:1387–404.
Stebbins WG, Lebwohl MG. Biologics in combination with nonbiologics: efficacy and safety. Dermatol Ther. 2004;17:432–40.
Foley PA, Quirk C, Sullivan JR, Dolianitis C, Hack SP, Thirunavukkarasu K, et al. Combining etanercept with traditional agents in the treatment of psoriasis: a review of the clinical evidence. J Eur Acad Dermatol Venereol. 2010;24:1135–43.
Menter A, Korman NJ, Elmets CA, Feldman SR, Gelfand JM, Gordon KB, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 4. Guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009;61:451–85.
Mrowietz U, de Jong EM, Kragballe K, Langley R, Nast A, Puig L, et al. A consensus report on appropriate treatment optimization and transitioning in the management of moderate-to-severe plaque psoriasis. J Eur Acad Dermatol Venereol. 2014;28:438–53.
Otley CC, Stasko T, Tope WD, Lebwohl M. Chemoprevention of nonmelanoma skin cancer with systemic retinoids: practical dosing and management of adverse effects. Dermatol Surg. 2006;32:562–8.
Oh CJ, Das KM, Gottlieb AB. Treatment with anti-tumor necrosis factor α (TNF-α) monoclonal antibody dramatically decreases the clinical activity of psoriasis lesions. J Am Acad Dermatol. 2000;42:829–30.
Gisondi P, Del Giglio M, Cotena C, Girolomoni G. Combining etanercept and acitretin in the therapy of chronic plaque psoriasis: a 24-week, randomized, controlled, investigator-blinded pilot trial. Br J Dermatol. 2008;158:1345–9.
Famenini S, Wu JJ. Combination therapy with tumor necrosis factor inhibitors in psoriasis treatment. Cutis. 2013;92:140–7.
Bailey EE, Ference EH, Alikhan A, Hession MT, Armstrong AW. Combination treatments for psoriasis: a systematic review and meta-analysis. Arch Dermatol. 2012;148:511–22.
Zachariae C, Mork NJ, Reunala T, Lorentzen H, Falk E, Karvonen SL, et al. The combination of etanercept and methotrexate increases the effectiveness of treatment in active psoriasis despite inadequate effect of methotrexate therapy. Acta Derm Venereol. 2008;88:495–501.
Gottlieb AB, Langley RG, Strober BE, Papp KA, Klekotka P, Creamer K, et al. A randomized, double-blind, placebo-controlled study to evaluate the addition of methotrexate to etanercept in patients with moderate to severe plaque psoriasis. Br J Dermatol. 2012;167:649–57.
Baranauskaite A, Raffayová H, Kungurov NV, Kubanova A, Venalis A, Helmle L, et al. Infliximab plus methotrexate is superior to methotrexate alone in the treatment of psoriatic arthritis in methotrexate-naive patients: the RESPOND study. Ann Rheum Dis. 2012;71:541–8.
Hørslev-Petersen K, Hetland ML, Junker P, Pødenphant J, Ellingsen T, Ahlquist P, et al. Adalimumab added to a treat-to-target strategy with methotrexate and intra-articular triamcinolone in early rheumatoid arthritis increased remission rates, function and quality of life. The OPERA study: an investigator-initiated, randomised, double-blind, parallel-group, placebo-controlled trial. Ann Rheum Dis. 2014;73:654–61.
Burmester GR, Kivitz AJ, Kupper H, Arulmani U, Florentinus S, Goss SL, et al. Efficacy and safety of ascending methotrexate dose in combination with adalimumab: the randomised CONCERTO trial. Ann Rheum Dis. 2014. doi: 10.1136/annrheumdis-2013-204769. [Epub ahead of print].
Driessen RJ, van de Kerkhof PC, de Jong EM. Etanercept combined with methotrexate for high-need psoriasis. Br J Dermatol. 2008;159:460–3.
Yamauchi PS, Lowe NJ. Etanercept therapy allows the tapering of methotrexate and sustained clinical responses in patients with moderate to severe psoriasis. Int J Dermatol. 2008;47:202–4.
Strober BE. Successful treatment of psoriasis and psoriatic arthritis with etanercept and methotrexate in a patient newly unresponsive to infliximab. Arch Dermatol. 2004;140:366.
Strober BE, Clarke S. Etanercept for the treatment of psoriasis: combination therapy with other modalities. J Drugs Dermatol. 2004;3:270–2.
Iyer S, Yamauchi P, Lowe NJ. Etanercept for severe psoriasis and psoriatic arthritis: observations on combination therapy. Br J Dermatol. 2002;146:118–21.
Langewouters AM, van Erp PE, de Jong EM, van de Kerkhof PC. The added therapeutic efficacy and safety of alefacept in combination with other (systemic) anti-psoriatics in refractory psoriasis. J Dermatolog Treat. 2006;17:362–9.
Krueger GG, Gottlieb AB, Sterry W, Korman N, Van De Kerkhof P. A multicenter, open-label study of repeat courses of intramuscular alefacept in combination with other psoriasis therapies in patients with chronic plaque psoriasis. J Dermatolog Treat. 2008;19:146–55.
Kirby B, Marsland AM, Carmichael AJ, Griffiths CE. Successful treatment of severe recalcitrant psoriasis with combination infliximab and methotrexate. Clin Exp Dermatol. 2001;26:27–9.
Heikkilä H, Ranki A, Cajanus S, Karvonen SL. Infliximab combined with methotrexate as long-term treatment for erythrodermic psoriasis. Arch Dermatol. 2005;141:1607–10.
Takahashi MD, Castro LG, Romiti R. Infliximab, as sole or combined therapy, induces rapid clearing of erythrodermic psoriasis. Br J Dermatol. 2007;157:828–31.
Warren RB, Brown BC, Lavery D, Ashcroft DM, Griffiths CE. Biologic therapies for psoriasis: practical experience in a U.K. tertiary referral centre. Br J Dermatol. 2009;160:162–9.
Wee JS, Petrof G, Jackson K, Barker JN, Smith CH. Infliximab for the treatment of psoriasis in the U.K.: 9 years’ experience of infusion reactions at a single centre. Br J Dermatol. 2012;167:411–6.
Kamili QU, Miner A, Hapa A, Menter A. Infliximab treatment for psoriasis in 120 patients on therapy for a minimum of one year: a review. J Drugs Dermatol. 2011;10:539–44.
van den Reek JM, van Lümig PP, Kievit W, Zweegers J, van de Kerkhof PC, Seyger MM, et al. Effectiveness of adalimumab dose escalation, combination therapy of adalimumab with methotrexate, or both in patients with psoriasis in daily practice. J Dermatolog Treat. 2013;24:361–8.
Barland C, Kerdel FA. Addition of low-dose methotrexate to infliximab in the treatment of a patient with severe, recalcitrant pustular psoriasis. Arch Dermatol. 2003;139:949–50.
Dalaker M, Bonesrønning JH. Long-term maintenance treatment of moderate-to-severe plaque psoriasis with infliximab in combination with methotrexate or azathioprine in a retrospective cohort. J Eur Acad Dermatol Venereol. 2009;23:277–82.
Karanikolas GN, Koukli EM, Katsalira A, Arida A, Petrou D, Komninou E, et al. Adalimumab or cyclosporine as monotherapy and in combination in severe psoriatic arthritis: results from a prospective 12-month nonrandomized unblinded clinical trial. J Rheumatol. 2011;38:2466–74.
Lee EJ, Shin MK, Kim NI. A clinical trial of combination therapy with etanercept and low dose cyclosporine for the treatment of refractory psoriasis. Ann Dermatol. 2010;22:138–42.
Yamauchi PS, Lowe NJ. Cessation of cyclosporine therapy by treatment with etanercept in patients with severe psoriasis. J Am Acad Dermatol. 2006;54:S135–8.
Costanzo A, Talamonti M, Spallone G, Botti E, Chimenti MS, Papoutsaki M, et al. Efficacy of short-term cyclosporine treatment to control psoriasis-related events during efalizumab therapy. Dermatology. 2009;218:146–50.
Owen CM, Harrison PV. Successful treatment of severe psoriasis with basiliximab, an interleukin-2 receptor monoclonal antibody. Clin Exp Dermatol. 2000;25:195–7.
Rallis E, Verros CD. Ustekinumab treats psoriasis refractory to seven conventional and biologic therapies. Dermatol Online J. 2011;17:14.
Pouplard C, Brenaut E, Horreau C, Barnetche T, Misery L, Richard MA, et al. Risk of cancer in psoriasis: a systematic review and meta-analysis of epidemiological studies. J Eur Acad Dermatol Venereol. 2013;27(Suppl 3):36–46.
Nijsten TE, Stern RS. The increased risk of skin cancer is persistent after discontinuation of psoralen + ultraviolet A: a cohort study. J Invest Dermatol. 2003;121:252–8.
Conley J, Nanton J, Dhawan S, Pearce DJ, Feldman SR. Novel combination regimens: biologics and acitretin for the treatment of psoriasis—a case series. J Dermatolog Treat. 2006;17:86–9.
Adisen E, Karaca F, Gürer MA. When there is no single best biological agent: psoriasis and psoriatic arthritis in the same patient responding to two different biological agents. Clin Exp Dermatol. 2008;33:164–6.
Smith EC, Riddle C, Menter MA, Lebwohl M. Combining systemic retinoids with biologic agents for moderate to severe psoriasis. Int J Dermatol. 2008;47:514–8.
Gisondi P, Girolomoni G. Combination of efalizumab and acitretin in chronic plaque psoriasis. J Eur Acad Dermatol Venereol. 2008;22:247–8.
Ingram JR, Anstey AV, Piguet V. Combination treatment with a tumour necrosis factor antagonist and an oral retinoid: efficacy in severe acral psoriasis? Br J Dermatol. 2012;167:949–51.
Gallo E, Llamas-Velasco M, Daudén E, García-Diez A. Refractory generalized pustular psoriasis responsive to a combination of adalimumab and acitretin. Int J Dermatol. 2013;52:1610–1.
Lynde CW, Gupta AK, Guenther L, Poulin Y, Levesque A, Bissonnette R. A randomized study comparing the combination of nbUVB and etanercept to etanercept monotherapy in patients with psoriasis who do not exhibit an excellent response after 12 weeks of etanercept. J Dermatolog Treat. 2012;23:261–7.
Park KK, Wu JJ, Koo J. A randomized, ‘head-to-head’ pilot study comparing the effects of etanercept monotherapy vs. etanercept and narrowband ultraviolet B (NB-UVB) phototherapy in obese psoriasis patients. J Eur Acad Dermatol Venereol. 2013;27:899–906.
Kircik L, Bagel J, Korman N, Menter A, Elmets CA, Koo J, et al. Utilization of narrow-band ultraviolet light B therapy and etanercept for the treatment of psoriasis (UNITE): efficacy, safety, and patient-reported outcomes. J Drugs Dermatol. 2008;7:245–53.
Bagel J. Adalimumab plus narrowband ultraviolet B light phototherapy for the treatment of moderate to severe psoriasis. J Drugs Dermatol. 2011;10:366–71.
De Simone C, D’Agostino M, Capizzi R, Capponi A, Venier A, Caldarola G. Combined treatment with etanercept 50 mg once weekly and narrow-band ultraviolet B phototherapy in chronic plaque psoriasis. Eur J Dermatol. 2011;21:568–72.
Gambichler T, Tigges C, Scola N, Weber J, Skrygan M, Bechara FG, et al. Etanercept plus narrowband ultraviolet B phototherapy of psoriasis is more effective than etanercept monotherapy at 6 weeks. Br J Dermatol. 2011;164:1383–6.
Wolf P, Hofer A, Legat FJ, Bretterklieber A, Weger W, Salmhofer W, et al. Treatment with 311-nm ultraviolet B accelerates and improves the clearance of psoriatic lesions in patients treated with etanercept. Br J Dermatol. 2009;160:186–9.
Wolf P, Hofer A, Weger W, Posch-Fabian T, Gruber-Wackernagel A, Legat FJ. 311 nm ultraviolet B-accelerated response of psoriatic lesions in adalimumab-treated patients. Photodermatol Photoimmunol Photomed. 2011;27:186–9.
Wolf P, Weger W, Legat FJ, Posch-Fabian T, Gruber-Wackernagel A, Inzinger M, et al. Treatment with 311-nm ultraviolet B enhanced response of psoriatic lesions in ustekinumab-treated patients: a randomized intraindividual trial. Br J Dermatol. 2012;166:147–53.
Blauvelt A, Brown M, Gordon KB, Kavanaugh A, Leonardi CT, Stockfleth E, et al. Updates on psoriasis and cutaneous oncology: proceedings from the 2013 MauiDerm meeting. J Clin Aesthet Dermatol. 2013;6(9 Suppl):S2–20.
Tausend W, Downing C, Tyring S. Systematic review of interleukin-12, interleukin-17, and interleukin-23 pathway inhibitors for the treatment of moderate-to-severe chronic plaque psoriasis: ustekinumab, briakinumab, tildrakizumab, guselkumab, secukinumab, ixekizumab, and brodalumab. J Cutan Med Surg. 2014;18:156–69.
Deal watch: Boosting TRegs to target autoimmune disease. Nat Rev Drug Discov. 2011;10:566.
ClinicalTrials.gov. Safety and efficacy of multiple doses of BT061 in patients with moderate to severe chronic plaque psoriasis. http://clinicaltrials.gov/ct2/show/NCT01072383?term=nct01072383&rank=1. Accessed 22 May 2014.
Acknowledgments
Technical assistance with editing and styling of the manuscript for submission was provided by Oxford PharmaGenesis Inc. and was funded by Novartis Pharmaceuticals Corporation. The authors were fully responsible for all content and editorial decisions and received no financial support or other form of compensation related to the development of this manuscript. The opinions expressed in the manuscript are those of the authors and were not influenced by Novartis Pharmaceuticals Corporation.
Conflict of Interest
Dr. Cather has served as a speaker or consultant for AbbVie, Janssen, Leo, and Novartis. She has served as an investigator in clinical trials for Amgen, Celgene, Galderma, Lilly, Merck, Novartis, Pfizer, and Tolmar. She owns no stock and has no ownership interest in any pharmaceutical company.
Dr. Crowley has served as a speaker or consultant for, and has received research grants from, AbbVie and Amgen. He has served as a consultant for, and has received research support from, Celgene, Janssen, Lilly, and Pfizer. He has received research grants from Maruho, Merck, and Regeneron. He owns no stock and has no ownership interest in any pharmaceutical company.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Cather, J.C., Crowley, J.J. Use of Biologic Agents in Combination with Other Therapies for the Treatment of Psoriasis. Am J Clin Dermatol 15, 467–478 (2014). https://doi.org/10.1007/s40257-014-0097-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40257-014-0097-1